Abstract
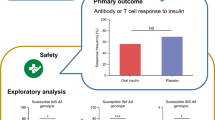
Mucosal administration of insulin represents an attractive antigen-specific therapeutic approach to preventing type 1 diabetes. It can prevent autoimmune diabetes in animal models, but although it has been shown to be safe, it has not yet been proven effective in human studies. Efficacy may depend on the dose and route at which insulin is administered, the stage in type 1 diabetes pathogenesis at which treatment is initiated, and the study cohort that is treated. We have proposed Pre-POINT (Primary Oral/intranasal INsulin Trial), a dose-finding safety and immune efficacy pilot study for primary mucosal insulin therapy in islet autoantibody-negative children at high genetic risk for type 1 diabetes who naturally first develop autoimmunity to insulin. Pre-POINT aims to identify an optimal insulin dose and route of application (orally or intranasally) that is well tolerated and can induce an immune response to insulin for additional use in a phase II/III primary prevention trial in children at risk.
Similar content being viewed by others
References and Recommended Reading
The DCCT/EDIC Research Group: Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic nephropathy: the Epidemiology of Diabetes Interventions and Complications (EDIC) study. JAMA 2003, 290:2159–2167.
Nathan DM, Lachin J, Cleary P, et al.: Intensive diabetes therapy and carotid intima-media thickness in type 1 diabetes mellitus. N Engl J Med 2003, 348:2294–2303.
The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group [no authors listed]. N Engl J Med 1993, 329:977–986.
Cyclosporin-induced remission of IDDM after early intervention. Association of 1 yr of cyclosporin treatment with enhanced insulin secretion. The Canadian-European Randomized Control Trial Group [no authors listed]. Diabetes 1988, 37:1574–1582.
Herold KC, Gitelman SE, Masharani U, et al.: Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. N Engl J Med 2002, 346:1692–1698.
Keymeulen B, Vandemeulebroucke E, Ziegler AG, et al.: Insulin needs after CD3-antibody therapy in new-onset type 1 diabetes. N Engl J Med 2005, 352:2598–2608.
Gale EA, Bingley PJ, Emmett CL, Collier T: European Nicotinamide Diabetes Intervention Trial (ENDIT): a randomised controlled trial of intervention before the onset of type 1 diabetes. Lancet 2004, 363:925–931.
Diabetes Prevention Trial — Type 1 Diabetes Study Group: Effects of insulin in relatives of patients with type 1 diabetes mellitus. N Engl J Med 2002, 346:1685–1691.
Skyler JS, Krischer JP, Wolfsdorf J, et al.: Effects of oral insulin in relatives of patients with type 1 diabetes: the Diabetes Prevention Trial-Type 1. Diabetes Care 2005, 28:1068–1076.
French MB, Allison J, Cram DS, et al.: Transgenic expression of mouse proinsulin II prevents diabetes in nonobese diabetic mice. Diabetes 1997, 46:34–39.
Thebault-Baumont K, Dubois-Laforgue D, Krief P, et al.: Acceleration of type 1 diabetes mellitus in proinsulin 2-deficient NOD mice. J Clin Invest 2003, 111:851–857.
Moriyama H, Abiru N, Paronen J, et al.: Evidence for a primary islet autoantigen (preproinsulin 1) for insulitis and diabetes in the nonobese diabetic mouse. Proc Natl Acad Sci U S A 2003, 100:10376–10381.
Jaeckel E, Lipes MA, von Boehmer H: Recessive tolerance to preproinsulin 2 reduces but does not abolish type 1 diabetes. Nat Immunol 2004, 5:1028–1035.
Nakayama M, Abiru N, Moriyama H, et al.: Prime role for an insulin epitope in the development of type 1 diabetes in NOD mice. Nature 2005, 435:220–223.
Krishnamurthy B, Dudek NL, McKenzie MD, et al.: Responses against islet antigens in NOD mice are prevented by tolerance to proinsulin but not IGRP. J Clin Invest 2006, 116:3258–3265.
Ziegler AG, Hummel M, Schenker M, Bonifacio E: Autoantibody appearance and risk for development of childhood diabetes in offspring of parents with type 1 diabetes: the 2-year analysis of the German BABYDIAB Study. Diabetes 1999, 48:460–468.
