Abstract
Purpose
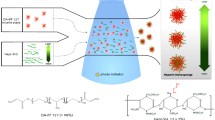
Heparin immobilized porous poly(d,l-lactic-co-glycolic acid) (PLGA) microspheres were prepared for sustained release of basic fibroblast growth factor (bFGF) to induce angiogenesis.
Materials and Methods
Porous PLGA microspheres having primary amine groups on the surface were prepared using an oil-in-water (O/W) single emulsion method using Pluronic F-127 as an extractable porogen. Heparin was surface immobilized via covalent conjugation. bFGF was loaded into the heparin functionalized (PLGA-heparin) microspheres by a simple dipping method. The bFGF loaded PLGA-heparin microspheres were tested for in vitro release and in vivo angiogenic activity.
Results
PLGA microspheres with an open-porous structure were formed. The amount of conjugated amine group onto the microspheres was 1.93 ± 0.01 nmol/mg-microspheres, while the amount of heparin was 95.8 pmol/mg-microspheres. PLGA-heparin microspheres released out bFGF in a more sustained manner with a smaller extent of initial burst than PLGA microspheres, indicating that surface immobilized heparin controlled the release rate of bFGF. Subcutaneous implantation of bFGF loaded PLGA-heparin microspheres in mice significantly induced the formation of new vascular microvessels.
Conclusions
PLGA microspheres with an open porous structure allowed significant amount of heparin immobilization and bFGF loading. bFGF loaded PLGA-HP microspheres showed sustained release profiles of bFGF in vitro, demonstrating reversible and specific binding of bFGF to immobilized heparin. They also induced local angiogenesis in vivo in an animal model.







Similar content being viewed by others
References
P. Carmeliet. Angiogenesis in health and disease. Nat. Med. 9:653–660 (2003).
P. Carmeliet. Mechanisms of angiogenesis and arteriogenesis. Nat. Med. 6:389–395 (2000).
J. Li and R. S. Kirsner. Angiogenesis in wound repair: angiogenic growth factors and the extracellular matrix. Microsc. Res. Tech. 60:107–114 (2003).
T. P. Richardson, M. C. Peters, A. B. Ennett, and D. J. Mooney. Polymeric system for dual growth factor delivery. Nat. Biotechnol. 19:1029–1034 (2001).
K. Y. Lee, M. C. Peters, K. W. Andersonm, and D. J. Mooney. Controlled growth factor release from synthetic extracellular matrices. Nature 408:998–1000 (2000).
H. Lee, R. A. Cusick, F. Browne, T. H. Kim, P. X. Ma, H. Utsunomiya, R. Langer, and J. Vacanti. Local delivery of basic fibroblast growth factor increases both angiogenesis and engraftment of hepatocytes in tissue-engineered polymer devices. Transplantation 73:1589–1593 (2002).
C. L. Casper, N. Yamaguchi, K. L. Kiick, and J. F. Rabolt. Functionalizing electrospun fibers with biologically relevant macromolecules. Biomacromolecules 6:1998–2007 (2005).
J. E. Markkanen, T. T. Rissanen, A. Kivela, and S. Yla-Herttuala. Growth factor-induced therapeutic angiogenesis and arteriogenesis in the heart-gene therapy. Cardiovasc. Res. 65:656–664 (2005).
Y. Cao, P. Lenden, D. Shima, F. Browne, and J. Folkman. In vivo angiogenic activity and hypoxia induction of heterodimers of placenta growth factor/vascular endothelial growth factor. J. Clin. Invest. 98:2507–2511 (1996).
J. L. Cleland, E. T. Duenas, A. Park, A. Daugherty, J. Kahn, and J. Kowalski. Development of poly-(d,l-lactide-coglycolide) microsphere formulations containing recombinant human vascular endothelial growth factor to promote local angiogenesis. J. Control. Release 72:13–24 (2001).
P. B. O'Donnell and J. W. McGinity. Preparation of microspheres by the solvent evaporation technique. Adv. Drug Del. Rev. 28:25–42 (1997).
H. K. Kim and T. G. Park. Microencapsulation of human growth hormone within biodegradable polyester microspheres: protein aggregation stability and incomplete release mechanism. Biotech. Bioeng. 65:659–667 (1999).
M. van de Weert, W. E. Hennink, and W. Jiskoot. Protein instability in poly(lactic-co-glycolic acid) microparticles. Pharm. Res. 17:1159–1167 (2000).
R. Sasisekharan, S. Ernst, and G. Venkataraman. On the regulation of fibroblast growth factor activity by heparin-like glycosaminoglycans. Angiogenesis 1:45–54 (1997).
