Abstract
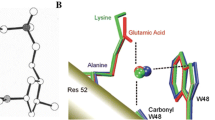
Over the last two decades, studies on lone pair-π interaction have attracted lot of attention of experimental as well as theoretical chemists due to its intriguing nature and its suspected presence in biological systems. The present Chapter begins with a brief overview of the earlier theoretical and experimental work done in this area. This is followed by exploration of the nuances of bonding in lone pair-π interaction, employing the tool of molecular electrostatic potential (MESP) since such weak interactions are mainly dominated by electrostatic features of host and guest molecules. The critical points associated with the scalar field of MESP are exploited for scrutinizing the directionality and bonding sites involved in the lone pair-π complexes. Furthermore, the electrostatic potential for intermolecular complexation (EPIC) model developed by Gadre et al., has been employed for finding out the electrostatically optimized structures and interaction energies of these complexes. The outcomes of EPIC model are compared with the results obtained from quantum chemical calculations of the complexes employing M06L/6-311++G(d,p) level of theory. The present study details out four different cases of lone pair-π complexes, which are currently in vogue. Hexafluorobenzene, one of the most explored π-deficient host in the present context, is initially taken up to demonstrate various facets of MESP for gaining insights into this interaction. This is followed by the scrutiny of special classes of recently synthesized highly π-deficient molecules, viz. tetraoxacalix [2]arene[2]triazine and naphthalenediimide, which are known to have specificity and large affinity, respectively, towards the electron rich species. The chapter ends with the description of lone pair-π interaction in the case of urate oxidase, an enzyme present in biological systems.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Némethy G, Scheraga HA (1962) J Chem Phys 36:3382–3400
Tzalis D, Tor Y (1996) Tetrahedron Lett 37:8293 – 8296
Müller-Dethlefs K, Hobza P (2000) Chem Rev 100:143–168
Duncan R, Kopeček J (1984) Soluble synthetic polymers as potential drug carriers. In: Polymers in medicine, vol. 57.Springer, Berlin
Huck WTS, Prins LJ, Fokkens RH, Nibbering NMM, van Veggel FCJM, Reinhoudt DN (1998) J Am Chem Soc 120:6240–6246
Zhong W, Gallivan JP, Zhang Y, Li L, Lester HA, Dougherty DA (1998) Proc Nat Acad Sci USA 95:12088–12093
Mecozzi S, West AP, Dougherty DA (1996) J Am Chem Soc 118:2307–2308
McCurdy A, Jimenez L, Stauffer DA, Dougherty DA (1992) J Am Chem Soc 114:10314–10321
Wheeler SE, Houk KN (2009) J Am Chem Soc 131:3126–3127
Sayyed FB, Suresh CH (2012) J Phys Chem A 116:5723–5732
Sayyed FB, Suresh CH (2012) Chem Phys Lett 523:11–14
Sayyed FB, Suresh CH (2011) J Phys Chem A 115:9300–9307
Mahadevi AS, Sastry GN (2013) Chem Rev 113:2100–2138
Egli M, Sarkhel S (2007)Acc Chem Res 40:197–205
Caltagirone C, Gale PA (2009) Chem Soc Rev 38:520–563
Wenzel M, Hiscock JR, Gale PA (2012) Chem Soc Rev 41:480–520
Gale PA, Busschaert N, Haynes CJE, Karagiannidis LE, Kirby IL (2014) Chem Soc Rev 43:205–241
Schottel BL, Chifotides HT, Dunbar KR (2008) Chem Soc Rev 37:68–83
Chifotides HT, Dunbar KR (2013) Acc Chem Res 46:894–906
Frontera A, Gamez P, Mascal M, Mooibroek TJ, Reedijk J (2011) Angew Chem Int Ed 50:9564–9583
