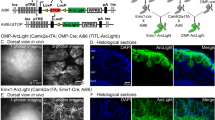
Abstract
Olfactory cilia are the obligate transducers of the odorant signal, and thus their study has been a focus of investigation in the olfactory field. Various methodologies have been established to visualize the cilia of olfactory sensory neurons; however, these approaches are limited to static imaging and often lack the ability to resolve individual cilia projecting from solitary neurons in the postnatal mouse. Here we detail a procedure of the visualization of olfactory cilia by ectopic expression of fluorescently tagged proteins. The procedure can be used for the observation and manipulation of the olfactory cilia and ciliary proteins in both static and dynamic conditions.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bloom G (1954) Studies on the olfactory epithelium of the frog and the toad with the aid of light and electron microscopy. Z Zellforsch Mikrosk Anat 41(1):89–100. https://doi.org/10.1007/BF00340285
Menco BP, Farbman AI (1985) Genesis of cilia and microvilli of rat nasal epithelia during pre-natal development. I. Olfactory epithelium, qualitative studies. J Cell Sci 78:283–310
Menco BP, Farbman AI (1985) Genesis of cilia and microvilli of rat nasal epithelia during pre-natal development. II. Olfactory epithelium, a morphometric analysis. J Cell Sci 78:311–336
Okano M, Weber AF, Frommes SP (1967) Electron microscopic studies on the distal border of the canine olfactory epithelium. J Ultrastruct Res 17(5):487–502. https://doi.org/10.1016/s0022-5320(67)80137-0
Kavoi B, Makanya A, Hassanali J, Carlsson HE, Kiama S (2010) Comparative functional structure of the olfactory mucosa in the domestic dog and sheep. Ann Anat 192(5):329–337. https://doi.org/10.1016/j.aanat.2010.07.004
Kratzing JE (1975) The fine structure of the olfactory and vomeronasal organs of a lizard (Tiliqua scincoides scincoides). Cell Tissue Res 156(2):239–252. https://doi.org/10.1007/BF00221807
Hansen A (2007) Olfactory and solitary chemosensory cells: two different chemosensory systems in the nasal cavity of the American alligator, Alligator mississippiensis. BMC Neurosci 8:64. https://doi.org/10.1186/1471-2202-8-64
Schulte E, Holl A (1971) Ultrastructure of the olfactory epithelium of Calamoichthys calabaricus J. A. Smoth (Pisces, Brachiopterygii). Z Zellforsch Mikrosk Anat 120(2):261–279. https://doi.org/10.1007/BF00335539
Hansen A, Zielinski BS (2005) Diversity in the olfactory epithelium of bony fishes: development, lamellar arrangement, sensory neuron cell types and transduction components. J Neurocytol 34(3–5):183–208. https://doi.org/10.1007/s11068-005-8353-1
Green WW, Uytingco CR, Ukhanov K, Kolb Z, Moretta J, McIntyre JC et al (2018) Peripheral gene therapeutic rescue of an olfactory ciliopathy restores sensory input, axonal pathfinding, and odor-guided behavior. J Neurosci 38(34):7462–7475. https://doi.org/10.1523/JNEUROSCI.0084-18.2018
Uytingco CR, Williams CL, Xie C, Shively DT, Green WW, Ukhanov K et al (2019) BBS4 is required for intraflagellar transport coordination and basal body number in mammalian olfactory cilia. J Cell Sci 132(5). https://doi.org/10.1242/jcs.222331
Williams CL, Uytingco CR, Green WW, McIntyre JC, Ukhanov K, Zimmerman AD et al (2017) Gene therapeutic reversal of peripheral olfactory impairment in Bardet-Biedl syndrome. Mol Ther 25(4):904–916. https://doi.org/10.1016/j.ymthe.2017.02.006
McIntyre JC, Davis EE, Joiner A, Williams CL, Tsai IC, Jenkins PM et al (2012) Gene therapy rescues cilia defects and restores olfactory function in a mammalian ciliopathy model. Nat Med 18(9):1423–1428. https://doi.org/10.1038/nm.2860
Ukhanov K, Uytingco C, Green W, Zhang L, Schurmans S, Martens JR (2022) INPP5E controls ciliary localization of phospholipids and the odor response in olfactory sensory neurons. J Cell Sci 135(5). https://doi.org/10.1242/jcs.258364