X. H. Wang, D. P. Li, W. J. Wang, Q. L. Feng, F. Z. Cui, Y. X. Xu, and X. H. Song. Covalent immobilization of chitosan and heparin on PLGA surface. Int. J. Biol. Macromol. 33:95–100 (2003).
A. Perets, Y. Baruch, F. Weisbuch, G. Shoshany, G. Neufeld, and S. Cohen. Enhancing the vascularization of three-dimensional porous alginate scaffolds by incorporating controlled release basic fibroblast growth factor microspheres. J. Biomed. Mater. Res. 65A:489–497 (2003).
G. C. M. Steffens, C. Yao, P. Prevel, M. Markowicz, P. Schenck, E. M. Noah, and N. Pallua. Modulation of angiogenic potential of collagen matrices by covalent incorporation of heparin and loading with vascular endothelial growth factor. Tissue Eng. 10:1502–1509 (2004).
H. K. Kim, H. J. Chung, and T. G. Park. Biodegradable polymeric microspheres with “open/closed” pores for sustained release of human growth hormone. J. Control. Release 112:167–174 (2006).
G. T. Hermanson. Bioconjugate Techniques, Academic, San Diego, California, 1996.
J. J. Yoon, S. H. Song, D. S. Lee, and T. G. Park. Immobilization of cell adhesive RGD peptide onto the surface of highly porous biodegradable polymer scaffolds fabricated by a gas foaming/salt leaching method. Biomaterials 25:5613–5620 (2004).
O. Jeon, S. W. Kang, H. W. Lim, J. H. Chung, and B. S. Kim. Long-term and zero-order release of basic fibroblast growth factor from heparin-conjugated poly(l-lactide-co-glycolide) nanospheres and fibrin gel. Biomaterials 27:1598–1607 (2006).
I. Kang, D. K. Baek, Y. M. Lee, and Y. K. Sung. Synthesis and surface characterization of heparin-immobilized polyurethanes. J. Polym. Sci., Part A: Polym. Chem. 36:2331–2338 (1998).
V. D. Nadkarni, A. Pervin, and R. J. Lindhardt. Directional immobilization of heparin onto beaded supports. Anal. Biochem. 222(1):59–67 (1994).
T. Magoshi, H. Ziani-Cherif, S. Ohya, Y. Nakayama, and T. Matsuda. Thermosensitive heparin coating: heparin conjugated with poly(N-isopropylacrylamide) at one terminus. Langmuir 18:4862–4872 (2002).
P. K. Smith, A. K. Mallia, and G. T. Hermanson. Colorimetric method for the assay of heparin content in immobilized heparin preparations. Anal. Biochem. 109:466–473 (1980).
G. Caponetti, J. S. Hrkach, B. Kriwet, M. Poh, N. Lotan, P. Colombo, and R. Langer. Microparticles of novel branched copolymers of lactic acid and amino acids: preparation and characterization. J. Pharm. Sci. 88:136–141 (1999).
A. Lamprecht, U. F. Schafer, and C. M. Lehr. Visualization and quantification of polymer distribution in microcapsules by confocal laser scanning microscopy (CLSM). Int. J. Pharm. 196:223–226 (2000).
A. Lamprecht, U. F. Schafer, and C. M. Lehr. Characterization of microcapsules by confocal laser scanning microscopy: structure, capsule wall composition and encapsulation rate. Eur. J. Pharm. Biopharm. 49:1–9 (2000).
L. D. Thompson, M. W. Patoliano, and B. A. Springer. Energetic characterization of the basic fibroblast growth factor-heparin interaction: identification of the heparin binding domain. Biochemistry 33:3831–3840 (1994).
M. Fugita, M. Ishihara, M. Simizu, K. Obara, T. Ishizuka, Y. Saito, H. Yura, Y. Morimoto, B. Takase, T. Matsui, M. Kikuchi, and T. Maehara. Vascularization in vivo caused by the controlled release of fibroblast growth factor-2 from an injectable chitosan/non-anticoagulant heparin hydrogel. Biomaterials 25:699–706 (2004).
Acknowledgements
This research was supported by grants from the Ministry of Commerce, Industry and Energy (10011366) and from the Ministry of Science and Technology, National Research Laboratory Program, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chung, H.J., Kim, H.K., Yoon, J.J. et al. Heparin Immobilized Porous PLGA Microspheres for Angiogenic Growth Factor Delivery. Pharm Res 23, 1835–1841 (2006). https://doi.org/10.1007/s11095-006-9039-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-006-9039-9