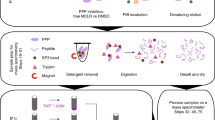
Abstract
When characterizing posttranslational modifications like phosphorylation, using efficient screening methods to map the phospho sites is essential, especially when dealing with large multi-domain proteins. NLRP3 (the NOD, LRR, and pyrin domain-containing protein 3), which initiates the formation of an NLRP3 inflammasome complex, is regulated posttranslationally by phosphorylation at several Ser and Tyr residues. However, determining sites of modification are not straightforward. For quick and reliable screening of the candidate phospho sites in NLRP3, we use a phospho dot blot assay which we describe here. This technique employs an in vitro kinase assay with a candidate kinase, Bruton’s Tyrosine Kinase (BTK), and peptides derived from the region of interest in the protein that contains the potential phosphorylation sites. The reaction containing the phosphorylated peptides is quickly screened by a dot blot where the peptides are blotted with a commercially available anti-phospho-tyrosine antibody. This method can also be adapted to detect modified Ser or Thr residues and is an ideal screening assay to map phospho residues in NLRP3 or other proteins. This can be an initial screening procedure or can be complemented by other approaches such as site directed mutagenesis and by generating phospho site-specific antibodies.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Broderick L, De Nardo D, Franklin BS, Hoffman HM, Latz E (2015) The inflammasomes and autoinflammatory syndromes. Annu Rev Pathol Mech Dis 10(1):395–424. https://doi.org/10.1146/annurev-pathol-012414-040431
Mangan MSJ, Olhava EJ, Roush WR, Seidel HM, Glick GD, Latz E (2018) Targeting the NLRP3 inflammasome in inflammatory diseases. Nat Rev Drug Discov 17(8):588–606. https://doi.org/10.1038/nrd.2018.97
Spalinger MR, Schwarzfischer M, Hering L et al (2019) Loss of PTPN22 abrogates the beneficial effect of cohousing-mediated fecal microbiota transfer in murine colitis. Mucosal Immunol 12(6):1336–1347. https://doi.org/10.1038/s41385-019-0201-1
Zhang Z, Meszaros G, He W-T et al (2017) Protein kinase D at the Golgi controls NLRP3 inflammasome activation. J Exp Med 214(9):2671–2693. https://doi.org/10.1084/jem.20162040
Bittner ZA, Liu X, Tortola MM et al (2019) BTK operates a phospho-tyrosine switch to regulate NLRP3 inflammasome activity. J Exp Med 218(11):e20201656. https://doi.org/10.1084/jem.20201656
Liu X, Pichulik T, Wolz OO et al (2017) Human NACHT, LRR, and PYD domain–containing protein 3 (NLRP3) inflammasome activity is regulated by and potentially targetable through Bruton tyrosine kinase. J Allergy Clin Immunol 140(4):1054–1067.e10. https://doi.org/10.1016/j.jaci.2017.01.017
Ito M, Shichita T, Okada M et al (2015) Bruton’s tyrosine kinase is essential for NLRP3 inflammasome activation and contributes to ischaemic brain injury. Nat Commun 6:1–11. https://doi.org/10.1038/ncomms8360
Mayor A, Martinon F, De Smedt T, Pétrilli V, Tschopp J (2007) A crucial function of SGT1 and HSP90 in inflammasome activity links mammalian and plant innate immune responses. Nat Immunol 8(5):497–503. https://doi.org/10.1038/ni1459
Hawkes R, Niday E, Gordon J (1982) A dot-immunobinding assay for monoclonal and other antibodies. Anal Biochem 119(1):142–147. https://doi.org/10.1016/0003-2697(82)90677-7
Hantschel O, Rix U, Schmidt U, Bürckstümmer T, Kneidinger M, Schütze G, Colinge J, Bennett KL, Ellmeier W, Valent P, Superti-Furga G (2007) The Btk tyrosine kinase is a major target of the Bcr-Abl inhibitor dasatinib. Proc Natl Acad Sci 104(33):13283. https://doi.org/10.1073/pnas.0702654104
Acknowledgments
We thank Xiaowu Zhang from Cell Signaling Technologies for helpful advice on the initial conceptual implementation of this method. The work was supported by the Else-Kröner-Fresenius Stiftung (to A.W.), the Deutsche Forschungsgemeinschaft (German Research Foundation, DFG) grant We-4195/15-1 (to A.W.), and the University Hospital Tübingen. Infrastructural funding was provided by the University of Tübingen, the University Hospital Tübingen, and the DFG Clusters of Excellence “iFIT—Image-Guided and Functionally Instructed Tumor Therapies” (EXC 2180, to AW), “CMFI—Controlling Microbes to Fight Infection” (EXC 2124, to AW), Gefördert durch die Deutsche Forschungsgemeinschaft (DFG) im Rahmen der Exzellenzstrategie des Bundes und der Länder—EXC 2180 and EXC 2124.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Shankar, S., Bittner, Z.A., Weber, A.N.R. (2022). NLRP3 Phospho-residue Mapping by Phospho Dot Blots. In: Abdul-Sater, A.A. (eds) The Inflammasome. Methods in Molecular Biology, vol 2459. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2144-8_10
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2144-8_10
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2143-1
Online ISBN: 978-1-0716-2144-8
eBook Packages: Springer Protocols