Xie C, Habif JC, Uytingco CR, Ukhanov K, Zhang L, de Celis C et al (2021) Gene therapy rescues olfactory perception in a clinically relevant ciliopathy model of Bardet-Biedl syndrome. FASEB J 35(9):e21766. https://doi.org/10.1096/fj.202100627R
Oberland S, Neuhaus EM (2014) Whole mount labeling of cilia in the main olfactory system of mice. J Vis Exp 94. https://doi.org/10.3791/52299
Alevra M, Schwartz P, Schild D (2012) Direct measurement of diffusion in olfactory cilia using a modified FRAP approach. PLoS One 7(7):e39628. https://doi.org/10.1371/journal.pone.0039628
Jenkins PM, Hurd TW, Zhang L, McEwen DP, Brown RL, Margolis B et al (2006) Ciliary targeting of olfactory CNG channels requires the CNGB1b subunit and the kinesin-2 motor protein, KIF17. Curr Biol 16(12):1211–1216. https://doi.org/10.1016/j.cub.2006.04.034
Delling M, DeCaen PG, Doerner JF, Febvay S, Clapham DE (2013) Primary cilia are specialized calcium signalling organelles. Nature 504(7479):311–314. https://doi.org/10.1038/nature12833
Fan S, Fogg V, Wang Q, Chen XW, Liu CJ, Margolis B (2007) A novel Crumbs3 isoform regulates cell division and ciliogenesis via importin beta interactions. J Cell Biol 178(3):387–398. https://doi.org/10.1083/jcb.200609096
Hansen JN, Kaiser F, Klausen C, Stüven B, Chong R, Bönigk W et al (2020) Nanobody-directed targeting of optogenetic tools to study signaling in the primary cilium. elife 9. https://doi.org/10.7554/eLife.57907
Iwadate Y (2003) Photolysis of caged calcium in cilia induces ciliary reversal in Paramecium caudatum. J Exp Biol 206(Pt 7):1163–1170. https://doi.org/10.1242/jeb.00219
Williams CL, McIntyre JC, Norris SR, Jenkins PM, Zhang L, Pei Q et al (2014) Direct evidence for BBSome-associated intraflagellar transport reveals distinct properties of native mammalian cilia. Nat Commun 5:5813. https://doi.org/10.1038/ncomms6813
Xie C, Habif JC, Ukhanov K, Uytingco CR, Zhang L, Campbell RJ et al (2022) Reversal of ciliary mechanisms of disassembly rescues olfactory dysfunction in ciliopathies. JCI Insight. https://doi.org/10.1172/jci.insight.158736
Uytingco CR, Martens JR (2019) Intranasal delivery of adenoviral and AAV vectors for transduction of the mammalian peripheral olfactory system. Methods Mol Biol 1950:283–297. https://doi.org/10.1007/978-1-4939-9139-6_17
Corey EA, Ukhanov K, Bobkov YV, McIntyre JC, Martens JR, Ache BW (2021) Inhibitory signaling in mammalian olfactory transduction potentially mediated by Gα. Mol Cell Neurosci 110:103585. https://doi.org/10.1016/j.mcn.2020.103585
McIntyre JC, Joiner AM, Zhang L, Iñiguez-Lluhí J, Martens JR (2015) SUMOylation regulates ciliary localization of olfactory signaling proteins. J Cell Sci 128(10):1934–1945. https://doi.org/10.1242/jcs.164673
Zhao H, Ivic L, Otaki JM, Hashimoto M, Mikoshiba K, Firestein S (1998) Functional expression of a mammalian odorant receptor. Science 279(5348):237–242. https://doi.org/10.1126/science.279.5348.237
Acknowledgments
We acknowledge Dr. Kirill Ukhanov for providing a representative whole-mount staining image and technical assistance. We thank Dr. Cedric Uytingco for providing a representative TIRF image. This work was supported by the National Institute of Deafness and Other Communication Disorders R01DC019345 (JRM), T32DC015994 (JCH), and F31DC020638 (JCH).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Habif, J.C., Xie, C., Martens, J.R. (2023). Visualizing and Manipulating Olfactory Cilia Through Viral Delivery Coupled with En Face Imaging of Intact OE. In: Goldstein, B.J., Matsunami, H. (eds) The Olfactory System. Methods in Molecular Biology, vol 2710. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-3425-7_1
Download citation
DOI: https://doi.org/10.1007/978-1-0716-3425-7_1
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-3424-0
Online ISBN: 978-1-0716-3425-7
eBook Packages: Springer Protocols