Abstract
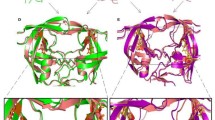
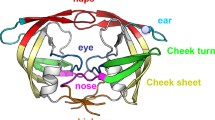
The aspartyl proteinase encoded within the genome of the type I human immunodeficiency virus (HIV-1 PR) is a valid and important target for the development of a therapeutic to treat HIV infections. Progress in this area has been rapid due to 1) the wealth of previous experience with other aspartyl proteinases and 2) the massive commitment by a large number of research groups worldwide. In this chapter we would like to discuss some of our efforts to develop a PR inhibitor by describing the structure of a complex between HIV-1 PR and a hydroxyethylene inhibitor. As a final note before continuing, we would like to acknowledge the significant contributions that Alex Wlodawer and his group have made to this field which have been important to the progress made not only by our group but by many others as well.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
M. Szelke, B. J. Leckie, A. Hallett, D. M. Jones, J. Sueiras, B. Atrash and A. F. Lever, Potent New Inhibitors of Human Renin, Nature 299: 555 (1982).
J. G. Dann, D. K. Stammers, C. J. Harris, R. J. Arrowsmith, D. E. Davies, G. W. Hardy and J. A. Morton, Human Renin: A New Class of Inhibitors, Biochem. Biophys. Res. Commun. 134: 71 (1986).
M. Szelke, D. M. Jones, B. Atrash, A. Hallett and B. J. Leckie, Novel Transition-State Analogue Inhibitors of Renin, in: “Peptides: Structure and Function, Proceedings of the 8th American Symposium,” V. J. Hruby and D. H. Rich, Eds., Pierce Chemical, Rockford, IL, (1983).
M. Miller, J. Schneider, B. K. Sathyanarayana, M. V. Toth, G. R. Marshall, L. Clawson, L. Selk, S. B. H. Kent and A. Wlodawer, Structure of Complex of Synthetic HIV-1 Protease with a Substrate-Based Inhibitor at 2.3 A Resolution, Science 246: 1149 (1989).
A. L. Swain, M. M. Miller, J. Green, D. H. Rich, J. Schneider, S. B. H. Kent and A. Wlodawer, X-ray Crystallographic Structure of a Complex Between a Synthetic Protease of Human Immunodeficiency Virus 1 and a Subtrate-based Hydroxyethylamine Inhibitor, Proc. Nat. Acad. Sci., U.S.A. 87: 8805 (1990).
P. M. D. Fitzgerald, B. M. McKeever, J. F. VanMiddlesworth, J. P. Springer, J. C. Heimbach, C.-T. Leu, W. K. Herber, R. A. F. Dixon and P. L. Darke, Crystallographic Analysis of a Complex Between HIV-1 Protease and Acetyl-Pepstatin at 2.0 Å Resolution, J. Biol. Chem. 265: 14209 (1990).
J. Erickson, D. J. Neidhart, J. VanDrie, D. J. Kempf, X. C. Wang, D. W. Norbeck, J. J. Plattner, J. W. Rittenhouse, M. Turon, N. Wideburg, W. E. Kohlbrenner, R. Simmer, R. Helfrich, D. A. Paul and M. Knigge, Design, Activity and 2.8 Å Crystal Structure of a C2 Symmetric Inhibitor Complexed to HIV-1 Protease, Science 249: 527 (1990).
P. M. D. Fitzgerald, MERLOT, An Integrated Package of Computer Programs for the Determination of Crystal Structures by Molecular Replacement, J. Appl. Crystallogr. 21: 273 (1988).
A. Wlodawer, M. Miller, M. Jaskólski, B. K. Sathyanarayana, E. Baldwin, I. T. Weber, L. M. Selk, L. Clawson, J. Schneider and S. B. H. Kent, Conserved Folding in Retroviral Proteases: Crystal Structure of a Synthetic HIV-1 Protease, Science 245: 616 (1989).
T. J. McQuade, A. G. Tomasselli, L. Liu, V. Karacostas, B. Moss, T. K. Sawyer, R. L. Heinrikson and W. G. Tarpley, A Synthetic HIV-1 Protease Inhibitor with Antiviral Activity Arrests HIV-like Particle Maturation, Science 247: 454 (1990).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1991 Plenum Press, New York
About this chapter
Cite this chapter
Graves, B.J. et al. (1991). The Three-Dimensional X-Ray Crystal Structure of HIV-1 Protease Complexed with a Hydroxyethylene Inhibitor. In: Dunn, B.M. (eds) Structure and Function of the Aspartic Proteinases. Advances in Experimental Medicine and Biology, vol 306. Springer, Boston, MA. https://doi.org/10.1007/978-1-4684-6012-4_58
Download citation
DOI: https://doi.org/10.1007/978-1-4684-6012-4_58
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4684-6014-8
Online ISBN: 978-1-4684-6012-4
eBook Packages: Springer Book Archive