Abstract
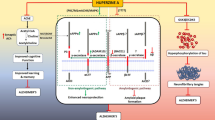
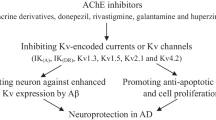
The use of acetylcholinesterase inhibitors to decrease the breakdown of the neurotransmitter acetylcholine has been the main symptomatic therapy for mild to moderate Alzheimer’s patients, though the etiology of Alzheimer’s disease remains unclear and seems to involve multiple factors. Further evidence has indicated that some of these acetylcholinesterase inhibitors also have non-cholinergic functions on the pathogenesis of Alzheimer’s disease including the formation and deposition of β-amyloid. Huperzine A, a potent and reversible inhibitor of acetylcholinesterase that was initially isolated from a Chinese herb, has been found to improve cognitive deficits in a broad range of animal models and has been used for Alzheimer’s disease treatment in China. The novel neuroprotective effects of huperzine A might yield beneficial effects in Alzheimer’s disease therapy and provide a potential template for the design of new selective and powerful anti-Alzheimer’s drugs. The present paper gives an overview on the neuroprotective effects of huperzine A beyond its acetylcholinesterase inhibition. These effects include regulating β-amyloid precursor protein metabolism, protecting against β-amyloid-mediated oxidative stress and apoptosis. The structure–function relationship of huperzine A is also discussed.

Similar content being viewed by others
References
Alvarez A, Alarcon R, Opazo C, Campos EO, Munoz FJ, Calderon FH, Dajas F, Gentry MK, Doctor, BP, De Mello FG, Inestrosa NC (1998) Stable complexes involving acetylcholinesterase and amyloid-beta peptide change the biochemical properties of the enzyme and increase the neurotoxicity of Alzheimer’s fibrils. J Neurosci 18:3213–3223
Alvarez A, Opazo C, Alarcon R, Garrido J, Inestrosa NC (1997) Acetylcholinesterase promotes the aggregation of amyloid-beta-peptide fragments by forming a complex with the growing fibrils. J Mol Biol 272:348–361
Anderson AJ, Pike CJ, Cotman CW (1995) Differential induction of immediate early gene proteins in cultured neurons by beta-amyloid (A beta): association of c-Jun with A beta-induced apoptosis. J Neurochem 65:1487–1498
Ashani Y, Peggins JO, 3rd Doctor BP (1992) Mechanism of inhibition of cholinesterases by huperzine A. Biochem Biophys Res Commun 184:719–726
Bartolini M, Bertucci C, Cavrini V, Andrisano V (2003) beta-Amyloid aggregation induced by human acetylcholinesterase: inhibition studies. Biochem Pharmacol 65:407–416
Behl C, Davis JB, Lesley R, Schubert D (1994) Hydrogen peroxide mediates amyloid beta protein toxicity. Cell 77:817–827
Blass JP (2001) Brain metabolism and brain disease: is metabolic deficiency the proximate cause of Alzheimer dementia? J Neurosci Res 66:851–856
Busciglio J, Gabuzda DH, Matsudaira P, Yankner BA (1993) Generation of beta-amyloid in the secretory pathway in neuronal and nonneuronal cells. Proc Natl Acad Sci USA 90:2092–2096
Buxbaum JD, Koo EH, Greengard P (1993) Protein phosphorylation inhibits production of Alzheimer amyloid beta/A4 peptide. Proc Natl Acad Sci USA 90:9195–9198
Casley CS, Land JM, Sharpe MA, Clark JB, Duchen MR, Canevari L (2002) Beta-amyloid fragment 25–35 causes mitochondrial dysfunction in primary cortical neurons. Neurobiol Dis 10:258–267
Colletier JP, Sanson B, Nachon F, Gabellieri E, Fattorusso C, Campiani G, Weik M (2006) Conformational flexibility in the peripheral site of torpedo californica acetylcholinesterase revealed by the complex structure with a bifunctional inhibitor. J Am Chem Soc 128:4526–4527
De Ferrari GV, Canales MA, Shin I, Weiner LM, Silman I, Inestrosa NC (2001) A structural motif of acetylcholinesterase that promotes amyloid beta-peptide fibril formation. Biochemistry 40:10447–10457
DeKosky ST, Scheff SW (1990) Synapse loss in frontal cortex biopsies in Alzheimer’s disease: correlation with cognitive severity. Ann Neurol 27:457–464
