Summary
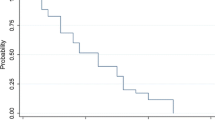
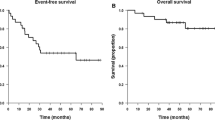
Sixteen patients who developed CT or MRI scan evidence of recurrent diffuse astrocytoma after radiation therapy and nitrosourea-containing chemotherapy received ifosfamide (2500 mg/m2/day for 3 consecutive days) and mesna (500 mg/m2/dose, 5 doses/day for 3 consecutive days). Toxicity consisted primarily of leukopenia in that 60 percent of patients developed leukocyte nadirs less than 1500/mcL. Excessive somnolence occurred in three patients and may have contributed to a case of fatal pneumonia in one patient but was reversible in the other two. No patient had CT or MRI scan evidence of tumor regression. One patient remains stable at 11.3+ months, but all other patients developed evidence of progressive disease less than 6 months from initiation of therapy. The median times to tumor progression and death were 2.0 and 4.8 months, respectively. In conclusion, while ifosfamide and mesna can be given safely at this dose and schedule, there is no evidence of antitumor effect. The degree of leukopenia observed likely would prevent further dose escalation of ifosfamide or addition of other myelosuppressive agents without additional means of bone marrow support in this population of patients.
Similar content being viewed by others
References
Brook N, Pohl J: The development of mesna for regional detoxification. Cancer Treat Rev 10 (Supplement A): 33–43,] 1983
Pratt CB, Horowitz ME, Meyer WH, Etcubanas E, Thompson EI, Douglass EC, Wilimas JAS, Hayes FA, Green AA: Phase II trial of ifosfamide in children with malignant solid tumors. Cancer Treat Rep 71: 131–135, 1987
Miser JS, Kinsella TJ, Triche TJ, Tsokos M, Jarosinski P, Forquer R, Wesley R, Magrath I: Ifosfamide with mesna uroprotection and etoposide: An effective regimen in the treatment of recurrent sarcomas and other tumors of children and young adults. J Clin One 5: 1191–1198, 1987
Lauer RC, Roth B, Loehrer PJ, Einhorn LH, Williams S: Cisplatin and ifosfamide and either VP-16 or vinblastine (VIP) as third line therapy for metastatic testicular cancer. (Abstract) Proc Am Soc Clin One 6: 99, 1987
Ninane J, Baurain R, de Kraker J, Cornu G: Pharmacokinetics of ifosfamide in childeren. Minutes of SIOP Meeting., 1986, Budapest, Hungary, pp 224–225
Kornblith PL, Walker MD, Cassady JR: Neurologic Oncology. JB Lippincott Company, Philadelphia, 1987, pp 130–149
Daumus-Duport C, Scheithauer B, O'Fallon J, Kelly P: Grading of astrocytomas: A simple and reproducible grading method. Cancer 62: 2152–2165, 1988
Miser J, Krailo M, Smithson W, Belasco J, Ortega J, Hammond GD: Treatment of children with recurrent brain tumors with ifosfamide (IFOS), etoposide (VP16), and mesna (M). Results of a Phase 11 trial. (Abstract) Proc Am Soc Clin One 8: 84, 1989
Finn GP, Bozek T, Souhami RL, Slevin ML, Thomas DG: High-dose etoposide in the treatment of relapsed primary brain tumors. Cancer Treat Rep 69: 603–605, 1985
Feun LG, Lee YY, Yung WK, Savaraj N, Wallace S: Intracarotid VP-16 in malignant brain tumors. J Neurooncol 4: 397–401, 1987
Tirelli U, D'Incalci M, Canetta R, Tumulo S, Franchin G, Veronesi A, Galligiono S, Trovo M, Rossi C, Grigoletto E: Etoposide (VP-16–213) in malignant brain tumors: A phase II study. J Clin Oncol 2: 432–437, 1984
Giannone L, Wolff SN: Phase II treatment of central nervous system gliomas with high-dose etoposide and autologous bone marrow transplantation. Cancer Treat Rep 71: 759–761,1987
Author information
Authors and Affiliations
Additional information
Additional participating institutions include: Geisinger Clinic and Medical Center CCOP, Danville, Pennsylvania 17822 (Richard M. Goldberg); University of Nebraska Medical Center, Omaha, Nebraska 68105 (John F. Foley); Illinois Oncology Research Association CCOP, Peoria, Illinois 61603 (James B. Gerstner); and The St. Cloud Clinic of Internal Medicine, Ltd., St. Cloud, Minnesota 56301 (John Weitz).
Rights and permissions
About this article
Cite this article
Elliott, T.E., Buckner, J.C., Cascino, T.L. et al. Phase II study of ifosfamide with mesna in adult patients with recurrent diffuse astrocytoma. J Neuro-Oncol 10, 27–30 (1991). https://doi.org/10.1007/BF00151244
Issue Date:
DOI: https://doi.org/10.1007/BF00151244