Abstract
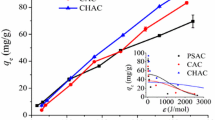
In this study, adsorption of catalase enzyme onto activated carbon obtained from apple shell, kinetic parameters, and activation data of adsorption process was investigated. Temperature (15, 25, 36.5, and 45 °C), solution pH (5.5, 7, and 9), initial catalase enzyme concentration (1.10−1, 2.10−1, and 2.5.10−1 g l−1), and ionic strength (1.10−2, 5.10−2, and 7.5.10−2 M) have taken as kinetic parameters for the adsorption of catalase enzyme on activated carbon. In all runs, common parameters of temperature, pH, initial enzyme concentration, and ionic strength were taken as 298 K, pH 7, 0.2 g l−1, and 5.10−2 M, respectively. The results of experiments revealed that the adsorption of catalase enzyme onto activated carbon increased with increasing temperature (15–45 °C), ionic strength, and initial catalase enzyme concentration. However, the adsorption process of catalase on activated carbon was affected negatively by increased pH. The thermodynamic functions such as enthalpy (ΔH), activation energy (Ea), entropy (ΔS), and Gibbs energy (ΔG) were investigated. ΔG, Ea, ΔH, and ΔS were found to be − 70.61, 4.19, − 1.69 kJ mol−1, and 231 J mol−1 K−1 for catalase enzyme adsorption, respectively. The adsorption of the process was investigated using Eyring and Arrhenius equations, and the findings showed that the adsorption kinetic is coherent with the pseudo-second-order model. Eventually, activated carbon can be used as an effective adsorbent for the adsorption of catalase.







Similar content being viewed by others
References
Sen, F., Karataş, Y., Gülcan, M., & Zahmakıran, M. (2014). Amylamine stabilized platinum (0) nanoparticles: active and reusable nanocatalyst in the room temperature dehydrogenation of dimethylamine-borane. RSC Advances, 4(4), 1526–1531.
Akocak, S., Şen, B., Lolak, N., Şavk, A., Koca, M., Kuzu, S., & Şen, F. (2017). One-pot three-component synthesis of 2-amino-4H-chromene derivatives by using monodisperse Pd nanomaterials anchored graphene oxide as a highly efficient and recyclable catalyst. Nano-Structures & Nano-Objects, 11, 25–31.
Pamuk, H., Aday, B., Kaya, M., & Sen, F. (2015). Pt Nps@GO as highly efficient and reusable catalyst for one-pot synthesis of acridinedione derivatives. RSC Advances, 5, 49295–49300.
Sen, B., Kuzu, S., Demir, E., Okyay, T. O., & Sen, F. (2017). Hydrogen liberation from the dehydrocoupling of dimethylamine-borane at room temperature by using novel and highly monodispersed RuPtNi nanocatalysts decorated with graphene oxide. International Journal of Hydrogen Energy, 42(36), 23299–23306.
Demir, E., Savk, A., Sen, B., & Sen, F. (2017). A novel monodisperse metal nanoparticles anchored graphene oxide as counter electrode for dye-sensitized solar cells. Nano-Structures & Nano-Objects, 12, 41–45.
Sahin, B., Aygun, A., Gunduz, H., Sahin, K., Demir, E., Akocak, S., & Sen, F. (2018). Cytotoxic effects of platinum nanoparticles obtained from pomegranate extract by the green synthesis method on the MCF-7 cell line. Colloids and Surfaces B: Biointerfaces, 163, 119–124.
Demirci, T., Celik, B., Yıldız, Y., Eriş, S., Arslan, M., Kilbas, B., & Sen, F. (2016). One-pot synthesis of Hantzsch dihydropyridines using highly efficient and stable PdRuNi@GO catalyst. RSC Advances, 6, 76948–76956.
Celik, B., Baskaya, G., Karatepe, O., Erken, E., & Sen, F. (2016). Monodisperse Pt(0)/DPA@GO nanoparticles as highly active catalysts for alcohol oxidation and dehydrogenation of DMAB. International Journal of Hydrogen Energy, 41, 5661–5669.
Abrahamson, J. T., Sen, F., Sempere, B., & Wal, M. P. (2013). Excess thermopower and the theory of thermopower waves. ACS Nano, 7(8), 6533–6544.
