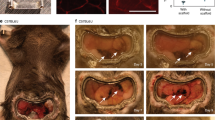
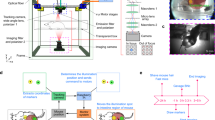
Abstract
Intravital microscopy is an imaging technique aimed at the visualization of the dynamics of biological processes in live animals. In the last decade, the development of nonlinear optical microscopy has enormously increased the use of this technique, thus addressing key biological questions in different fields such as immunology, neurobiology and tumor biology. In addition, new upcoming strategies to minimize motion artifacts due to animal respiration and heartbeat have enabled the visualization in real time of biological processes at cellular and subcellular resolution. Recently, intravital microscopy has been applied to analyze different aspect of mucosal immunity in the gut. However, the majority of these studies have been performed on the small intestine. Although crucial aspects of the biology of this organ have been unveiled, the majority of intestinal pathologies in humans occur in the large intestine.
Here, we describe a method to surgically expose and stabilize the large intestine in live mice and to perform short-term (up to 2 h) intravital microscopy.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Change history
23 July 2021
Chapter 15 “Method for Acute Intravital Imaging of the Large Intestine in Live Mice” was previously published with incorrect ESM videos and figure captions. This has now been rectified in the revised version of this book.
References
Weigert R, Porat-Shliom N, Amornphimoltham P (2013) Imaging cell biology in live animals: ready for prime time. J Cell Biol 201(7):969–979. https://doi.org/10.1083/jcb.201212130
Fish KN (2009) Total internal reflection fluorescence (TIRF) microscopy. Curr Protoc Cytom. Chapter 12:Unit12 18. https://doi.org/10.1002/0471142956.cy1218s50
Sahl SJ, Hell SW, Jakobs S (2017) Fluorescence nanoscopy in cell biology. Nat Rev Mol Cell Biol 18(11):685–701. https://doi.org/10.1038/nrm.2017.71
Wagner R (1839) Erlauterungstaflen zur Physiologie und Entwicklungs-geschichte. Leopold Voss, Leipzig, Germany
Pittet M, Weissleder R (2011) Intravital imaging. Cell 147(5):983–991. https://doi.org/10.1016/j.cell.2011.11.004
Secklehner J, Lo Celso C, Carlin LM (2017) Intravital microscopy in historic and contemporary immunology. Immunol Cell Biol 95(6):506–513. https://doi.org/10.1038/icb.2017.25
Perry SW, Burke RM, Brown EB (2012) Two-photon and second harmonic microscopy in clinical and translational cancer research. Ann Biomed Eng 40(2):277–291. https://doi.org/10.1007/s10439-012-0512-9
Wang BG, Konig K, Halbhuber KJ (2010) Two-photon microscopy of deep intravital tissues and its merits in clinical research. J Microsc 238(1):1–20. https://doi.org/10.1111/j.1365-2818.2009.03330.x
Masedunskas A, Weigert R (2008) Intravital two-photon microscopy for studying the uptake and trafficking of fluorescently conjugated molecules in live rodents. Traffic 9(10):1801–1810. https://doi.org/10.1111/j.1600-0854.2008.00798.x
Bestvater F, Spiess E, Stobrawa G, Hacker M, Feurer T, Porwol T, Berchner-Pfannschmidt U, Wotzlaw C, Acker H (2002) Two-photon fluorescence absorption and emission spectra of dyes relevant for cell imaging. J Microsc 208(Pt 2):108–115
Spiess E, Bestvater F, Heckel-Pompey A, Toth K, Hacker M, Stobrawa G, Feurer T, Wotzlaw C, Berchner-Pfannschmidt U, Porwol T, Acker H (2005) Two-photon excitation and emission spectra of the green fluorescent protein variants ECFP, EGFP and EYFP. J Microsc 217(Pt 3):200–204. https://doi.org/10.1111/j.1365-2818.2005.01437.x
You S, Tu H, Chaney EJ, Sun Y, Zhao Y, Bower AJ, Liu YZ, Marjanovic M, Sinha S, Pu Y, Boppart SA (2018) Intravital imaging by simultaneous label-free autofluorescence-multiharmonic microscopy. Nat Commun 9(1):2125. https://doi.org/10.1038/s41467-018-04470-8
Weigelin B, Bakker GJ, Friedl P (2016) Third harmonic generation microscopy of cells and tissue organization. J Cell Sci 129(2):245–255. https://doi.org/10.1242/jcs.152272
Masedunskas A, Milberg O, Porat-Shliom N, Sramkova M, Wigand T, Amornphimoltham P, Weigert R (2012) Intravital microscopy: a practical guide on imaging intracellular structures in live animals. BioArchitecture 2(5):143–157. https://doi.org/10.4161/bioa.21758
Ebrahim S, Weigert R (2019) Intravital microscopy in mammalian multicellular organisms. Curr Opin Cell Biol 59:97–103. https://doi.org/10.1016/j.ceb.2019.03.015
Ebrahim S, Chen D, Weiss M, Malec L, Ng Y, Rebustini I, Krystofiak E, Hu L, Liu J, Masedunskas A, Hardeman E, Gunning P, Kachar B, Weigert R (2019) Dynamic polyhedral actomyosin lattices remodel micron-scale curved membranes during exocytosis in live mice. Nat Cell Biol 21(8):933–939. https://doi.org/10.1038/s41556-019-0365-7
Kolesnikov M, Farache J, Shakhar G (2015) Intravital two-photon imaging of the gastrointestinal tract. J Immunol Methods 421:73–80. https://doi.org/10.1016/j.jim.2015.03.008