Mooibroek TJ, Black CA, Gamez P, Reedijk J (2008) Cryst Growth Des 8:1082–1093
Quiñonero D, Garau C, Rotger C, Frontera A, Ballester P, Costa A, Deyà PM (2002) Angew Chem Int Ed 41:3389–3392
Boden N, Davis P, Stam C, Wesselink G (1973) Mol Phys 25:81–86
Vrbancich J, Ritchie GLD (1980) J Chem Soc Faraday Trans 2 76:648–659
Schneider H-J, Werner F, Blatter T (1993) J Phys Org Chem 6:590–594
Schneider H-J, Blatter T, Palm B, Pfingstag U, Ruediger V, Theis I (1992) J Am Chem Soc 114:7704–7708
Schneider H-J (1991) Angew Chem Int Ed 30:1417–1436
Alkorta I, Rozas I, Elguero J (1997) J Org Chem 62:4687–4691
Alkorta I, Rozas I, Elguero J (2002) J Am Chem Soc 124:8593–8598
Gallivan JP, Dougherty DA (1999) Org Lett 1:103–106
Alkorta I, Elguero J (2003) J Phys Chem A 107:9428–9433
Garau C, Frontera A, Quiñonero D, Ballester P, Costa A, Deyà PM (2003) Chem Phys Chem 4:1344–1348
Estarellas C, Frontera A, Quiñonero D, Deyà PM (2011) Chem Asian J 6:2316–2318
Estarellas C, Frontera A, Quiñonero D, Deyà PM (2011) Angew Chem Int Ed 50:415–418
Jeziorski B, Moszynski R, Szalewicz K (1994) Chem Rev 94:1887–1930
Lao KU, Herbert JM (2014) J Chem Phys 140:044108–8
Kim D, Tarakeshwar P, Kim KS (2004) J Phys Chem A 108:1250–1258
Demeshko S, Decher S, Meyer F (2004) J Am Chem Soc 126:4508–4509
de Hoog P, Gamez P, Mutikainen I, Turpeinen U, Reedijk J (2004) Angew Chem Int Ed 43:5815–5817
Frohn H, Giesen M, Welting D, Henkel G (1996) Eur J Solid State Inorg Chem 33:841–853
Sessler JL, Gale PA, Cho W-S (2006) Anion receptor chemistry, vol 8. Royal Society of Chemistry
Rosokha YS, Lindeman SV, Rosokha SV, Kochi JK (2004) Angew Chem Int Ed 43:4650–4652
Müller M, Albrecht M, Gossen V, Peters T, Hoffmann A, Raabe G, Valkonen A, Rissanen K (2010) Chem Eur J 16:12446–12453
Li S, Wang D-X, Wang M-X (2012) Tetrahedron Lett 53:6226–6229
Arranz-Mascarós P, Bazzicalupi C, Bianchi A, Giorgi C, Godino-Salido M-L, Gutiérrez-Valero M-D, Lopez-Garzón R, Savastano M (2013) J Am Chem S 135:102–105
Giese M, Albrecht M, Krappitz T, Peters M, Gossen V, Raabe G, Valkonen A, Rissanen K (2012) Chem Commun 48:9983–9985
Ballester P (2013) Acc Chem Res 46:874–884
Gil-Ramírez G, Escudero-Adán EC, Benet-Buchholz J, Ballester P (2008) Angew Chem 120:4182–4186
Schottel BL, Chifotides HT, Shatruk M, Chouai A, Pérez LM, Bacsa J, Dunbar KR (2006) J Am Chem Soc 128:5895–5912
Mareda J, Matile S (2009) Chem Eur J 15:28–37
Zhao Y, Domoto Y, Orentas E, Beuchat C, Emery D, Mareda J, Sakai N, Matile S (2013) Angew Chem Int Ed 52:9940–9943
Gorteau V, Julliard MD, Matile S (2008) J Membr Sci 321:37–42
Vargas Jentzsch A, Emery D, Mareda J, Metrangolo P, Resnati G, Matile S (2011) Angew Chem Int Ed 50:11675–11678
Lin N-T, Vargas Jentzsch A, Guenee L, Neudorfl J-M, Aziz S, Berkessel A, Orentas E, Sakai N, Matile S (2012) Chem Sci 3:1121–112
Berman HM, Westbrook J, Feng Z, Gilliland G, Bhat TN, Weissig H, Shindyalov IN, Bourne PE (2000) Nucleic Acids Res 28:235–242
Scrocco E, Tomasi J (1973) The electrostatic molecular potential as a tool for the interpretation of molecular properties. In New concepts II, vol 42. Springer, Berlin, pp 95–170
Tomasi J, Mennucci B, Cammy M (1996) Molecular electrostatic potentials: concepts and applications. Elsevier, Amsterdam
Tomasi J, Mennucci B, Cammi R (1996) Theor Comp Chem 3:1–103
Politzer P, Murray JS (2002) Theor Chem Acc 108:134–142
Yeole SD, Gadre SR (2011) J Phys Chem A 115:12769–12779
Balanarayan P, Kavathekar R, Gadre SR (2007) J Phys Chem A 111:2733–2738
