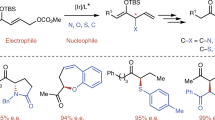
Abstract
Enantioenriched sp 3-hybridized 1-oxy-alkyllithium compounds are accessible by lithiodestannylation of the appropriate chiral stannanes or by deprotonation of 1-alkyl carbamates by means of sec-butyllithium/(-)-sparteine. These are usually configurationally stable at temperatures below-40°C and are substituted by a wide array of electrophiles with strict stereoretention. When applying chiral substrates, bearing an adjacent stereogenic center, often a high internal chiral induction occurs, being the basis for an efficient kinetic resolution in the deprotonation step. α-Oxybenzyllithium derivatives are usually more easily accessible due to mesomeric stabilization, but most of these compounds undergo facile racemization or epimerization at temperatures around-70 to -78°C. The sense of stereospecificity of the electrophilic substitution is less predictable: both — retention or inversion — are common, depending on the individual situation. 1-Oxy-2-alkenyllithium reagents have similar stereochemical properties. In a number of cases configurational stability is recorded. Procedures for efficient dynamic kinetic resolution, involving a crystallization step, have been developed. In particular, 1-lithio-2-alkenyl carbamates — after titanation — are valuable homoenolate reagents for achieving highly stereoselective homoaldol reactions. Chiral 1-oxy-2-alkynyllithium derivatives allow for a facile entry to enantioenriched allenes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
For the sake of simplicity, the 1-oxy-substituted lithium compounds such as 2 are drawn in a simplified form although it is known that the C-Li-bond has only a small covalent portion (if at all) and a high tendency for the formation of dimers or tetramers exists. Furthermore, the lithium cation is coordinatively saturated by Lewis-basic ligands; see: Lambert C, Schleyer PvR (1993) Carbanionen-polare Organometall-Verbindungen. In: Hanack M (ed) Houben-Weyl, Carbanionen, vol. E 19 d, 4th edn. Thieme-Verlag, Stuttgart, p 1
Still WC, Sreekumar C (1980) J Am Chem Soc 102:1201
Jephcote VJ, Pratt AJ, Thomas EJ (1989) J Chem Soc Perkin Trans 1 1529
Evans DA, Andrews GC, Buckwalter B (1974) J Am Chem Soc 96:5560
Ahlbrecht H, Beyer U (1999) Synthesis 365
Hoppe D (1984) Angew Chem 96:930; Angew Chem Int Ed Engl 23:932
Hoppe D, Krämer T (1986) Angew Chem 98:171; Angew Chem Int Ed Engl 25:160
Dreller S, Dyrbusch M, Hoppe D (1991) Synlett 397
Chong JM, Mar EK (1991) Tetrahedron Lett 32:5683
Chan PCM, Chong JM (1988) J Org Chem 53:5584
Tomooka K, Igarashi T, Nakai T (1994) Tetrahedron Lett 35:1913
Matteson DS, Tripathy PB, Sarkar A, Sadhu KM (1989) J Am Chem Soc 111:4399
Marshall JA, Gung WY (1989) Tetrahedron 45:1043
Marshall JA ( 1992) Chemtracts, Org Chem 5:75