Dvir H, Jiang HL, Wong DM, Harel M, Chetrit M, He XC, Jin GY, Yu GL, Tang XC, Silman I, Bai DL, Sussman JL (2002) X-ray structures of Torpedo californica acetylcholinesterase complexed with (+)-huperzine A and (-)-huperzine B: structural evidence for an active site rearrangement. Biochemistry 41:10810–10818
Er E, Oliver L, Cartron PF, Juin P, Manon S, Vallette FM (2006) Mitochondria as the target of the pro-apoptotic protein Bax. Biochim Biophys Acta 1757:1301–1311
Esch FS, Keim PS, Beattie EC, Blacher RW, Culwell AR, Oltersdorf T, McClure D, Ward PJ (1990) Cleavage of amyloid beta peptide during constitutive processing of its precursor. Science 248:1122–1124
Fayuk D, Yakel JL (2004) Regulation of nicotinic acetylcholine receptor channel function by acetylcholinesterase inhibitors in rat hippocampal CA1 interneurons. Mol Pharmacol 66:658–666
Fuentealba RA, Farias G, Scheu J, Bronfman M, Marzolo MP, Inestrosa NC (2004) Signal transduction during amyloid-beta-peptide neurotoxicity: role in Alzheimer disease. Brain Res Brain Res Rev 47:275–289
Gao X, Tang XC (2006) Huperzine A attenuates mitochondrial dysfunction in beta-amyloid-treated PC12 cells by reducing oxygen free radicals accumulation and improving mitochondrial energy metabolism. J Neurosci Res 83:1048–1057
Giacobini E, Mori F, Lai CC (1996) The effect of cholinesterase inhibitors on the secretion of APPS from rat brain cortex. Ann NY Acad Sci 777:393–398
Haass C, Selkoe DJ (1993) Cellular processing of beta-amyloid precursor protein and the genesis of amyloid beta-peptide. Cell 75:1039–1042
Hardy J (1997) Amyloid, the presenilins and Alzheimer’s disease. Trends Neurosci 20:154–159
Hardy J, Selkoe DJ (2002) The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 297:353–356
Haring R, Fisher A, Marciano D, Pittel Z, Kloog Y, Zuckerman A, Eshhar N, Heldman E (1998) Mitogen-activated protein kinase-dependent and protein kinase C-dependent pathways link the m1 muscarinic receptor to beta-amyloid precursor protein secretion. J Neurochem 71:2094–2103
Hensley K, Carney JM, Mattson MP, Aksenova M, Harris M, Wu JF, Floyd RA, Butterfield DA (1994) A model for beta-amyloid aggregation and neurotoxicity based on free radical generation by the peptide: relevance to Alzheimer disease. Proc Natl Acad Sci USA 91:3270–3274
Inestrosa NC, Alarcon R (1998) Molecular interactions of acetylcholinesterase with senile plaques. J Physiol Paris 92:341–344
Inestrosa NC, Alvarez A, Calderon F (1996a) Acetylcholinesterase is a senile plaque component that promotes assembly of amyloid beta-peptide into Alzheimer’s filaments. Mol Psychiatry 1:359–361
Inestrosa NC, Alvarez A, Perez CA, Moreno RD, Vicente M, Linker C, Casanueva OI, Soto C, Garrido J (1996b) Acetylcholinesterase accelerates assembly of amyloid-beta-peptides into Alzheimer’s fibrils: possible role of the peripheral site of the enzyme. Neuron 16:881–891
Jiang H, Luo X, Bai D (2003) Progress in clinical, pharmacological, chemical and structural biological studies of huperzine A: a drug of traditional chinese medicine origin for the treatment of Alzheimer’s disease. Curr Med Chem 10:2231–2252
Kim HS, Lee JH, Lee JP, Kim EM, Chang KA, Park CH, Jeong SJ, Wittendorp MC, Seo JH, Choi SH, Suh YH (2002) Amyloid beta peptide induces cytochrome C release from isolated mitochondria. Neuroreport 13:1989–1993
Knudson CM, Korsmeyer SJ (1997) Bcl-2 and Bax function independently to regulate cell death. Nat Genet 16:358–363
Ko LJ, Prives C (1996) p53: puzzle and paradigm. Genes Dev 10:1054–1072
Li Y, Hu GY (2002a) Huperzine A inhibits the sustained potassium current in rat dissociated hippocampal neurons. Neurosci Lett 329:153–156
Li Y, Hu GY (2002b) Huperzine A, a nootropic agent, inhibits fast transient potassium current in rat dissociated hippocampal neurons. Neurosci Lett 324:25–28
