Purpose
The purpose of this study was to determine the systemic absorption and the release of etoposide in cervical tissue administered via a vaginal ovule to women diagnosed with cervical intraepithelial lesions associated with human papillomavirus (HPV).
Methods
Fifteen women with low- and high-grade intraepithelial neoplasia confirmed by colposcopic test received a 50-mg intravaginal etoposide dose three times a week for 3 weeks. At the end of the study period, paralleled with the last ovule administered, blood samples were collected over a period of 24 h, and in situ cervical samples were obtained at 3 and 10 h after drug administration. Etoposide concentrations were determined in plasma and in in situ cervical samples using the high-performance liquid chromatography method with electrochemical detection.
Results
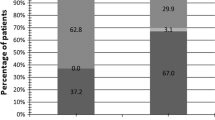
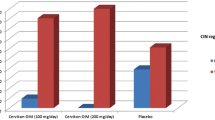
Pharmacokinetic analyses of plasma data indicated low or lack of systemic exposure of etoposide after the vaginal administration. Nevertheless, high concentrations of etoposide were found in all in situ cervical samples, indicating that etoposide could be released from its pharmaceutical formulation.
Conclusions
The results of the study suggest that the etoposide administered as intravaginal ovule is safe and tolerable and apparently could be a suitable option in patients with cervical intraepithelial neoplasia. Clinical results and the true impact on HPV infection and evolution of dysplasia need to be confirmed.




Similar content being viewed by others
References
F. X. Bosch (2003) ArticleTitleEpidemiology of human papillomavirus infections: new option for cervical cancer prevention Salud Publica Mex. 45 S326–S329 Occurrence Handle14746025
A. T. Lorincz (2003) ArticleTitleScreening for cervical cancer: new alternatives and research Salud Publica Mex. 45 S376–S387 Occurrence Handle14746031
M. Fukushima Y. Yamakawa S. Shimano M. Hashimoto Y. Sawada K. Fujinaga (1990) ArticleTitleThe physical state of human papillomavirus 16 DNA in cervical carcinoma and cervical intraepithelial neoplasia Cancer 66 2155–2161 Occurrence Handle1:CAS:528:DyaK3MXitVClu7Y%3D Occurrence Handle2171746
A. Iwasawa P. Nieminen M. Lehtinen J. Paavoneen (1996) ArticleTitleHuman papillomavirus DNA in uterine cervix squamous cell carcinoma and adenocarcinoma detected by polymerase chain reaction Cancer 77 2275–2279 Occurrence Handle10.1002/(SICI)1097-0142(19960601)77:11<2275::AID-CNCR14>3.0.CO;2-U Occurrence Handle1:STN:280:DyaK283isVyqsQ%3D%3D Occurrence Handle8635095
H. Hausen (2000) ArticleTitlePapillomaviruses causing cancer: evasion from host-cell control in early events in carcinogenesis J. Natl. Cancer Inst. 92 690–698 Occurrence Handle10.1093/jnci/92.9.690 Occurrence Handle10793105
H. Furumoto M. Irahara (2002) ArticleTitleHuman papilloma virus (HPV) and cervical cancer J. Med. Investig. 49 124–133
M. M. Holmes S. H. Weaver S. T. Vermillion (1999) ArticleTitleA randomized, double-blind, placebo-controlled trial of 5-fluoruracil for the treatment of cervicovaginal human papillomavirus Infect. Dis. Obstet. Gynecol. 7 186–189 Occurrence Handle10.1002/(SICI)1098-0997(1999)7:4<186::AID-IDOG4>3.0.CO;2-Z Occurrence Handle1:STN:280:DyaK1Mzns1GjtA%3D%3D Occurrence Handle10449266
Y. Liu A. McKalip B. Herman (2000) ArticleTitleHuman papillomavirus type 16 E6 and HPV-16 E6/E7 sensitize human keratinocytes to apoptosis induced by chemotherapeutic agents: roles of p53 and caspase activation J. Cell. Biochem. 78 334–349 Occurrence Handle1:CAS:528:DC%2BD3cXkt1aqtLw%3D Occurrence Handle10842327
G. L. Chen L. Yang T. C. Rowe B. D. Halligan K. M. Tewey L. F. Liu (1984) ArticleTitleNonintercalitive antitumor drugs interfere with the breakage reunion reaction of mammalian DNA topoisomerase II J. Biol. Chem. 259 13560–13566 Occurrence Handle1:CAS:528:DyaL2MXhslahtw%3D%3D Occurrence Handle6092381
M. Sioud P. Forterre A. M. Recondo Particlede (1987) ArticleTitleEffects of the antitumor drug VP16 (etoposide) on the archaebacterial. Halobacterium GRB1.7 Kb plasmid in vivo Nucleic Acids Res. 15 8217–8234 Occurrence Handle1:CAS:528:DyaL1cXht1yn Occurrence Handle3671082