Sawyer JS, Kucerovy A, Macdonald TL, McGarvey GJ ( 1988) J Am Chem Soc 110:842
McGarvey GJ, Kimura M ( 1982) J Org Chem 47:5420
Stork G, Manabe K, Liu L (1998) J Am Chem Soc 120:1337
Yamada JI, Abe H, Yamamoto Y (1990) JAm Chem Soc 112:6118
Yamamoto Y (1991) Chemtracts, Org Chem 4:255
Chong JM, Mar EK (1989) Tetrahedron 45:7709
Smyj RP, Chong JM (2001) Org Lett 3:2903
Chan PCM, Chong JM (1990) Tetrahedron Lett 31:1985
Chong JM, Mar EK (1990) Tetrahedron Lett 31:1981
Linderman RJ, Griedel BD (1990) J Org Chem 55:5428
Linderman RJ, Griedel BD (1991) J Org Chem 56:5491
Hutchinson DK, Fuchs PL (1987) J Am Chem Soc 109:4930
Christopher JA, Kocienski PJ, Procter MJ (1998) Synlett 425
Christopher JA, Kocienski PJ, Kuhl A, Bell R (2000) Synlett 463
Screttas CG, Micha-Screttas M (1978) J Org Chem 43:1064
Cohen T, Daniewski WM, Weisenfeld RB (1978) Tetrahedron Lett 4665
Cohen T, Bhupathy M (1989) Acc Chem Res 22:152
Cohen T, Lin MT (1984) J Am Chem Soc 106:1130
Rychnovsky SD, Mickus DE ( 1989) Tetrahedron Lett 30:3011
Lancelin JM, Morin-Allory L, Sinaÿ P (1984) J Chem Soc Chem Commun 355
Fernandez-Mayoralas A, Marra A, Trumtel M, Veyrières A, Sinaÿ P (1989) Tetrahedron Lett 30:2537
Pedretti V, Veyrières A, Sinaÿ P (1990) Tetrahedron 46:77
Beau JM, Sinaÿ P (1985) Tetrahedron Lett 26:6185
Beau JM, Sinaÿ P (1985) Tetrahedron Lett 26:6189
Beau JM, Sinaÿ P (1985) Tetrahedron Lett 26:6193
Frey O, Hoffmann M, Kessler H (1995) Angew Chem 107:2194; Angew Chem Int Ed 34:2026
Hoffmann M, Kessler H (1995) Tetrahedron Lett 35:6067
Rychnovsky SD, Mickus DE (1989) Tetrahedron Lett 30:3011
Rychnovsky SD, Buckmelter AJ, Dahanukar VH, Skalitzky DJ (1999) J Org Chem 64:6849
Rychnovsky SD, Zeller S, Skalitzky DJ, Griesgraber G ( 1990) J Org Chem 55:5550
Rychnovsky SD (1995) Chem Rev 95:2021
Rychnovsky SD, Griesgraber G, Schlegel R (1994) J Am Chem Soc 116:2623
Rychnovsky SD, Griegraber G, Kim J (1994) J Am Chem Soc 116:2621
Rychnovsky SD, Hoye RC (1994) J Am Chem Soc 116:1753
Richardson TI, Rychnovsky SD (1996) J Org Chem 61:4219
Rychnovsky SD, Khire UR, Yang G (1997) J Am Chem Soc 119:2058
Richardson TL, Rychnovsky SD (1997) J Am Chem Soc 119:12360
Ley SV, Jones P ( 1998) Chemtracts, Org Chem 11:1005
Schneider C (1998) Angew Chem 110:1445; Angew Chem Int Ed Engl37:1375
Yamauchi Y, Katagiri T, Uneyama K (2002) Org Lett 4:173
Hoppe D, Paetow M, Hintze F (1993) Angew Chem 105:430; Angew Chem Int Ed Engl 32:394
Paetow M, Kotthaus M, Grehl M, Fröhlich R, Hoppe D (1994) Synlett 1034
Schlosser M (2002) Organoalkali chemistry. In: Schlosser M (ed) Organometallics in synthesis, 2nd edn. Wiley, Chichester, p 1
Schlosser M (1992) Superbases as powerful tools in organic synthesis. In: Scheffold R (ed) Modern synthetic methods, vol. 6, Springer, Berlin, p 227
Mordini A (1992) Superbases and their use in organic synthesis. In: Snieckus V (ed) Advances in carbanion chemistry, vol. 1, Jai Press Inc., Greenwich, Connecticut, p 1
