Abstract
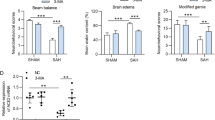
Autophagy is a self-degradative process and it plays a housekeeping role in removing misfolded or aggregated proteins, clearing damaged organelles, and eliminating intracellular pathogens. Previous studies have demonstrated that autophagy pathway was activated in brain after experimental subarachnoid hemorrhage (SAH); however, the role of autophagy in the pathogenesis of early brain injury (EBI) following SAH remains unknown. Experiment 1 aimed to investigate the time–course of the autophagy in the cortex following SAH. In experiment 2, we chose the maximum time pointof autophagy activation and assessed the effects of rapamycin (RAP, autophagy activator) and 3-methyladenine (3-MA, autophagy inhibitor) on regulation of EBI. All SAH animals were subjected to injection of 0.3 ml fresh arterial, nonheparinized blood into prechiasmatic cistern in 20 s. As a result, microtubule-associated protein light chain-3 (LC3), a biomarker of autophagosome, and beclin-1, a Bcl-2-interacting protein required for autophagy, were significantly increased at the early stage of SAH and their expressions peaked at 24 h after SAH. In RAP-treated group, the early brain damage such as brain edema, blood–brain barrier (BBB) impairment, cortical apoptosis, and clinical behavior scale was significantly ameliorated in comparison with vehicle-treated SAH rats. Conversely, 3-MA decreased expression of LC3 and beclin-1, increased the average value of brain edema and BBB disfunction, and aggravated neurological deficits. Our results suggest that autophagy pathway is activated in the brain after SAH and may play a beneficial role to EBI development.








Similar content being viewed by others
References
Hop JW, Rinkel GJ, Algra A, van Gijn J (1997) Case-fatality rates and functional outcome after subarachnoid hemorrhage: a systematic review. Stroke 28:660–664
Broderick JP, Brott TG, Duldner JE, Tomsick T, Leach A (1994) Initial and recurrent bleeding are the major causes of death following subarachnoid hemorrhage. Stroke 25:1342–1347
Ostrowski RP, Colohan AR, Zhang JH (2006) Molecular mechanisms of early brain injury after subarachnoid hemorrhage. Neurol Res 28:399–414
Cahill J, Zhang JH (2009) Subarachnoid hemorrhage: is it time for a new direction? Stroke 40:S86–S87
Glick D, Barth S, Macleod KF (2010) Autophagy: cellular and molecular mechanisms. J Pathol 221:3–12
Liu C, Gao Y, Barrett J, Hu B (2010) Autophagy and protein aggregation after brain ischemia. J Neurochem 115:68–78
Carloni S, Buonocore G, Balduini W (2008) Protective role of autophagy in neonatal hypoxia-ischemia induced brain injury. Neurobiol Dis 32:329–339
Zhang YB, Li SX, Chen XP, Yang L, Zhang YG, Liu R et al (2008) Autophagy is activated and might protect neurons from degeneration after traumatic brain injury. Neurosci Bull 24:143–149
Lee JY, He Y, Sagher O, Keep R, Hua Y, Xi G (2009) Activated autophagy pathway in experimental subarachnoid hemorrhage. Brain Res 1287:126–135
Wang Z, Chen G, Zhu WW, Bian JY, Shen XO, Zhou D (2010) Influence of simvastatin on microthrombosis in the brain after subarachnoid hemorrhage in rats: a preliminary study. Ann Clin Lab Sci 40:32–42
Smith RA, Balis FM, Ott KH, Elsberry DD, Sherman MR, Saifer MG (1995) Pharmacokinetics and tolerability of ventricularly administered superoxide dismutase in monkeys and preliminary clinical observations in familial ALS. J Neurol Sci 129:13–18
Wen YD, Sheng R, Zhang LS, Han R, Zhang X, Zhang XD et al (2008) Neuronal injury in rat model of permanent focal cerebral ischemia is associated with activation of autophagic and lysosomal pathways. Autophagy 4:762–769
