Abstract
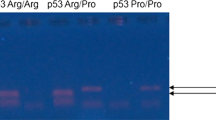
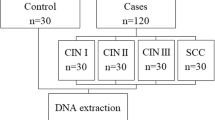
Infection by high-risk human papillomavirus (HR-HPV) and single nucleotide polymorphism (SNP) in genes involved in cell cycle control, as p21 and p27, are important factors in the development of different types of human cancers. This study aims at investigating whether both the p21 Ser31Arg and p27 V109G polymorphisms are associated with susceptibility to the development of cervical lesions in women HR-HPV positive. We analyzed 132 women HPV positive and with cervical lesions or CC and 154 healthy control (HPV negative and without cervical lesions). p21 Ser31Arg and p27 V109G polymorphisms were analyzed using the polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) method and sequencing. The p21 31Arg allele was associated with susceptibility for the development of cervical lesions (P* = 0.0009), while p27 V109G polymorphism showed no significant differences for this association (P* = 0.89). However, the combined effect of the polymorphisms showed that the presence of the CC genotype (SNP p21 Ser31Arg) conferred protection for the development of cervical lesions (OR = 0.39). p21 Ser31Arg and p27 V109G polymorphisms were not associated with the grade of cervical lesions (CINI, CINII, and CINIII) or CC (P* > 0.05). The HR-HPV more frequent in this study were of 16 (57.6 %) and 18 (37.1 %) types; however, no association was observed when both polymorphisms and risk factors analyzed were compared (P* > 0.05). Our findings suggest a possible association between p21 Ser31tabArg polymorphism and susceptibility to the development of cervical lesions in women from Pernambuco. Brazil.
Similar content being viewed by others
References
BRASIL. INCA. Instituto Nacional do Câncer. Ministério da Saúde. Cancer Prevalence Estimates 2014. Available online at: http://www.inca.gov.br/estimativa/2014/sintese-de-resultadocomentarios.asp (accessed May 13, 2014).
Snijders PJ, Steenbergen RD, Heideman DA, Meijer CJ. HPV-mediated cervical carcinogenesis: concepts and clinical implications. J Pathol. 2006;208:152–64.
Insinga RP, Dasbach EJ, Elbasha EH. Epidemiologic natural history and clinical management of Human Papillomavirus (HPV) Disease: a critical and systematic review of the literature in the development of an HPV dynamic transmission model. BMC Infect Dis. 2009;9:119.
Pei J, Jianxin L, Wen L, Xiaoxi Z, Jianxin T. Role of p53 and p21 polymorphisms in the risk of cervical cancer among Chinese women. Acta Biochim Biophys Sin. 2010;42:671–6.
Tavares MCM, Macêdo JL, Júnior SFL, Heráclio AS, Amorim MMR, Maia MMD, et al. Chlamydia trachomatis infection and human papillomavirus in women with cervical neoplasia in Pernambuco-Brazil. Mol Biol Rep. 2014;41:865–74.
Cotran RS, Kumar V, Collins T. Robbins: patologia estrutural e funcional. 6.ed. Rio de Janeiro: Guanabara Koogan. 2000
Kumar V, Abbas A, Fausto N, Mitchell RN. Robbins: patologia básica. 8.ed. Rio de Janeiro: Guanabara Koogan. (2010)
Singh S, Johnson J, Chellappan S. Small molecule regulators of Rb-E2F pathway as modulators of transcription. Biochim Biophys Acta. 2010;1799(10–12):788–94.
Eleutério Junior J, Giraldo PC, Gonçalves AK. Marcadores imunoistoquímicos de lesões precursoras do câncer de colo uterino associadas ao HPV: o papel da proteína de supressão tumoral p16ink4a. DST J Bras Doenças Sex Transm. 2006;18(1):62–5.
Knebel MD. p16INK4a as a biomarker for differentiating replicating and transforming high risk HPV infections: the theoretical concept and its potential diagnostic impact HPV today, Newsletter on Human Papillomavirus. 2009; 7–8, M– 35437–2001.
Itamochi H, Yoshida T, Walker CL, Bartholomeusz C, Aoki D, Ishihara H, et al. Novel mechanism of reduced proliferation in ovarian clear cell carcinoma cells: cytoplasmic sequestration of CDK2 by p27. Gynecol Oncol. 2011;122:641.