Lochmann L (2000) Eur J Inorg Chem 1115
Hoppe D, Hintze F, Tebben P (1990) Angew Chem 102:1457; Angew Chem Int Ed Engl 29:1422
Hoppe D, Hintze F, Tebben P, Paetow M, Ahrens H, Schwerdtfeger J, Sommerfeld P, Haller J, Guarnieri W, Kolczewski S, Hense T, Hoppe I (1994) Pure Appl. Chem 66:1479
Hoppe D, Hense T (1997) Angew Chem 109:2376; Angew Chem Int Ed Engl 36:2282
Haller J, Hense T, Hoppe D (1993) Synlett 726
Haller J, Hense T, Hoppe D (1996) Liebigs Ann Chem 489
Schwerdtfeger J, Hoppe D (1992) Angew Chem 104:1547; Angew Chem Int Ed Engl 31:1505
Schwerdtfeger J, Kolczewski S, Weber B, Fröhlich R, Hoppe D (1999) Synthesis 1573
O’Donnell MJ, Delgado F, Hostettler C, Schwesinger R (1998) Tetrahedron Lett 39:8775, and previous work cited therein
Boie C, Hoppe D (1997) Synthesis 176
Weber B, Kolczewski S, Fröhlich R, Hoppe D (1999) Synthesis 1593
Masamune S, Choy W, Petersen JS, Sita LR (1985) Angew Chem 97:1; Angew Chem Int Ed Engl24:1
Weber B, Schwerdtfeger J, Fröhlich R, Göhrt A, Hoppe D (1999) Synthesis 1915
Woltering MJ, Fröhlich R, Wibbeling B, Hoppe D (1998) Synlett 797
Guarnieri W, Grehl M, Hoppe D (1994) Angew Chem 106:1815; Angew Chem Int EdEngl 33:1734
Guarnieri W, Sendzik M, Fröhlich R, Hoppe D (1998) Synthesis 1274
Sendzik M, Guarnieri W, Hoppe D (1998) Synthesis 1287
Ahrens H, Paetow M, Hoppe D (1992) Tetrahedron Lett 33:5327
Helmke H, Hoppe D (1995) Synlett 978
Helmke H (1995) Dissertation, University of Münster
Hoppe D (1995) (-)-Sparteine. In: Paquette LA (ed), Encyclopedia of reagents for organic synthesis, vol 7, 1st edn. Wiley, Chichester, p 4662
Aggarwal VK (1994) Angew Chem 106:185; Angew Chem Int Ed Engl 33:175
Ebner T, Eichelbaum M, Fischer P, Meese CO (1989) Arch Pharm 322:399
Würthwein E-U, Behrens K, Hoppe D (1999) Chem Eur J 5:3459
Hintze F, Hoppe D (1992) Synthesis 1216
Hoppe I, Marsch M, Harms K, Boche G, Hoppe D (1995) Angew Chem 107:2328; Angew Chem Int Ed Eng 34:2158
Hintze F (1993) Dissertation, University of Kiel
Rein KS, Chen Z-H, Perumal PT, Echegoyen L, Gawley RE (1991) Tetrahedron Lett 32:1941 and references therein
Papillon JPN, Taylor RJK (2002) Org Lett 4:119
Tomooka K, Shimizu H, Nakai T (2001) J Organomet Chem 624:364
Behrens K, Fröhlich R, Meyer O, Hoppe D (1998) Eur J Org Chem 2397
Hoppe I, unpublished results
Boie C (1996) Dissertation, University of Münster
Sommerfeld P, Hoppe D (1992) Synlett 764
Paetow M, Ahrens H, Hoppe D (1992) Tetrahedron Lett 33:5323
Ahrens H (1994) Dissertation, University of Münster
Christoph G, Hoppe D (2002) Org Lett 4:2189
Hoppe D, Ahrens H, Guarnieri W, Helmke H, Kolczewski S (1996) Pure Appl Chem 68:613
For a more recent example, combined with chiral amplification, see Sato I, Omiya D, Saito T, Soai K (2000) J Am Chem Soc 122:11739
Würthwein E-U, unpublished results