Zhang JY, Peng C, Shi H, Wang S, Wang Q, Wang JZ (2009) Inhibition of autophagy causes tau proteolysis by activating calpain in rat brain. J Alzheimers Dis 16:39–47
Turkoglu OF, Eroglu H, Okutan O, Tun MK, Bodur E, Sargon MF, Oner L, Beskonakli E (2008) A comparative study of treatment for brain edema: magnesium sulphate versus dexamethasone sodium phosphate. J Clin Neurosci 15:60–65
Kohta M, Minami H, Tanaka K, Kuwamura K, Kondoh T, Kohmura E (2007) Delayed onset massive oedema and deterioration in traumatic brain injury. J Clin Neurosci 14:167–170
Yamaguchi M, Zhou C, Nanda A, Zhang JH (2004) Ras protein contributes to cerebral vasospasm in a canine double-hemorrhage model. Stroke 35:1750–1755
He Y, Wan S, Hua Y, Keep RF, Xi G (2008) Autophagy after experimental intracerebral hemorrhage. J Cereb Blood Flow Metab 28:897–905
Prunell GF, Mathiesen T, Svendgaard NA (2004) Experimental subarachnoid hemorrhage: cerebral blood flow and brain metabolism during the acute phase in three different models in the rat. Neurosurgery 54:426–436
Kusaka G, Ishikawa M, Nanda A, Granger DN, Zhang JH (2004) Signaling pathways for early brain injury after subarachnoid hemorrhage. J Cereb Blood Flow Metab 24:916–925
Claassen J, Carhuapoma JR, Kreiter KT, Du EY, Connolly ES, Mayer SA (2002) Global cerebral edema after subarachnoid hemorrhage: frequency, predictors, and impact on outcome. Stroke 33:1225–1232
Kassell NF, Torner JC, Haley EC, Jane JA, Adams HP, Kongable GL (1990) The international cooperative study on the timing of aneurysm surgery. Part 1: overall management results. J Neurosurg 73:18–36
Cuervo AM (2004) Autophagy: in sickness and in health. Trends Cell Biol 14:70–77
Kegel KB, Kim M, Sapp E, McIntyre C, Castano JG, Aronin N et al (2000) Huntingtin expression stimulates endosomal–lysosomal activity, endosome tubulation, and autophagy. J Neurosci 20:7268–7278
Bursch W, Ellinger A, Gerner C, Fröhwein U, Schulte-Hermann R (2000) Programmed cell death (PCD). Apoptosis, autophagic PCD, or others? Ann N Y Acad Sci 926:1–12
Rubinsztein DC, DiFiglia M, Heintz N, Nixon RA, Qin ZH, Ravikumar B et al (2005) Autophagy and its possible roles in nervous system diseases, damage and repair. Autophagy 1:11–22
Shintani T, Klionsky DJ (2004) Autophgy in health and disease: a doubleedged sword. Science 306:990–995
Rangaraju S, Verrier JD, Madorsky I, Nicks J, Dunn WA Jr, Notterpek L (2010) Rapamycin activates autophagy and improves myelination in explant cultures from neuropathic mice. J Neurosci 30:11388–11397
Nührenberg TG, Voisard R, Fahlisch F, Rudelius M, Braun J, Gschwend J et al (2005) Rapamycin attenuates vascular wall inflammation and progenitor cell promoters after angioplasty. FASEB J 19:246–248
Sarkar S, Ravikumar B, Floto RA, Rubinsztein DC (2009) Rapamycin and mTOR-independent autophagy inducers ameliorate toxicity of polyglutamine-expanded huntingtin and related proteinopathies. Cell Death Differ 16:46–56
Ravikumar B, Vacher C, Berger Z, Davies JE, Luo S, Oroz LG et al (2004) Inhibition of mTOR induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. Nat Genet 36:585–595
Acknowledgments
This work was supported by the 135 grant from Health Department of Jiangsu Province (RC2007081) and a grant from the China Postdoctoral Science Foundation (No.20090460085).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Zhong Wang and Xiao-Yong Shi contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, Z., Shi, XY., Yin, J. et al. Role of Autophagy in Early Brain Injury after Experimental Subarachnoid Hemorrhage. J Mol Neurosci 46, 192–202 (2012). https://doi.org/10.1007/s12031-011-9575-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-011-9575-6