Kimpimaki T, Kulmala P, Savola K, et al.: Natural history of beta-cell autoimmunity in young children with increased genetic susceptibility to type 1 diabetes recruited from the general population. J Clin Endocrinol Metab 2002, 87:4572–4579.
Barker JM, Barriga KJ, Yu L, et al.: Prediction of autoantibody positivity and progression to type 1 diabetes: Diabetes Autoimmunity Study in the Young (DAISY). J Clin Endocrinol Metab 2004, 89:3896–3902.
Colman PG, Steele C, Couper JJ, et al.: Islet autoimmunity in infants with a type 1 diabetic relative is common but is frequently restricted to one autoantibody. Diabetologia 2000, 43:203–209.
Hummel M, Bonifacio E, Schmid S, et al.: Brief communication: early appearance of islet autoantibodies predicts childhood type 1 diabetes in offspring of diabetic parents. Ann Intern Med 2004, 140:882–886.
Kimpimaki T, Kupila A, Hamalainen AM, et al.: The first signs of beta-cell autoimmunity appear in infancy in genetically susceptible children from the general population: the Finnish Type 1 Diabetes Prediction and Prevention Study. J Clin Endocrinol Metab 2001, 86:4782–4788.
Yu L, Robles DT, Abiru N, et al.: Early expression of antiinsulin autoantibodies of humans and the NOD mouse: evidence for early determination of subsequent diabetes. Proc Natl Acad Sci U S A 2000, 97:1701–1706.
Vardi P, Ziegler AG, Mathews JH, et al.: Concentration of insulin autoantibodies at onset of type 1 diabetes. Inverse log-linear correlation with age. Diabetes Care 1988, 11:736–739.
Kent SC, Chen Y, Bregoli L, et al.: Expanded T cells from pancreatic lymph nodes of type 1 diabetic subjects recognize an insulin epitope. Nature 2005, 435:224–228.
Achenbach P, Koczwara K, Knopff A, et al.: Mature high-affinity immune responses to (pro)insulin anticipate the autoimmune cascade that leads to type 1 diabetes. J Clin Invest 2004, 114:589–597.
Chaillous L, Lefevre H, Thivolet C, et al.: Oral insulin administration and residual beta-cell function in recent-onset type 1 diabetes: a multicentre randomised controlled trial. Diabete Insuline Orale group. Lancet 2000, 356:545–549.
Pozzilli P, Pitocco D, Visalli N, et al.: No effect of oral insulin on residual beta-cell function in recent-onset type I diabetes (the IMDIAB VII). IMDIAB Group. Diabetologia 2000, 43:1000–1004.
Harrison LC, Honeyman MC, Steele CE, et al.: Pancreatic [beta]-cell function and immune responses to insulin after administration of intranasal insulin to humans at risk for type 1 diabetes. Diabetes Care 2004, 27:2348–2355.
Kupila A, Sipila J, Keskinen P, et al.: Intranasally administered insulin intended for prevention of type 1 diabetes—a safety study in healthy adults. Diabetes Metab Res Rev 2003, 19:415–420.
Faria AMC, Weiner HL: Oral tolerance. Immunol Rev 2005, 206:232–259.
Harrison LC, Hafler DA: Antigen-specific therapy for autoimmune disease. Curr Opin Immunol 2000, 12:704–711.
Gottlieb PA, Eisenbarth GS: Insulin-specific tolerance in diabetes. Clin Immunol 2002, 102:2–11.
Mayer L, Shao L: Therapeutic potential of oral tolerance. Nat Rev Immunol 2004, 4:407–419.
Bergerot I, Fabien N, Maguer V, Thivolet C: Oral administration of human insulin to NOD mice generates CD4+ T cells that suppress adoptive transfer of diabetes. J Autoimmun 1994, 7:655–663.
Harrison LC, Dempsey-Collier M, Kramer DR, Takahashi K: Aerosol insulin induces regulatory CD8 gamma delta T cells that prevent murine insulin-dependent diabetes. J Exp Med 1996, 184:2167–2174.
Zhang ZJ, Davidson L, Eisenbarth G, Weiner HL: Suppression of diabetes in nonobese diabetic mice by oral administration of porcine insulin. Proc Natl Acad Sci U S A 1991, 88:10252–10256.
Maron R, Melican NS, Weiner HL: Regulatory Th2-type T cell lines against insulin and GAD peptides derived from orally-and nasally-treated NOD mice suppress diabetes. J Autoimmun 1999, 12:251–258.