For calculations on the (-)-sparteine-mediated N-Boc-pyrrolidines, see Wiberg KB, Bailey WF (2000) Angew Chem 112:2211; Angew Chem Int Ed 39:2127
Paetow M (1993) Dissertation, University of Kiel
van Bebber J ( 1997) Dissertation, University of Münster
Tomooka K, Shimizu H,I noue T, Shibata H, Nakai T (1999) Chem Lett 759
Boche G, Bosold F, Lohrenz JCW, Opel A, Zulauf P (1993) Chem Ber 126:1873
For a similar reaction of-(dibenzylamino) alkyl carbamates, see ref. Kolczewski S, Fröhlich R, Hoppe D (1999) Synthesis 1593 [71]
Kotthaus M (1997) Dissertation, University of Münster
Marek I (1999) J Chem Soc Perkin Trans 1 535
Woltering MJ, Fröhlich R, Hoppe D (1997) Angew Chem 109:1804; Angew Chem Int Ed Engl 36:1764
Tomooka K, Komine N, Sasaki T, Shimizu H, Nakai T (1998) Tetrahedron Lett 39:9715
Hoppe D, Woltering MJ, Oestreich M, Fröhlich R (1999) Helv Chim Acta 82:1860
Oestreich M, Fröhlich R, Hoppe D (1998) Tetrahedron Lett 39:1745
Oestreich M, Fröhlich R, Hoppe D (1999) J Org Chem 64:8616
Oestreich M, Hoppe D (1999) Tetrahedron Lett 40:1881,3283
Gralla G, Wibbeling B, Hoppe D (2002) Org Lett 4:2193
Christoph G (2002) Dissertation, University of Münster
Seebach D, Prelog V (1982) Angew Chem 94:696; Angew Chem Int Ed Engl 21:654
Hense T, Hoppe D (1997) Synthesis 1394
van Bebber J, Ahrens H, Fröhlich R, Hoppe D (1999) Chem Eur J 5:1905
Laqua H, Fröhlich R, Wibbeling B, Hoppe D (2001) J Organomet Chem 624:96
Hodgson DM, Lee GP (1996) J Chem Soc Chem Commun 1015
Hodgson DM, Robinson LA (1999) J Chem Soc Chem Commun 309
Hodgson DM, Cameron ID (2001) Org Lett 3:441
Hodgson DM, Gras E (2002) Angew Chem 114:2482; Angew Chem Int 41:2376
Streitwieser Jr. A, Juaristi E, Nebenzahl LL (1980) Equilibrium carbon acidities in solution. In: Buncel E, Durst T (eds) Comprehensive carbanion chemistry, vol. 5A, Elsevier, Amsterdam, p 323
Ruhland T, Hoffmann RW, Schade S, Boche G (1995) ChemBer 128:551
Beak P, Basu A, Gallagher DJ, Park YS, Thayumanavan S (1996) Acc Chem Res 29:552
Basu A, Thayumanavan S (2002) Angew Chem 114:740; Angew Chem Int 41:717
Hoppe D, Carstens A, Krämer T (1990) Angew Chem 102:1455; Angew Chem Int Ed Engl 29:1424
Hammerschmidt F, Hanninger A (1995) Chem Ber 128:1069
Carstens A, Hoppe D (1994) Tetrahedron 50:6097
Derwing C (1995) Dissertation, University of Münster
Carstens A (1993) Dissertation, University of Kiel
Derwing C, Hoppe D (1996) Synthesis 149
Brewster JH, Braden WE Jr (1964) Chem Ind 1759
Mitsui S, Imaizumi S, Senda Y, Konno K(1964) Chem Ind 233
Mitsui S, Senda Y, Konno K (1963) Chem Ind 1354
Zhang P, Gawley RE (1993) J Org Chem 58:3223
Hoppe D (1997) Generation and reactions of chiral, mesomerically stabilized lithium carbanions. In: Nair V, Kumar S (eds) New horizons in organic synthesis. New Age International Publishers, New Delhi, p 130
Derwing C, Frank H, Hoppe D ( 1999) Eur J Org Chem 3519