J. M. Henwood R. N. Brogden (1990) ArticleTitleEtoposide: a review of itspharmacodynamic and pharmacokinetics properties and therapeutic potential in combination chemotherapy of cancer Drugs 39 438–490 Occurrence Handle1:STN:280:DyaK3c3ivVWquw%3D%3D Occurrence Handle2184009 Occurrence Handle10.2165/00003495-199039030-00008
D. Busse G. Wurthwein C. Hinske G. Hempel M. F. Fromm M. Eichelbaum H. K. Kroemer F. W. Busch (2002) ArticleTitlePharmacokinetics of intravenous etoposide in patients with breast cancer: influence of dose escalation and cyclophosphamide and doxorubicin coadministration Naunyn-Schmiedebergs Arch. Pharmacol. 366 218–225 Occurrence Handle1:CAS:528:DC%2BD38XlvVCjs70%3D Occurrence Handle12172704
A. Ballestrero A. Rubagotti P. Stura F. Ferrando D. Amoroso M. Rinaldini P. Sismondi F. Genta M. Mesiti F. Brema F. Patrone F. Boccardo InstitutionalAuthorNameGROETA (Italian Breast Cancer Adjuvant study) (2001) ArticleTitleAdjuvant chemotherapy with high-dose cyclophosphamide, etoposide and cisplatin intensification without progenitor cell support in breast cancer patients with ten or more involved nodes: 5 year results of a pilot trial Oncology 60 221–227 Occurrence Handle10.1159/000055322 Occurrence Handle1:CAS:528:DC%2BD3MXjvVWqtLw%3D Occurrence Handle11340373
H. Kobayashi Y. Kawashima (1989) ArticleTitleA case of recurrent uterine cervical cancer successfully treated with etoposide Gan To Kagaku Ryoho 16 3279–3282 Occurrence Handle1:STN:280:DyaL1MzptFGnug%3D%3D Occurrence Handle2782919
M. Ikeda K. Noda M. Hiura T. Tamaya M. Ozaki M. Hatae M. Ozawa T. Yamabe K. Tanaka R. Izumi H. Okada Y. Ogita H. Hoshiai (1998) ArticleTitleLate phase II trial oral etoposide administered for 21 consecutive days in patients with cervical cancer. ETP 21 Study Group-Cervical Cancer Group Gan To Kagaku Ryoho 25 2249–2257 Occurrence Handle1:STN:280:DyaK1M%2FpslOmtQ%3D%3D Occurrence Handle9881082
K. Noda K. Tanaka M. Ozaki K. Hirabayasi K. Hasegawa I. Nishiya M. Yakushiji R. Izumi Y. Tomoda Y. Ogita H. Sugimori T. Yamabe R. Kudo A. Yajima Y. Terashima S. Fujii Y. Suzuoki H. Okada I. Kono K. Ochiai T. Yamamoto M. Ikeda M. Umesaki T. Saito H. Niitani (1998) ArticleTitleEarly phase II trial of oral etoposide administered for 21 consecutive days in patients with cervical or ovarian cancer. ETP 21 Study Group-Cervical-Ovarian Cancer Group Gan To Kagaku Ryoho 25 2061–2068 Occurrence Handle1:STN:280:DyaK1M%2Fls1artQ%3D%3D Occurrence Handle9838908
P. G. Rose J. A. Blessing L. Van S. Waggonner (1998) ArticleTitleProlonged oral etoposide in recurrent or advanced squamous cell carcinoma of the cervix: a gynecologic oncology group study Gynecol. Oncol. 70 263–266 Occurrence Handle1:CAS:528:DyaK1cXmsVClt7w%3D Occurrence Handle9740702
F. L. Meyskens E. Surwit T. E. Moon J. M. Childers J. R. Davis R. T. Dorr C. S. Johnson D. S. Alberto (1994) ArticleTitleEnhancement of regression of cervical intraepithelial neoplasia II (moderate dysplasia) with topically applied all-trans-retinoic acid: a randomized trial J. Natl. Cancer Inst. 86 539–543 Occurrence Handle8133537
M. Maiman D. H. Watts J. Anderson P. Clax M. Merino M. A. Kendall (1999) ArticleTitleVaginal 5-fluoruracil for high-grade cervical dysplasia in human immunodeficiency virus infection; a randomized trial Obstet. Gynecol. 94 954–961 Occurrence Handle10.1016/S0029-7844(99)00407-X Occurrence Handle1:CAS:528:DyaK1MXntlensbg%3D Occurrence Handle10576182
M. M. Alper B. N. Barwin W. M. McLean I. J. McGilveray S. Sved (1985) ArticleTitleSystemic absorption of metronidazole by the vaginal route Obstet. Gynecol. 65 781–784 Occurrence Handle1:STN:280:DyaL2M3hsVOlsQ%3D%3D Occurrence Handle4000568
C. Hoffman N. Focke G. Franke M. Zschiesche W. Siegmund (1995) ArticleTitleComparative bioavailability of metronidazole formulations (Vagimid) after oral and vaginal administration Int. J. Clin. Pharmacol. Ther. 33 232–239
J. Pérez-Urizar Y. F. Picazo B. Navarro-González F. J. Flores-Murrieta G. Castañeda-Hernández (1996) ArticleTitleA new rapid and economic high performance liquid chromatographic assay with electrochemical detection for determination of etoposide (VP16) in human plasma samples J. Liq. Chromatogr. Relat. Technol. 19 939
Estadísticas de Mortalidad en México: muertes registradas en el año 2002. Salud Publica Mex. 46:169–185 (2004).