Gadre SR, Shirsat RN (2000) Electrostatics of atoms and molecules. Universities Press, Hyderabad
Gadre SR, Kulkarni SA, Shrivastava IH (1992) J Chem Phys 96:5253–5260
Balanarayan P, Gadre SR (2003) J Chem Phys 119:5037–5043
Shirsat RN, Bapat SV, Gadre SR (1992) Chem Phys Lett 200:373–378
Politzer P, Murray JS, Peralta-Inga Z (2001) Int J Quantum Chem 85:676–684
Politzer P, Landry SJ, Waernheim T (1982) J Phys Chem 86:4767–4771
Murray JS, Lane P, Politzer P (2007) Int J Quantum Chem 107:2286–2292
Politzer P, Murray JS, Lane P (2007) Int J Quantum Chem 107:3046–3052
Shields ZP, Murray JS, Politzer P (2010) Int J Quantum Chem 110:2823–2832
Politzer P, Murray JS, Clark T (2010) Phys Chem Chem Phys 12:7748–7757
Suresh CH, Koga N, Gadre SR (2000) Organometallics 19:3008–3015
Elango M, Subramanian V, Rahalkar AP, Gadre SR, Sathyamurthy N (2008) J Phys Chem A 112:7699–7704
Kumar A, Gadre SR, Mohan N, Suresh CH (2014) J Phys Chem A 118:526–532
Mohan N, Suresh CH, Kumar A, Gadre SR (2013) Phys Chem Chem Phys 15:18401–18409
Suresh CH, Gadre SR (2007) J Phys Chem A 111:710–714
Suresh CH, Alexander P, Vijayalakshmi KP, Sajith P, Gadre SR (2008) Phys Chem Chem Phys 10:6492–6499
Pullman B (1990) Int J Quantum Chem 38:81–92
Pichon-Pesme V, Lecomte C (1998) Acta Crystallogr Sect B 54:485–493
Benabicha F, Pichon-Pesme V, Jelsch C, Lecomte C, Khmou A (2000) Acta Crystallogr Sect B 56:155–165
Bouhmaida N, Ghermani N-E, Lecomte C, Thalal A (1997) Acta Crystallogr Sect A 53:556–563
Bouhmaida N, Thalal A, Ghermani Ne, Lecomte C (1999) Acta Crystallogr Sect A 55:729–738
Gadre SR, Shrivastava IH (1991) J Chem Phys 94:4384–4390
Gadre SR, Pathak RK (1990) Proc Ind Acad Sci (Chem Sci) 102:189–192
Gadre SR, Pundlik SS (1997) J Phys Chem B 101:3298–3303
Pundlik SS, Gadre SR (1997) J Phys Chem B 101:9657–9662
Pingale SS, Gadre SR, Bartolotti LJ (1998) J Phys Chem A 102:9987–9992
Sivanesan D, Babu K, Gadre SR, Subramanian V, Ramasami T (2000) J Phys Chem A 104:10887–10894
Zhao Y, Truhlar DG (2006) J Chem Phys 125:194101–194118
Frisch MJ et al (2009) Gaussian09 Revision D.01. Gaussian Inc. Wallingford
Boys S, Bernardi F (1970) Mol Phys 19:553–566
Gadre SR, Kulkarni SA, Suresh C, Shrivastava IH (1995) Chem Phys Lett 239:273–281
Yeole SD, López R, Gadre SR (2012) J Chem Phys 137:074116–074117
Rico JF, López R, Ramírez G, Ema I, Ludeñ EV (2004) J Comput Chem 25:1355–1363
Yeole SD, Gadre SR (2011) J Chem Phys 134:084111–084118
Wang D-X, Wang M-X (2013) J Am Chem Soc 135:892–897
Acknowledgements
Authors are thankful to Dr. C. H. Suresh and Dr. P. Balanarayan for fruitful discussions. Anmol Kumar thanks the Council of Scientific and Industrial Research (CSIR) for research fellowship. Professor Shridhar Gadre is grateful to the Department of Science and Technology (DST), New Delhi for the award of J. C. Bose National Fellowship.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Gadre, S., Kumar, A. (2015). Understanding Lone Pair-π Interactions from Electrostatic Viewpoint. In: Scheiner, S. (eds) Noncovalent Forces. Challenges and Advances in Computational Chemistry and Physics, vol 19. Springer, Cham. https://doi.org/10.1007/978-3-319-14163-3_13
Download citation
DOI: https://doi.org/10.1007/978-3-319-14163-3_13
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-14162-6
Online ISBN: 978-3-319-14163-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)