Hammerschmidt F, Hanninger A, Simov BP, Völlenkle H, Werner A (1999) Eur J Org Chem 3511
Peters JG, Seppi M, Fröhlich R, Wibbeling B, Hoppe D (2002) Synthesis 381
Superchi S, Sotomayor N, Miao G, Joseph B, Campbell MG, Snieckus V (1996) Tetrahedron Lett 37:6061
Hoffmann RW, Rühl T, Harbach J ( 1992) Liebigs Ann Chem 725
Caddick S, Jenkins K (1996) Chem Soc Rev:447
Retzow S (1993) Dissertation, University of Kiel
Heinl T, Retzow S, Hoppe D, Fraenkel G, Chow A (1999) Chem Eur J 5:3464
For calculations on a higher level concerning the problem, see: Wiberg KB, Bailey WF (2000) J Mol Struct (special issue) 556:239
Komine N, Wang L-F, Tomooka K, Nakai T (1999) Tetrahedron Lett 40:6809
Tomooka K, Wang L-F, Komine N, Nakai T (1999) Tetrahedron Lett 40:6813
Schwark J-R, Hoppe D (1990) Synthesis 291
Zschage O, Schwark J-R, Hoppe D (1990) Angew Chem 102:336; Angew Chem Int Ed Engl 29:296
Kunz H, Waldmann H (1990) Chemtracts Org Chem 3:421
Zschage O, Hoppe D (1992) Tetrahedron 48:8389
Marsch M, Harms K, Zschage O, Hoppe D, Boche G (1991) Angew Chem 103:338; Angew Chem Int Ed Engl 30:321
Hoppe D, Krämer T, Schwark JR, Zschage O (1990) Pure Appl Chem 62:1999
Hoppe D, Zschage O (1990) Chiral metallated carbamates: Tools for new strategies in asymmetric synthesis. In: Dötz KH, Hoffmann RW (eds) Organic synthesis via organometallics, 1st edn. Vieweg, Braunschweig, p 267
Hoppe D (1986) Synthesis of enantiomerically pure unnatural compounds via nonbiomimetic homoaldol reactions. In: Schneider MP (ed) Enzymes as catalysts in organic synthesis. D. Reidel, Dordrecht, p 177
Hoppe D (1995) Formation of C-C bonds by addition of allyl-type organometallic compounds to carbonyl compounds. General aspects. In: Helmchen G, Hoffmann RW, Mulzer J, Schaumann E (eds) Houben-Weyl, Stereoselective synthesis, vol. E 21b, 4th edn. Thieme-Verlag, Stuttgart, p 1357
Hoppe D (1995) Formation of C-C bonds by addition of allyl-type organometallic compounds to carbonyl compounds. Allyl alkali metal reagents (M=Li, Na, K). In: Helmchen G, Hoffmann RW, Mulzer J, Schaumann E (eds) Houben-Weyl, Stereoselective synthesis, vol. E 2 1b, 4th edn. Thieme-Verlag, Stuttgart, p 1379
Hoppe D (1995) Formation of C-C bonds by addition of allyl-type organometallic compounds to carbonyl compounds. Allyltitanium and allylzirconium reagents. In: Helmchen G, Hoffmann RW, Mulzer J, Schaumann E (eds) Houben-Weyl, Stereoselective synthesis, vol. E 21b, 4th edn. Thieme-Verlag, Stuttgart, p 1551
Hoppe D (1995) (E)-1-(N,N-Diisopropylcarbamoyloxy)crotyllithium. In: Paquette LA (ed) Encyclopedia of reagents for organic synthesis, vol. 3, 1st edn. Wiley, Chichester, p 1927
Hoffmann RW (1989) Chem Rev 89:1841
Zimmermann HE, Traxler MD (1957) J Am Chem Soc 79:1920
Zschage O, Schwark J-R, Krämer T, Hoppe D (1992) Tetrahedron 48:8377
Krämer T, Hoppe D ( 1987) Tetrahedron Lett 28:5149