Liang YQ, Tang XC (2004) Comparative effects of huperzine A, donepezil and rivastigmine on cortical acetylcholine level and acetylcholinesterase activity in rats. Neurosci Lett 361:56–59
Lustbader JW, Cirilli M, Lin C, Xu HW, Takuma K, Wang N, Caspersen C, Chen X, Pollak S, Chaney M, Trinchese F, Liu S, Gunn-Moore F, Lue LF, Walker DG, Kuppusamy P, Zewier ZL, Arancio O, Stern D, Yan SS, Wu H (2004) ABAD directly links Abeta to mitochondrial toxicity in Alzheimer’s disease. Science 304:448–452
Moran MA, Mufson EJ, Gomez-Ramos P (1993) Colocalization of cholinesterases with beta amyloid protein in aged and Alzheimer’s brains. Acta Neuropathol (Berl) 85:362–369
Mori F, Lai CC, Fusi F, Giacobini E (1995) Cholinesterase inhibitors increase secretion of APPs in rat brain cortex. Neuroreport 6:633–666
Nitsch RM, Slack BE, Wurtman RJ, Growdon JH (1992) Release of Alzheimer amyloid precursor derivatives stimulated by activation of muscarinic acetylcholine receptors. Science 258:304–307
Pang YP, Kozikowski AP (1994) Prediction of the binding sites of huperzine A in acetylcholinesterase by docking studies. J Comput Aided Mol Des 8:669–681
Raves ML, Harel M, Pang YP, Silman I, Kozikowski AP, Sussman JL (1997) Structure of acetylcholinesterase complexed with the nootropic alkaloid, (-)-huperzine A. Nat Struct Biol 4:57–63
Rossner S, Ueberham U, Schliebs R, Perez-Polo JR, Bigl V (1998) The regulation of amyloid precursor protein metabolism by cholinergic mechanisms and neurotrophin receptor signaling. Prog Neurobiol 56:541–569
Saille C, Marin P, Martinou JC, Nicole A, London J, Ceballos-Picot I (1999) Transgenic murine cortical neurons expressing human Bcl-2 exhibit increased resistance to amyloid beta-peptide neurotoxicity. Neuroscience 92:1455–1463
Selkoe DJ (1994) Cell biology of the amyloid beta-protein precursor and the mechanism of Alzheimer’s disease. Annu Rev Cell Biol 10:373–403
Selkoe DJ (1996) Amyloid beta-protein and the genetics of Alzheimer’s disease. J Biol Chem 271:18295–18298
Shang YZ, Ye JW, Tang XC (1999) Improving effects of huperzine A on abnormal lipid peroxidation and superoxide dismutase in aged rats. Zhongguo Yao Li Xue Bao 20:824–828
Siek GC, Katz LS, Fishman EB, Korosi TS, Marquis JK (1990) Molecular forms of acetylcholinesterase in subcortical areas of normal and Alzheimer disease brain. Biol Psychiatry 27:573–580
Sussman JL, Harel M, Frolow F, Oefner C, Goldman A, Toker L, Silman I (1991) Atomic structure of acetylcholinesterase from Torpedo californica: a prototypic acetylcholine-binding protein. Science 253:872–879
Szalai G, Krishnamurthy R, Hajnoczky G (1999) Apoptosis driven by IP(3)-linked mitochondrial calcium signals. Embo J 18:6349–6361
Tang LL, Wang R, Tang XC (2005a) Effects of huperzine A on secretion of nerve growth factor in cultured rat cortical astrocytes and neurite outgrowth in rat PC12 cells. Acta Pharmacol Sin 26:673–678
Tang LL, Wang R, Tang XC (2005b) Huperzine A protects SHSY5Y neuroblastoma cells against oxidative stress damage via nerve growth factor production. Eur J Pharmacol 519:9–15
Tang XC, Kindel GH, Kozikowski AP, Hanin I (1994) Comparison of the effects of natural and synthetic huperzine-A on rat brain cholinergic function in vitro and in vivo. J Ethnopharmacol 44:147–155
Taylor P, Radic Z (1994) The cholinesterases: from genes to proteins. Annu Rev Pharmacol Toxicol 34:281–320
Wang LM, Han YF, Tang XC (2000) Huperzine A improves cognitive deficits caused by chronic cerebral hypoperfusion in rats. Eur J Pharmacol 398:65–72
Wang R, Xiao XQ, Tang XC (2001a) Huperzine A attenuates hydrogen peroxide-induced apoptosis by regulating expression of apoptosis-related genes in rat PC12 cells. Neuroreport 12:2629–2634
Wang R, Yan H, Tang XC (2006a) Progress in studies of huperzine A, a natural cholinesterase inhibitor from Chinese herbal medicine. Acta Pharmacol Sin 27:1–26