Bahnassy AA, Zekri AR, Saleh M, Lotayef M, Moneir M, Shawki O. The possible role of cell cycle regulators in multistep process of HPV-associated cervical carcinoma. BMC Clin Pathol. 2007;7:94–9.
Koushik A, Platt RW, Franco EL. p53 Codon 72 Polymorphism and cervical neoplasia: a meta-analysis review. Cancer Epidemiol Biomarkers Prev. 2004;13:11–22.
Harima Y, Sawada S, Nagata K, Sougawa M, Ostapenko V, Ohnishi T. Polymorphism of the WAF1 gene is related to susceptibility to cervical cancer in Japanese women. Int J Mol Med. 2001;7:261–4.
Xi YG, Ding KY, Su XL, Chen DF, You WC, Shen Y, et al. p53 polymorphism and p21WAF1/CIP1 haplotype in the intestinal gastric cancer and the precancerous lesions. Carcinogenesis. 2004;25:2201–6.
Choi YY, Kang HK, Choi JE, Jang JS, Kim EJ, Cha SI, et al. Comprehensive assessment of p21 polymorphisms and lung cancer risk. J Hum Genet. 2008;53:87–95.
Lei D, Sturgis EM, Liu Z, Zafereo ME, Wei Q, Li G. Genetic polymorphisms of p21 and risk of second primary malignancy in patients with index squamous cell carcinoma of the head and neck. Carcinogenesis. 2010;31:222–7.
Van den Broeke C, Radu M, Chernoff J, Favoreel HW. An emerg- ing role for p21-activated kinases (paks) in viral infections. Trends Cell Biol. 2010;20:160–9.
Jiang P, Liu J, Li W, Zeng X, Tang J. Role of p53 and p21 polymorphisms in the risk of cervical cancer among Chinese women. Acta Biochim Biophys Sin. 2010;42(9):671–6.
Pasquali D, Circelli L, Faggiano A, Pancione M, Renzullo A, Elisei R, et al. CDKN1B V109G polymorphism a new prognostic factor in sporadic medullary thyroid carcinoma. Eur J Endocrinol. 2011;164(3):397–404.
Lloyd RV, Erickson LA, Jin L, Kulig E, Qian X, Cheville JC, et al. p27kip1: a multifunctional cyclin-dependent kinase inhibitor with prognostic significance in human cancers. Am J Pathol. 1999;154(2):313–23.
Huang LW, Chao SL, Hwang JL, Chou YY. Downregulation of p27 is associated with malignant transformation and aggressive phenotype of cervical neoplasms. Gynecol Oncol. 2002;85:524–8.
Kibel AS, Suarez BK, Belani J, Oh J, Webster R, Brophy-Ebbers M, et al. CDKN1A and CDKN1B polymorphisms and risk of advanced prostate carcinoma. Cancer Res. 2003;63:2033.
Li G, Sturgis EM, Wang LE, Chamberlain RM, Spitz MR, El-Naggar AK, et al. Association between the V109G polymorphism of the p27 gene and the risk and progression of oral squamous cell carcinoma. Clin Cancer Res. 2004;10:3996.
Camargo-Kosugi CM, da Silva ID, Sato H, D’Amora P, Carvalho CV, Nogueira-de-Souza NC, et al. Eur J Obstetrics Gynecology Reproductive Biol. 2009;145:180–3.
Tae KY, Kyoung CE, Hoon CN, Hung KJ, Ick Yang W, Wook KJ, et al. Expression of cyclin E and p27KIP1 in cervical carcinoma. Cancer Lett. 2000;153(1–2):41–50.
Goff BA, Sallin J, Garcia R, VanBlaricom A, Paley PJ, Muntz HG. Evaluation of p27 in preinvasive and invasive malignancies of the cervix. Gynecol Oncol. 2003;88:40–4.
Sgambato A, Zannoni GF, Faraglia B, Camerini A, Tarquini E, Spada D, et al. Decreased expression of the CDK inhibitor p27KIP1 and increased oxidative DNA damage in the multistep process of cervical carcino- genesis. Gynecol Oncol. 2004;92(3):776–83.
Solomon MR, Bamossy GJ, Askegaard S. Consumer behaviour: European perspective. London: Pearson Education Limited; 2002.