Daniel D, Wegmann DR: Protection of nonobese diabetic mice from diabetes by intranasal or subcutaneous administration of insulin peptide B-(9-23). Proc Natl Acad Sci U S A 1996, 93:956–960.
von Herrath MG, Dyrberg T, Oldstone MB: Oral insulin treatment suppresses virus-induced antigen-specific destruction of beta cells and prevents autoimmune diabetes in transgenic mice. J Clin Invest 1996, 98:1324–1331.
Hancock WW, Polanski M, Zhang J, et al.: Suppression of insulitis in non-obese diabetic (NOD) mice by oral insulin administration is associated with selective expression of interleukin-4 and-10, transforming growth factor-beta, and prostaglandin-E. Am J Pathol 1995, 147:1193–1199.
Bergerot I, Fabien N, Mayer A, Thivolet C: Active suppression of diabetes after oral administration of insulin is determined by antigen dosage. Ann N Y Acad Sci 1996, 778:362–367.
Maron R, Guerau-de-Arellano M, Zhang X, Weiner HL: Oral administration of insulin to neonates suppresses spontaneous and cyclophosphamide induced diabetes in the NOD mouse. J Autoimmun 2001, 16:21–28.
Monetini L, Cavallo MG, Sarugeri E, et al.: Cytokine profile and insulin antibody IgG subclasses in patients with recent onset type 1 diabetes treated with oral insulin. Diabetologia 2004, 47:1795–1802.
Weiner HL, Mackin GA, Matsui M, et al.: Double-blind pilot trial of oral tolerization with myelin antigens in multiple sclerosis. Science 1993, 259:1321–1324.
Trentham DE, Dynesius-Trentham RA, Orav EJ, et al.: Effects of oral administration of type II collagen on rheumatoid arthritis. Science 1993, 261:1727–1730.
Bonifacio E, Hummel M, Walter M, et al.: IDDM1 and multiple family history of type 1 diabetes combine to identify neonates at high risk for type 1 diabetes. Diabetes Care 2004, 27:2695–2700.
Aly TA, Ide A, Barker JM, et al.: HLA genotype and haplotype sharing determine extremely high risk of early childhood type 1 diabetes. Proc Natl Acad Sci U S A 2006, 103:14074–14079.
Walter M, Albert E, Conrad M, et al.: IDDM2/insulin VNTR modifies risk conferred by IDDM1/HLA for development of type 1 diabetes and associated autoimmunity. Diabetologia 2003, 46:712–720.
Laine AP, Hermann R, Knip M, et al.: The human leukocyte antigen genotype has a modest effect on the insulin gene polymorphism-associated susceptibility to type 1 diabetes in the Finnish population. Tissue Antigens 2004, 63:72–74.
Steck AK, Bugawan TL, Valdes AM, et al.: Association of non-HLA genes with type 1 diabetes autoimmunity. Diabetes 2005, 54:2482–2486.
Bonifacio E, Scripoli M, Kredel K, et al.: Early autoantibody responses in prediabetes are IgG1 dominated and suggest antigen-specific regulation. J Immunol 1999, 163:525–532.
Barker JM, McFann KK, Orban T: Effect of oral insulin on insulin autoantibody levels in the Diabetes Prevention Trial Type 1 oral insulin study. Diabetologia 2007, 50:1603–1606.
Roep BO: Standardization of T-cell assays in Type I diabetes. Immunology of Diabetes Society T-cell Committee. Diabetologia 1999, 42:636–637.
Arif S, Tree TI, Astill TP, et al.: Autoreactive T cell responses show proinflammatory polarization in diabetes but a regulatory phenotype in health. J Clin Invest 2004, 113:451–463.
Danke NA, Yang J, Greenbaum C, Kwok WW: Comparative study of GAD65-specific CD4+ T cells in healthy and type 1 diabetic subjects. J Autoimmun 2005, 25:303–311.
Monti P, Scripoli M, Rigamonti A, et al.: Evidence for in vivo primed and expanded autoreactive T cells as a specific feature of patients with type 1 diabetes. J Immunol 2007, 179:5785–5792.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Achenbach, P., Barker, J. & Bonifacio, E. Modulating the natural history of type 1 diabetes in children at high genetic risk by mucosal insulin immunization. Curr Diab Rep 8, 87–93 (2008). https://doi.org/10.1007/s11892-008-0017-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11892-008-0017-y