Unfortunately, the assignments in the preliminary communication (Krämer T, Schwark J-R, Hoppe D (1989) Tetrahedron Lett 30:7037) are incorrect
Schwark JR (1991) Dissertation, University of Kiel
Seppi M (2001) Dissertation, University of Münster
Paulsen H, Graeve C, Hoppe D (1996) Synthesis 141
Hoppe D, Hanko R, Brönneke A, Lichtenberg F, van Hülsen E (1985) Chem Ber 118:2822
van Hülsen E. Hoppe D (1985) Tetrahedron Lett 26:411
Deiters A, Hoppe D (1999) Angew Chem 111:529; Angew Chem Int Ed 38:546
Deiters A, Hoppe D (2001) J Org Chem 66:2842
Deiters A, Fröhlich R, Hoppe D (2000) Angew Chem 112:2189; Angew Chem Int Ed 39:2105
Deiters A, Mück-Lichtenfeld C, Fröhlich R, Hoppe D (2002) Chem Eur J 8:1833; (2000) Org Lett 2:2415
Deiters A, Wibbeling B, Hoppe D (2001) Adv Synth Catal 343:181
Özlügedik M, Kristensen J, Wibbeling B, Fröhlich R, Hoppe D (2002) Eur J Org Chem:414
Özlügedik M, Hoppe D (2002) Synthesis, in preparation
Heimbach D, Fröhlich R, Wibbeling B, Hoppe D (2000) Synlett 950
Reggelin M, Tebben P, Hoppe D (1989) Tetrahedron Lett 30:2915
Tebben P, Reggelin M, Hoppe D (1989) Tetrahedron Lett 30:2919
Hoppe D, Tebben P, Reggelin M, Bolte M (1997) Synthesis 183
Hanko R, Hoppe D (1982) Angew Chem 94:378; Angew Chem Int Ed Engl 21:372
Hoppe D, Tarara G, Wilckens M, Jones PG, Schmidt D, Stezowski JJ (1987) Angew Chem 99:1079; Angew Chem Int Ed Engl 26:1034
Hoffmann RW, Lanz J, Metternich R, Tarara G, Hoppe D (1987) Angew Chem 99:1196; Angew Chem Int Ed Engl 26:1145
Hoppe D, Tarara G, Wilckens M (1989) Synthesis 83
Hirsch R, Hoffmann RW (1992) Chem Ber 125:975
Hoffmann RW, Weidmann U (1985) Chem Ber 118:3980
Landmann B, Hoffmann RW (1987) Chem Ber 120:331
Hanko R, Rabe K, Dally R, Hoppe D (1991) Angew Chem 103:1725; Angew Chem Int Ed Engl 30:1690
Rehders F, Hoppe D ( 1992) Synthesis 859
Berque I, Le Ménez P, Razon P, Pancrazi A, Ardisson J, Neuman A, Prangé T, Brion J-D (1998) Synlett 1132 199. Kocienski P,Dixon NJ (1989) Synlett 52
Berque I, Le Ménez P, Razon P, Pancrazi A, Ardisson J, Brion J-D ( 1998) Synlett 1135
Zschage O, Hoppe D (1992) Tetrahedron 48:5657
Paulsen H, Hoppe D (1992) Tetrahedron 48:5667
Peschke B, Lüßmann J, Dyrbusch M, Hoppe D (1992) Chem Ber 125:1421
Le Ménez P, Fargeas V, Poisson J, Ardisson J, Lallemand J-Y, Pancrazi A (1994) Tetrahedron Lett 35:7767
Le Menez P, Firmo N, Fargeas V, Ardisson J, Pancrazi A (1994) Synlett 995
Férézou JP, Julia M, Khourzom R, Pancrazi A, Robert P ( 1991 ) Synlett 611
Le Ménez P, Fargeas V, Berque I, Poisson J, Ardisson J, Lallemand J-Y, Pancrazi A (1995) J Org Chem 60:3592
Ashworth P. Broadbelt B. Jankowski P. Pimm A. Kocienski P (1995) Synthesis 199
Smith ND, Kocienski PJ, Street SDA (1996) Synthesis 652
Marshall JA (1996) Chem Rev 96:31
Marshall JA (1998) Chemtracts, Org Chem 11:697
Paulsen H, Graeve C, Fröhlich R, Hoppe D (1996) Synthesis 145