Wang R, Zhang HY, Tang XC (2001b) Huperzine A attenuates cognitive dysfunction and neuronal degeneration caused by beta-amyloid protein-(1–40) in rat. Eur J Pharmacol 421:149–156
Wang ZF, Tang LL, Yan H, Wang YJ, Tang XC (2006b) Effects of huperzine A on memory deficits and neurotrophic factors production after transient cerebral ischemia and reperfusion in mice. Pharmacol Biochem Behav 83:603–611
Wolf BA, Wertkin AM, Jolly YC, Yasuda RP, Wolfe BB, Konrad RJ, Manning D, Ravi S, Williamson JR, Lee VM (1995) Muscarinic regulation of Alzheimer’s disease amyloid precursor protein secretion and amyloid beta-protein production in human neuronal NT2N cells. J Biol Chem 270:4916–4922
Xiao XQ, Wang R, Han YF, Tang XC (2000a) Protective effects of huperzine A on beta-amyloid(25–35) induced oxidative injury in rat pheochromocytoma cells. Neurosci Lett 286:155–158
Xiao XQ, Wang R, Tang XC (2000b) Huperzine A and tacrine attenuate beta-amyloid peptide-induced oxidative injury. J Neurosci Res 61:564–569
Xiao XQ, Yang JW, Tang XC (1999) Huperzine A protects rat pheochromocytoma cells against hydrogen peroxide-induced injury. Neurosci Lett 275:73–76
Xiao XQ, Zhang HY, Tang XC (2002) Huperzine A attenuates amyloid beta-peptide fragment 25–35-induced apoptosis in rat cortical neurons via inhibiting reactive oxygen species formation and caspase-3 activation. J Neurosci Res 67:30–36
Yan H, Zhang HY, Tang XC (2007) Involvement of M1-muscarinic acetylcholine receptors, protein kinase C and mitogen-activated protein kinase in the effect of huperzine A on secretory amyloid precursor protein-alpha. Neuroreport 18:689–692
Yu SP, Farhangrazi ZS, Ying HS, Yeh CH, Choi DW (1998) Enhancement of outward potassium current may participate in beta-amyloid peptide-induced cortical neuronal death. Neurobiol Dis 5:81–88
Zamzami N, Kroemer G (2001) The mitochondrion in apoptosis: how Pandora’s box opens. Nat Rev Mol Cell Biol 2:67–71
Zhang HY, Brimijoin S, Tang XC (2003a) Apoptosis induced by beta-amyloid25–35 in acetylcholinesterase-overexpressing neuroblastoma cells. Acta Pharmacol Sin 24:853–858
Zhang HY, Liang YQ, Tang XC, He XC, Bai DL (2002) Stereoselectivities of enantiomers of huperzine A in protection against beta-amyloid(25–35)-induced injury in PC12 and NG108–15 cells and cholinesterase inhibition in mice. Neurosci Lett 317:143–146
Zhang HY, Tang XC (2003b) Huperzine A attenuates the neurotoxic effect of staurosporine in primary rat cortical neurons. Neurosci Lett 340:91–94
Zhang HY, Tang XC (2006) Neuroprotective effects of huperzine A: new therapeutic targets for neurodegenerative disease. Trends Pharmacol Sci 27:619–625
Zhang HY, Yan H, Tang XC (2004) Huperzine A enhances the level of secretory amyloid precursor protein and protein kinase C-alpha in intracerebroventricular beta-amyloid-(1–40) infused rats and human embryonic kidney 293 Swedish mutant cells. Neurosci Lett 360:21–24
Zhao Q, Tang XC (2002) Effects of huperzine A on acetylcholinesterase isoforms in vitro: comparison with tacrine, donepezil, rivastigmine and physostigmine. Eur J Pharmacol 455:101–107
Zhou J, Tang XC (2002) Huperzine A attenuates apoptosis and mitochondria-dependent caspase-3 in rat cortical neurons. FEBS Lett 526:21–25
Acknowledgement
This work was supported by grants from the Ministry of Science and Technology of China (G199805110, G1998051115, 2004CB518907) and the National Natural Science Foundation of China (39170860, 39770846, 3001161954, 30123005, 30271494 and 30572169).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, H.Y., Yan, H. & Tang, X.C. Non-cholinergic Effects of Huperzine A: Beyond Inhibition of Acetylcholinesterase. Cell Mol Neurobiol 28, 173–183 (2008). https://doi.org/10.1007/s10571-007-9163-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-007-9163-z