Bozkurt, S., Tosun, B., Sen, B., Akocak, S., Savk, A., Ebeoğlugil, M. F., & Sen, F. (2017). A hydrogen peroxide sensor based on TNM functionalized reduced graphene oxide grafted with highly monodisperse Pd nanoparticles. Analytica Chimica Acta, 989C, 88–94.
Ayranci, R., Baskaya, G., Guzel, M., Bozkurt, S., Ak, M., Savk, A., & Sen, F. (2017). Carbon-based nanomaterials for high-performance optoelectrochemical systems. Chemistry Select., 2(4), 1548–1555.
Dasdelen, Z., Yıldız, Y., Eris, S., & Sen, F. (2017). Enhanced electrocatalytic activity and durability of Pt nanoparticles decorated on GO-PVP hybride material for methanol oxidation reaction. Applied Catalysis B: Environmental, 219C, 511–516.
Karatepe, O., Yildiz, Y., Pamuk, H., Eris, S., Dasdelen, Z., & Sen, F. (2016). Enhanced electrocatalytic activity and durability of highly monodisperse Pt@PPy-PANI nanocomposites as a novel catalyst for electro-oxidation of methanol. RSC Advances, 6, 50851–50857.
Baskaya, G., Esirden, I., Erken, E., Kaya, M., & Sen, F. (2017). Synthesis of 5-substituted-1H-tetrazole derivatives using monodisperse carbon black decorated Pt nanoparticles as heterogeneous nanocatalysts. Journal of Nanoscience and Nanotechnology, 17, 1992–1999.
Erkan, A., Bakir, U., & Karakas, G. (2006). Photocatalytic microbial inactivation over Pd doped SnO2 and TiO2 thin films. Journal of Photochemistry and Photobiology A: Chemistry, 184, 313–321.
Preety, V. H. (2014). Immobilization and kinetics of catalase on calcium carbonate nanoparticles attached epoxy support. Applied Biochemistry and Biotechnology, 172, 115–130.
Gianfreda, L., & Scarfi, M. R. (1991). Enzyme stabilization: state of the art. Molecular and Cellular Biochemistry, 100(2), 97–128.
Dogac, Y. I., Cinar, M., & Teke, M. (2015). Improving catalase stability properties by encapsulation in alginate=Fe3O4 magnetic composite beads for enzymatic removal of H2O2. Preparative Biochemistry & Biotechnology, 45, 144–157.
Gao, X., Yang, W., Pang, P., LiaoCai, S. Q., Zeng, K., & Grimes, C. A. (2007). A wireless magneto elastic biosensor for rapid detection of glucose concentrations in urine samples. Sensors and Actuators B: Chemical, 128, 161–167.
Amorim, A. M., Gasques, M. D. G., Jurgen, A. J., & Scharf, M. (2002). The application of catalase for the elimination of hydrogen peroxide residues after bleaching of cotton fabrics. Anais da Academia Brasileira de Ciências, 74, 433–436.
Tatsuma, T., Watanabe, T., Tatsuma, S., & Watanabe, T. (1994). Substrate-purging enzyme electrodes, peroxidase/catalase electrodes for hydrogen peroxide with an improved upper sensing limit. Analytical Chemistry, 66(2), 290–294.
Madaras, M. B., Spokane, R. B., Johnson, J. M., & Woodward, J. R. (1997). Glutamine biosensors for biotechnology applications, with suppression of the endogenous glutamate signal. Analytical Chemistry, 69(18), 3674–3678.
Santoni, T., Santianni, D., Manzoni, A., Zanardi, S., & Mascini, M. (1997). Enzyme electrode for glucose determination in whole blood. Talanta, 44(9), 1573–1580.
Hnaien, M., Lagarde, F., & Jaffrezic-Renault, N. (2010). A rapid and sensitive alcohol oxidase/catalase conductometric biosensor for alcohol determination. Talanta, 81(1–2), 222–227.
Felhofer, J. L., Caranto, J. D., & Garcia, C. D. (2010). Adsorption kinetics of catalase to thin films of carbon nanotubes. Langmuir, 26(22), 17178–17183.
