Abstract
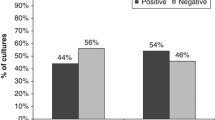
The increase in the prevalence of antibiotic resistance in Helicobacter pylori had a drastic effect on successful treatment. Up-to-date information on H. pylori antibiotic therapy in Iran is still limited. In this study, we aim to determine the prevalence of antibiotic resistance among the H. pylori strains. Furthermore, the possibility of using fluoroquinolones for antibiotic treatment was investigated. Antral biopsy specimens obtained from dyspeptic patients were investigated for H. pylori. Bacterial culture and susceptibility tests were done based on standard methods. H. pylori ATCC 43504 was used as a quality control. In the current study, 30 H. pylori strains were selected randomly and retested to confirm our susceptibility tests. Of 170 patients, 150 were identified as positive for H. pylori (88.2%). In this study, 150 single colonies of H. pylori strains [81 women (54%), 69 men (46%); mean age 38.6; aged 21–70 years] were collected. Primary resistance of H. pylori isolates were clarithromycin (34%), metronidazole (78.6%), tetracycline (9.3%), amoxicillin (10%), levofloxacin (5.3%) and moxifloxacine (4.6%). In conclusion, our results show that we are confronting a new generation of resistant strains of H. pylori in Iran. This alarming finding indicates an urgent need for introduction of new effective antibiotics in our country. Since the majority of clinicians prefer to continue with the ineffective antibiotics as therapeutic regimens, they must also be prepared to deal with treatment failures.

Similar content being viewed by others
References
O’Connor A, Gisbert J, McNamara D, O’Morain C (2010) Treatment of Helicobacter pylori infection 2010 Helicobacter 15:46–52.
Mansour-Ghanaei F, YousefiMashhour M, Joukar F, Sedigh M, Bagher-Zadeh AH, Jafarshad R (2009) Prevalence of Helicobacter Pylori infection among children in Rasht, Northern Iran. Middle East J Dig Dis 1:84–88
Goldman C, Barrado A, Janjetic M, Balcarce N, CuetoRua E, Oshiro M et al (2006) Factors associated with H. pylori epidemiology in symptomatic children in Buenos Aires, Argentina. World J Gastroenterol 12:5384–5388
Douraghi M, Mohammadi M, Oghalaie A, Abdirad A, Mohagheghi MA et al (2008) dupA as a risk determinant in Helicobacter pylori infection. J Med Microbiol 57:554–562
Salehi Z, HalimiJelodar M, Rassa M, Ahaki M, Mollasalehi H, Mashayekhi F (2009) Helicobacter pylori cagA status and peptic ulcer disease in Iran. Dig Dis Sci 54:608–613
Megraud F (2004) H. pylori antibiotic resistance: prevalence, importance, and advances in testing. Gut 53:1374–1384
Saberi-Firoozi M, Nejabat M (2006) Experiences with Helicobacter pylori treatment in Iran. Iran J Med Sci 31:181–185
Malekzadeh R, Derakhshan MH, Malekzadeh Z (2009) Gastric cancer in Iran: epidemiology and risk factors. Arch Iran Med 12:576–583
TalebiBezminAbadi A, MohabatiMobarez A, Taghvaei T, Wolfram L (2010) Antibiotic resistance of Helicobacter pylori in Mazandaran, North of Iran. Helicobacter 15:505–509
HaghiTomatari F, MohabbatiMobarez A, Amini M, Hosseini D, TalebiBezminAbadi A (2010) Helicobacter pylori resistance to metronidazole and clarithromycin in dyspeptic patients in Iran. IRCMJ 12:409–412
Fakheri H, Merat S, Hosseini V, Malekzadeh R (2004) Low-dose furazolidone in triple and quadruple regimens for Helicobacter pylori eradication. Aliment Pharmacol Ther 19:89–93
John Albert M, Al-Mekhaizeem K, Neil L, Dhar R et al (2006) High prevalence and level of resistance to metronidazole, but lack of resistance to other antimicrobials in Helicobacter pylori, isolated from a multiracial population in Kuwait. Aliment Pharmacol Ther 24:1359–1366
Gatta L, Zullo A, Perna F, Ricci C, De Francesco V, Tampieri A et al (2005) A 10-day levofloxacin-based triple therapy in patients who have failed two eradication courses. Aliment Pharmacol Ther 22:45–49
Zullo A, De Francesco V, Hassan C, Panella C, Morini S, Ierardi E (2006) Second-line treatment for Helicobacter pylori eradication after sequential therapy failure: a pilot study. Therapy 3:251–254
Kuei-Hsiang Hung, Bor-Shyang Sheu, Wei-Lun Chang, Hsiu-Mei Wu, Chin-Cheng Liu, Jiunn-Jong Wu (2009) Prevalence of primary fluoroquinolone resistance among clinical isolates of Helicobacter pylori at a University Hospital in Southern Taiwan. Helicobacter 14:61–65
Graham DY, Abudayyeh S, El-Zimaity HM, Hoffman J, Reddy R, Opekun AR (2006) Sequential therapy using high-dose esomeprazole–amoxicillin followed by gatifloxacin for Helicobacter pylori infection. Aliment Pharmacol Ther 24:845–850
Nista EC, Candelli M, Zocco MA et al (2005) Moxifloxacin-based strategies for first-line treatment of Helicobacter pylori infection. Aliment Pharmacol Ther 21:1241–1247
Boyanova L, Gergova G, Knikolov R, Davidkov L, Kamburov V, Jelev C, Mitov I (2008) Prevalence and evolution of Helicobacter pylori resistance to 6 antimicrobial agents over 12 years and correlation between susceptibility testing methods. Diagn Microbiol Infect Dis 60:409–415
