Summary.
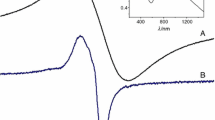
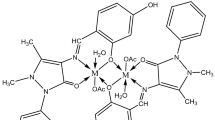
Fe(III), Co(II), Ni(II), and Cu(II) complexes of the title azodyes have been synthesized and characterized by elemental analysis, molar conductance, TGA, DTA, magnetic susceptibility measurements, IR, electronic and ESR spectral studies. The spectral studies suggest an octahedral geometry for Fe(III) and Co(II) complexes but a square planar geometry for Ni(II) and Cu(II) complexes. The kinetics of the catalysed oxidation of N,N,N′,N′-tetramethyl-p-phenylenediamine dihydrochloride (TMPPD) with mononuclear and binuclear copper complexes were studied to check the activity of these copper complexes in oxidizing organic amines. The electrochemical behaviour of the metal complexes was studied using DC polarography and cyclic voltammetry. Antimicrobial activity of the azo compounds and its complexes have been tested against different microorganisms.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
El-Baradie, K. Mononuclear and Binuclear Fe(III), Co(II), Ni(II), and Cu(II) Complexes of 3,4′-Dihydroxyazobenzene-3′,4-dicarboxylic Acid and 3-Carboxy-4-hydroxyphenylazo-3-carboxy-4-hydroxynaphthalene. Monatshefte für Chemie 136, 677–692 (2005). https://doi.org/10.1007/s00706-004-0250-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-004-0250-2