K. Bodner B. Bodner-Adler F. Wierrani O. Kimberger C. Denk W. Grunberger (2002) ArticleTitleIs therapeutic conization sufficient to eliminate a high-risk HPV infection of the uterine cervix? A clinicopathological analysis Anticancer Res. 22 3733–3736 Occurrence Handle12552985
K. Nakayama Y. Sano Y. Endo H. Sato T. Ohyama M. Yamaguchi (1992) ArticleTitleClinical studies on intravaginal administration of CDDP for dysplasia, carcinoma in situ and microinvasive carcinoma of the uterine cervix Nippon Sanka Fujinka Gakkai Zasshi 44 208–214 Occurrence Handle1:STN:280:DyaK387osFehtw%3D%3D Occurrence Handle1545173
J. L. Gonzalez-Sanchez G. Flores-Murrieta J. Chavez-Brambilia J. M. Deolarte-Manzano A. F. Andrade-Manzano (2002) ArticleTitleTopical 5-Fluoruracil for treatment of vaginal intraepithelial neoplasms Ginecol. Obstet. Mex. 70 244–247 Occurrence Handle12148464
M. L. Slevin P. I. Clark S. P. Joel S. Malik R. J. Osborne W. M. Gregory D. G. Lowe R. H. Reznek P. F. Wrigley (1989) ArticleTitleA randomised trial to evaluate the effect of schedule on the activity of etoposide in small-cell lung cancer J. Clin. Oncol. 7 1333–1340 Occurrence Handle1:STN:280:DyaL1MzmtFGluw%3D%3D Occurrence Handle2549204
P. I. Clark M. L. Slevin S. P. Joel R. J. Osborne D. I. Talbot P. W. Johnson R. Reznek T. Masud W. Gregory P. F. Wrigley (1994) ArticleTitleA randomized trial of two etoposide schedules in small-cell lung cancer: the influence of pharmacokinetics on efficacy and toxicity J. Clin. Oncol. 12 1427–1435 Occurrence Handle1:STN:280:DyaK2c3ptlSksw%3D%3D Occurrence Handle8021734
P. I. Clark S. P. Joel M. L. Slevin (1989) ArticleTitleA pharmacokinetics hypothesis for the clinical efficacy of etoposide in small-cell lung cancer Proc. ASCO 8 66
J. D. Jonkman-de Vries H. Rosing O. Tellingen Particlevan C. Neef A. C. Dubbelman A. Bult W. W. Bokkel-Huinink Particleten B. G. Taal J. H. Beijnen (1995) ArticleTitlePharmacokinetics of etoposide after oral and intravenous administration in patients with gastric carcinoma Clin. Drug Investig. 10 86
J. L. Aguilar-Ponce Y. Flores-Picazo J. Pérez-Urizar G. Castañeda-Hernández J. W. Zinser-Sierra A. Dueñas-Gonzalez E. Calderon-Flores B. A. Segura-Pacheco J. Garza-Salazar Particlede la (1999) ArticleTitleBioavailability of etoposide after oral administration of the solution marketed for intravenous use: therapeutic and pharmaeconomic perspectives Arch. Med. Res. 30 212–215 Occurrence Handle1:STN:280:DyaK1Mzls1ykuw%3D%3D Occurrence Handle10427872
Acknowledgment
This study was supported by the Research Medical Division of Laboratorios Lemery S.A. de C.V. (México City).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
García-López, P., Coll, M., Cervera, E. et al. The Systemic Absorption of Etoposide after Intravaginal Administration in Patients with Cervical Intraepithelial Lesions Associated with Human Papillomavirus Infection. Pharm Res 23, 378–383 (2006). https://doi.org/10.1007/s11095-005-9142-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-005-9142-3