Cattoir V, Nectoux J, Lascols C, Deforges L, Delchier JC, Megraud F, Soussy CJ, Cambau E (2007) Update on fluoroquinolone resistance in Helicobacter pylori: new mutations leading to resistance and first description of a gyrA polymorphism associated with hypersusceptibility. Int J Antimicrob Agent 29:389–396
Sherif M, Mohran Z, Fathy H, Rockabrand DM, Rozmajzl PJ, Frenck RW (2004) Universal high-level primary metronidazole resistance in Helicobacter pylori isolated from children in Egypt. J Clin Microbiol 42:4832–4834
Watanabe Y, Aoyama N, Shirasaka D, Maekawa S, Kuroda K, Miki I et al (2003) Levofloxacin-based triple therapy as a second-line treatment after failure of Helicobacter pylori eradication with standard triple therapy. Dig Liver Dis 35:711–715
Gisbert JP, De La Morena F (2006) Systematic review and meta-analysis: levofloxacin-based rescue regimens after Helicobacter pylori treatment failure. Aliment Pharmacol Ther 23:35–44
Saad RJ, Schoenfeld P, Kim HM, Chey WD (2006) Levofloxacin-based triple therapy versus bismuth-based quadruple therapy for persistent Helicobacter pylori infection: a meta-analysis. Am J Gastroenterol 101:488–496
Leszczynska K, Namiot A, Namiot Z, Leszczynska JK, Jakoniuk P, Chilewicz M, Namiot DB, Kemona A, Milewski R, Bucki R (2010) Patient factors affecting culture of Helicobacter pylori isolated from gastric mucosal specimens. Advances in medical sciences 55. doi:10.2478/v10039-010-0028-1
Megraud F, Lehours P (2007) Helicobacter pylori detection and antimicrobial susceptibility testing. Clin Microbiol Rev 20:280–322
Kusters JG, ArnoudVliet HM, Kuipers EJ (2006) Pathogenesis of Helicobacter pylori infection. Clin Microbiol Rev 19:449–490
Clinical and Laboratory Standards Institute (CLSI) (2007) Performance standards for antimicrobial susceptibility testing; fifteenth information supplement. Wayne, PA
Ebrahimi-Dariani N, Mirmomen S, Mansour-Ghanaei F (2003) The efficacy of furazolidone-based quadruple therapy for eradication of Helicobacter pylori infection in Iranian patients resistant to metronidazole-based quadruple therapy. Med Sci Monit 9:PI105–8
Rafeey M, Ghotaslou R, Nikvash S, AshrafyHafez A (2007) Primary resistance in Helicobacter pylori isolated in children from Iran. J Infect Chemother 13:291–295
Rimbara E, Noguchi N, Tanabe M et al (2005) Susceptibilities to clarithromycin, amoxycillin and metronidazole of Helicobacter pylori isolates from the antrum and corpus in Tokyo, Japan, 1995–2001. Clin Microbiol Infect 11:307–311
De Francesco V, Margiotta M, Zullo A, Hassan C, Giorgio F, Burattini O, Stoppino G et al (2007) Prevalence of primary clarithromycin resistance in Helicobacter pylori strains over a 15 year period in Italy. J Antimicrob Chemother 59:783–785
Falsafi T, Mobasheri F, Nariman F, Najafi M (2004) Susceptibilities to different antibiotics of Helicobacter pylori strains isolated from patients at the Pediatric Medical Center of Tehran, Iran. J Clin Microbiol 42:387–389
Siavoshi F, Pourkhajeh AH, Merat S, Asl-Soleimani H, Heydarian E, Khatibian M et al (2000) Susceptibility of various strains of Helicobacter pylori to selected agents. Arch Iran Med 3:60–63
FallahiGh H, Maleknejad Sh (2007) Helicobacter pylori culture and antimicrobial resistance in Iran. Iran J Pediatr 74:127–130
Khashei R, Shojaei H, Adibi P, Shavakhi A, Aslani MM, Naser AD (2008) Genetic diversity and drug resistance of Helicobacter pylori strains in Isfahan, Iran. Iran J Basic Med Sci 11:174–182
Koletzko S, Richy F, Bontems P, Crone J, Kalach N, Monteiro ML, European Pediatric Task Force on Helicobacter pylori et al (2006) Prospective multicenter study on antibiotic resistance of Helicobacter pylori strains obtained from children living in Europe. Gut 55:1711–1716
Street ME, Caruana P, Caffarelli C, Magliani W, Manfredi M, Fornaroli F et al (2001) Antibiotic resistance and antibiotic sensitivity based treatment in Helicobacter pylori infection: advantages and outcome. Arch Dis Child 84:419–422
Boyanova L, Markovska R, Yordanov D, Marina M, Ivanova K, Panayotov S, Gergova G, Mitov I (2009) High prevalence of virulent Helicobacter pylori strains in symptomatic Bulgarian patients. Diagn Microbiol Infect Dis 6:374–380
Agudo S, Peerez-Perez G, Alarcon T, lopez-Brea M (2010) High prevalence of clarithromycin-resistant Helicobacter pylori strains and risk factors associated with resistance in Madrid, Spain. J Clin Microbiol 48:3703–3707
Boyanova L, Nikolov R, Lazarova E, Gergova G et al (2006) Antibacterial resistance in Helicobacter pylori strains isolated from Bulgarian children and adult patients over 9 years. J Med Microbiol 55:65–68
Acknowledgments
This project was supported financially by M Sc thesis at Tarbiat Modares University at 2007.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Talebi Bezmin Abadi, A., Ghasemzadeh, A., Taghvaei, T. et al. Primary resistance of Helicobacter pylori to levofloxacin and moxifloxacine in Iran. Intern Emerg Med 7, 447–452 (2012). https://doi.org/10.1007/s11739-011-0563-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-011-0563-1