Abstract
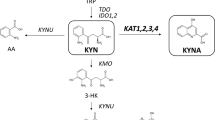
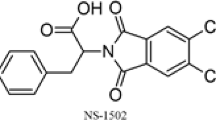
Chirality, a phenomenon of dissimetry, is a fundamental characteristic of biological systems. A large amount of endogenous compounds including drug-interacting macromulecules are chiral, thus giving rise to differences in the intermolecular energy involved in the interaction of the chiral drug and the biological target. As a result of such chiral discrimination, drug enantiomers may differ in their pharmacodynamic and pharmacokinetic effects. Due to such differences, particularly in vivo the use of 1:1 mixture of enantiomers (racemates), often fails to display, and occasionally masks the properties of each enantiomer. In searching of selective inhibitors of tryptophane catabolism enzymes, we identified two classes of potent and selective inhibitors represented by 2’-methoxy-benzoylalanine (R,S)-1 and 3’, 4’-dichloro-benzoylalanine (R,S)-2, acting respectively as kynureninase and kynuren- ine 3-hydroxylase selective inhibitors with no activity on the kynurenic acid synthesising enzyme (Kynurenine aminotransferase). Moreover, it is noteworthy that the unsubstituted benzoyl-alanine (R, S)-3, even though endowed with lower potency and selectivity, is by itself an inhibitor of kynurenine-3-hydroxylase.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Reference
Melillo D.G., Larsen R.D., Mathre D.J., Shukis W.F., Wood A.W., and Colleluori J.R., 1987, Pratical enantioselective synthesis of a homotyrosine derivative and (R,R)-4-propyl-9-hydroxynaphtoxazine, a potent dopamine agonist, J.Org.Chem.52: 5143.
Nordlander D.S., Payne M.J., Njorge EG., Vishwanath V.M., Han G.R., Laikos G.D., and Balk M.A., 1985, A short enantiospecific synthesis of 2-amino-6,7-dihydroxy-l,2,3,4-tetrahydronaphtalene, J.Org.Chem., 50: 3619.
Pellicciari R., Gallo-Mezo M.A., and Amer A.M., 1992, Stereospecific synthesis of the enantiomers of nicotinylalanine, a neuroprotecting agent, Tetrahedron Letters, 33: 3003.
Quallich G.J, Williams M.T., and Friedman R.C., 1990, Friedel-Crafts synthesis of 4-(3,4-dichlorophenyl)- 3,4-dihydro- l(2H)-naphtalenone, a key intermediate in the preparation of the antidepressant Sertaline, J.Org.Chem.55: 4971.
Reifenrath W.D., Bertelli D.J., and Fries D.S., 1976, Stereochemistry of Friedel-Crafts addition of phtaloylaspartic anhydride to benzene, Tetrahedron Letters, 23: 1959.
Saito K., Nowak T.S., Markey S.P., and Heyes M.P., 1993, Mechanism of delayed increases in kynurenine pathway metabolism in damaged brain regions following transient cerebral ischemia. J Neurochem. 60: 180.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1996 Plenum Press, New York
About this chapter
Cite this chapter
Giordani, A. et al. (1996). Enantiospecific Synthesis and in vitro Activity of Selective Inhibitors of Rat Brain Kynureninase and Kynurenine-3-Hydroxylase. In: Filippini, G.A., Costa, C.V.L., Bertazzo, A. (eds) Recent Advances in Tryptophan Research. Advances in Experimental Medicine and Biology, vol 398. Springer, Boston, MA. https://doi.org/10.1007/978-1-4613-0381-7_83
Download citation
DOI: https://doi.org/10.1007/978-1-4613-0381-7_83
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-8026-9
Online ISBN: 978-1-4613-0381-7
eBook Packages: Springer Book Archive