Chieppa M, Rescigno M, Huang AY, Germain RN (2006) Dynamic imaging of dendritic cell extension into the small bowel lumen in response to epithelial cell TLR engagement. J Exp Med 203(13):2841–2852. https://doi.org/10.1084/jem.20061884
Farache J, Koren I, Milo I, Gurevich I, Kim KW, Zigmond E, Furtado GC, Lira SA, Shakhar G (2013) Luminal bacteria recruit CD103+ dendritic cells into the intestinal epithelium to sample bacterial antigens for presentation. Immunity 38(3):581–595. https://doi.org/10.1016/j.immuni.2013.01.009
Chennupati V, Worbs T, Liu X, Malinarich FH, Schmitz S, Haas JD, Malissen B, Forster R, Prinz I (2010) Intra- and intercompartmental movement of gammadelta T cells: intestinal intraepithelial and peripheral gammadelta T cells represent exclusive nonoverlapping populations with distinct migration characteristics. J Immunol 185(9):5160–5168. https://doi.org/10.4049/jimmunol.1001652
Spadoni I, Zagato E, Bertocchi A, Paolinelli R, Hot E, Di Sabatino A, Caprioli F, Bottiglieri L, Oldani A, Viale G, Penna G, Dejana E, Rescigno M (2015) A gut-vascular barrier controls the systemic dissemination of bacteria. Science 350(6262):830–834. https://doi.org/10.1126/science.aad0135
Toiyama Y, Mizoguchi A, Okugawa Y, Koike Y, Morimoto Y, Araki T, Uchida K, Tanaka K, Nakashima H, Hibi M, Kimura K, Inoue Y, Miki C, Kusunoki M (2010) Intravital imaging of DSS-induced cecal mucosal damage in GFP-transgenic mice using two-photon microscopy. J Gastroenterol 45(5):544–553. https://doi.org/10.1007/s00535-009-0187-7
Fumagalli A, Drost J, Suijkerbuijk SJ, van Boxtel R, de Ligt J, Offerhaus GJ, Begthel H, Beerling E, Tan EH, Sansom OJ, Cuppen E, Clevers H, van Rheenen J (2017) Genetic dissection of colorectal cancer progression by orthotopic transplantation of engineered cancer organoids. Proc Natl Acad Sci U S A 114(12):E2357–E2364. https://doi.org/10.1073/pnas.1701219114
Ritsma L, Steller EJ, Ellenbroek SI, Kranenburg O, Borel Rinkes IH, van Rheenen J (2013) Surgical implantation of an abdominal imaging window for intravital microscopy. Nat Protoc 8(3):583–594. https://doi.org/10.1038/nprot.2013.026
Ritsma L, Ellenbroek SIJ, Zomer A, Snippert HJ, de Sauvage FJ, Simons BD, Clevers H, van Rheenen J (2014) Intestinal crypt homeostasis revealed at single-stem-cell level by in vivo live imaging. Nature 507(7492):362–365. https://doi.org/10.1038/nature12972
Puccini A, Battaglin F, Lenz HJ (2018) Management of Advanced Small Bowel Cancer. Curr Treat Options in Oncol 19(12):69. https://doi.org/10.1007/s11864-018-0592-3
Oh G, Yoo SW, Jung Y, Ryu YM, Park Y, Kim SY, Kim KH, Kim S, Myung SJ, Chung E (2014) Intravital imaging of mouse colonic adenoma using MMP-based molecular probes with multi-channel fluorescence endoscopy. Biomed Opt Express 5(5):1677–1689. https://doi.org/10.1364/BOE.5.001677
Ntziachristos V (2006) Fluorescence molecular imaging. Annu Rev Biomed Eng 8:1–33. https://doi.org/10.1146/annurev.bioeng.8.061505.095831
Masedunskas A, Sramkova M, Parente L, Sales KU, Amornphimoltham P, Bugge TH, Weigert R (2011) Role for the actomyosin complex in regulated exocytosis revealed by intravital microscopy. Proc Natl Acad Sci U S A 108(33):13552–13557. https://doi.org/10.1073/pnas.1016778108
Muzumdar MD, Tasic B, Miyamichi K, Li L, Luo L (2007) A global double-fluorescent Cre reporter mouse. Genesis 45(9):593–605. https://doi.org/10.1002/dvg.20335
Acknowledgments
This research was supported by the Intramural Research Program of the National Institutes of Health, National Cancer Institute, Center for Cancer Research (RW). This work has been supported by a UICC International Cancer Research Technology Transfer Fellowship (UICC ICRETT ICR/2015/40447 fellowship to ME).
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
1 Electronic Supplementary Materials
Blood flow in the large intestine mucosal layer. Green: FITC-conjugated dextran. Scale bar: 20 μm. The original pixel size has been computationally modified for a better video quality. (AVI 1223 kb)
Z-stack acquisition of the large intestine of B5/EGFP mice, from the muscular layer to the mucosal layer. Scale bar: 100 μm. (AVI 4461 kb)
Blood flow in the large intestine muscular layer. Red: TRITC-conjugated dextran, Green: CX3CR1gfp- positive tissue macrophages. Scale bar: 50 μm. The original pixel size has been computationally modified for a better video quality. (AVI 372 kb)
Rights and permissions
Copyright information
© 2021 This is a U.S. government work and not under copyright protection in the U.S.; foreign copyright protection may apply and Springer Nature US
About this protocol
Cite this protocol
Erreni, M., Doni, A., Weigert, R. (2021). Method for Acute Intravital Imaging of the Large Intestine in Live Mice. In: Brzostowski, J., Sohn, H. (eds) Confocal Microscopy. Methods in Molecular Biology, vol 2304. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1402-0_15
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1402-0_15
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1401-3
Online ISBN: 978-1-0716-1402-0
eBook Packages: Springer Protocols