Associacão Brasileira de Genitoscopia. Nomenclatura para laudos Colposcópico. 2002; Available online at: http://www.colposcopy.org.br (accessed august 2015).
De Roda Husman AM, Walboomers JMM, van den Brule AJC, Meijer CJLM, Snijders PJF. The use of general primers GP5 and GP6 elongated at their 30 ends with adjacent highly conserved sequences improves human papillomavirus detection by PCR. J Gen Vir. 1995;76:1057–62.
Manos MM, Ting Y, Wright DK, Lewis AJ, Broker TR, Wolinsky SM. The use of polymerase chain reaction amplification for the detection of genital human papillomaviruses. Cancer Cells. 1989;7:209–14.
Karlsen F, Kalantari M, Jenkins A, Pettersen E, Kristensen G, Holm R, et al. Use of multiple PCR primer sets for optimal detection of human papillomavirus. J Clin Microbiol. 1996;34:2095–100.
Li YJ, Laurent-Puig P, Salmon RJ, Thomas G, Hamelin R. Polymorphisms and probable lack of mutation in the WAF1-CIP1 gene in colorectal cancer. Oncogene. 1995;10:599–601.
Chedid M, Michieli P, Lengel C, Huppi K, Givol D. A single nucleotide substitution at codon 31 (Ser/Arg) defines a polymorphism in a highly conserved region of the p53-inducible gene WAF1/CIP1. Oncogene. 1994;9(10):3021–4.
Wang Z, Sturgis EM, Zhang F, Lei D, Liu Z, Xu L, et al. Genetic variants of p27 and p21 as predictors for risk of second primary malignancy in patients with index squamous cell carcinoma of head and neck. Mol Cancer. 2012;11:17.
Bhattacharya P, Sengupta S. Lack of evidence that proline homozygos- ity at codon 72 of p53 and rare arginine allele at codon 31 of p21, jointly mediate cervical cancer susceptibility among Indian women. Gynecol Oncol. 2005;99:176–82.
Ning W, Shizhuo W, Qiao Z, Yanming L, Heng W, Wei L, et al. Association of p21 SNPs and risk of cervical cancer among Chinese women. BMC Cancer. 2012;12:589.
Roh J, Kim M, Kim J, Park N, Song Y, Kang S, et al. Polymorphisms in codon 31 of p21 and cervical cancer susceptibility in Korean women. Cancer Lett. 2001;165:59–62.
Tian Q, Lu W, Chen H, Ye F, Xie X. The nonsynonymous single-nucleotide polymorphisms in codon 31 of p21 gene and the susceptibility to cervical cancer in Chinese women. Int J Gynecol Cancer. 2009;19:1011–4.
Birgander R, Sjalander A, Saha N, Spitsyn V, Beckman L, Beckman G. The codon 31 polymorphism of the p53-inducible gene p21 shows dis- tinct differences between major ethnic groups. Hum Hered. 1996;46:148–54.
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100:57–70.
Stah M, Dijkers PF, Kops GJ, Lens SM, Coffer PJ, Burgering BM, et al. The forkhead transcription factor FoxO regulates transcription of p27Kip1 and Bim in response to IL-2. JImmuno. 2002;168:5024–31.
Oliveira-Silva M, Lordello CX, Zardo LM, Bonvicino CR, Moreira MA. Human papillomavirus in Brazilian women with and without cervical lesions. 2011; Virol J 8:4.
da Silva MF B, Chagas BS, Guimarães V, Katz LM, Felix PM, Miranda PM, et al. HPV31 and HPV33 incidence in cervical samples from women in Recife, Brazil. Genet Mol Res. 2009;8:1437–43.
Lorenzato F, Ho L, Terry G, Singer A, Santos LC, De Lucena Batista R, et al. The use of human papillomavirus typing in detection of cervical neoplasia in Recife (Brazil). Int J Gynecol Cancer. 2000;10:143–50.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
The work was performed at the Rural Federal University of Pernambuco (UFRPE), Recife, PE, Brazil.
Rights and permissions
About this article
Cite this article
Lima, G., Santos, E., Angelo, H. et al. Association between p21 Ser31Arg polymorphism and the development of cervical lesion in women infected with high risk HPV. Tumor Biol. 37, 10935–10941 (2016). https://doi.org/10.1007/s13277-016-4979-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-4979-0