Grieco PA, Oguri T, Yokoyama Y (1978) Tetrahedron Lett 419
Hoppe D, Brönneke A (1983) Tetrahedron Lett 24:1687
Zschage O, Hoppe D (1992) Tetrahedron 48:5657
Paulsen H, Hoppe D (1992) Tetrahedron 48:5667
Prasad KRK, Hoppe D (2000) Synlett 1067
Madec D, Henryon V, Férézou J-P ( 1999) Tetrahedron Lett 40:8103
Sharpless KB, Verhoeven TR (1979) Aldrichimica Acta 12:63
Rossiter BE, Verhoeven TR, Sharpless KB (1979) Tetrahedron Lett 4733
Mihelich ED (1979) Tetrahedron Lett 4729
Hoppe D, Lüßmann J, Jones PG, Schmidt D, Sheldrick GM (1986) Tetrahedron Lett 27:3591
Tarara G, Hoppe D (1989) Synthesis 89
Nakata M, Toshima K, Kai T, Kinoshita M ( 1985) Bull Chem Soc Jpn 58:3457 and previous publications
Lüßmann J, Hoppe D, Jones PG, Fittschen C, Sheldrick GM (1986) Tetrahedron Lett 27:3595
Hoppe D, Krämer T, Freire Erdbrügger C, Egert E (1989) Tetrahedron Lett 30:1233
Braun M (1998) Angew Chem 110:445; Angew Chem Int Ed 37:430
Sengupta S, Snieckus V (1990) J Org Chem 55:5680
Peschke B (1991) Dissertation, University of Kiel
Howarth JA, Owton WM, Percy JM (1994) Synlett 503
Kocienski P, Barber C (1990) Pure Appl Chem 62:1933
Pimm A, Kocienski P, Street SDA (1992) Synlett 886
Hareau-Vittini G, Kocienski PJ (1995) Synlett 893
Madec D, Pujol S, Henryon V, Férézou JP (1995) Synlett 435
Sanford MS, Love JA, Grubbs RH (2001) J Am Chem Soc 123:6543
Grubbs RH, Miller SJ, Fu GC (1995) Acc Chem Res 28:446
Schuster M, Blechert S (1997) Angew Chem 109:2124; Angew Chem Int Ed Engl 36:2036
Hoppe D, Riemenschneider C (1983) Angew Chem 95:64; Angew Chem Int Ed Engl 22:54
Hoppe D, Gonschorrek C, Schmidt D, Egert E (1987) Tetrahedron 43:2457
Reich HJ, Holladay JE ( 1995) J Am Chem Soc 117:8470
Hoppe D, Gonschorrek C (1987) Tetrahedron Lett 28:785
Egert E, Beck H, Schmidt D, Gonschorrek C, Hoppe D (1987) Tetrahedron Lett 28:789
Hoppe D, Gonschorrek C, Egert E, Schmidt D (1985) Angew Chem 97:706; Angew Chem Int Ed Engl 24:700
Marshall JA, Lebreton J (1987) Tetrahedron Lett 28:3323
Schultz-Fademrecht C, Wibbeling B, Fröhlich R, Hoppe D (2001) Org Lett 3:1221
Schultz-Fademrecht C, Tius MA, Grimme S, Wibbeling B, Hoppe D (2002) Angew Chem 115:1610; Angew Chem Int Ed 41:1532
Tomooka K, Komine N, Nakai T (1997) Synlett 1045
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2003 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Hoppe, D., Marr, F., Brüggemann, M. (2003). Enantioselective Synthesis by Lithiation Adjacent to Oxygen and Electrophile Incorporation. In: Hodgson, D.M. (eds) Organolithiums in Enantioselective Synthesis. Topics in Organometallic Chemistry, vol 5. Springer, Berlin, Heidelberg. https://doi.org/10.1007/3-540-36117-0_4
Download citation
DOI: https://doi.org/10.1007/3-540-36117-0_4
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-00104-1
Online ISBN: 978-3-540-36117-6
eBook Packages: Springer Book Archive