Alptekin, O., Tukel, S. S., Yıldırım, D., & Alagöz, D. (2010). Immobilization of catalase onto Eupergit C and its characterization. Journal of Molecular Catalysis B: Enzymatic, 64(3–4), 177–183.
Yang, J. J., Ma, X. O., Zhang, Z. S., Cheng, B., Li, S., & Wang, G. (2010). Lipase immobilized by modificationcoupled and adsorption-cross-linking methods: a comparative study. Biotechnology Advances, 28, 644–650.
Reguly, J. C. (2000). Biotechnology of fermentative process (Portuguese) (Vol. 3). Pelotas, Brasil: Editora Universitário/UFPel ISBN: 85-7192-128-8.
Burns, R. G. (1986). Interaction of enzymes with soil mineral and organic colloids. Madison: Soil Science Society of America.
Kise, H., & Hayakawa, A. (1991). Immobilization of proteases to porous chitosan beads and their catalysis for ester and peptide synthesis in organic solvents. Enzyme and Microbial Technology, 13, 584–588.
Huang, X. L. (1997). Comparıson of the propertıes of trypsın immobilized on 2 celite(TM) derıvatıves, Catignani H.L, and Swaisgood H.E. Journal of Biotechnology, 53, 21.
Gooding, J. J., & Hal, E. A. H. (1996). Membrane properties of acrylate bulk polymers for. Biosensor applications. Biosensors & Bioelectronics, 11, 1031.
Yıldız, A., & GÜR, A. (2007). Adsorption of phenol and chlorophenols on pure and modified sepiolite. Journal of the Serbian Chemical Society, 72, 467.
Yıldız, A., GÜR, A., & Ceylan, H. (2006). Adsorption of aniline, phenol, and chlorophenols on pure and modified bentonite. The Russıan Journal of Physıcal Chemıstry, 80, S172–S176.
Yıldız, A., & GÜR, A. (2006). Adsorption of phenol and phenol derivatives on pure and modified kaolinite. Asian Journal of Chemistry, 18, 2650.
Baileey, J. E., & Ollis, D. F. (1986). Applied enzyme catalysis (p. 180). Singapore: Mc Graw-Hill International.
Kennedy, J. F., & Melo, E. H. M. (1990). Immobilized enzymes and cells. Chemical Engineering Progress, 86, 81.
Bjorlıng, F., Godtfredsen, S. E., & Kırk, O. (1991). The future impact of industrial lipases. Trends in Biotechnology, 9, 360–363.
Demirbas, Ö., Calimli, M. H., Kuyuldar, E., Alma, M. H., Nas, M. S., & Sen, F. Equilibrium, kinetics, and thermodynamic of adsorption of enzymes on diatomite clay materials. BioNanoscience. https://doi.org/10.1007/s12668-019-00615-1.
Çalımlı, M. H., Demirbaş, Ö., Aygün, A., Alma, M. H., Nas, M. S., & Şen, F. (2018). Immobilization kinetics and mechanism of bovine serum albumin on diatomite clay from aqueous solutions. Applied Water Science, 8, 209.
Akkuş, P., (2006). “Lipaz Kullanılarak Şeker Esteri Sentezi”, G.Y.T.E. Mühendislik ve Fen Bilimleri Enstitüsü, Yüksek Lisans Tezi, Gebze.
Hunter, J. (1999). Introduction to modern colloid science. New York, USA: Oxford University Press.
Doğan, M., Alkan, M., Demirbaş, O., Özdemir, Y., & Özmetin, C. (2006). Adsorption kinetics of Maxilon blue GRL onto sepiolite from aqueous solutions. Chemical Engineering Journal, 124, 89–101.
Demirbaş, O., (2006). Doctorate Thesis, Balikesir University Institute of Science, Balıkesir.
Tekin, N., Demirbaş, O., & Alkan, M. (2005). Adsorption of cationic polyacrylamide onto kaolinite. Microporous and Mesoporous Materials, 85, 340–350.
Vermöhlen, K., Lewandowski, H.-D., Narres, H. D., & Schwuger, M. J. (2000). Adsorption of polyelectrolytes onto oxides—the influence of ionic strength, molar mass, and Ca2+ ions. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 163, 45–53.
Vecchia, R. D., Sebrao, D., Nascımento, M. G., & Soldı, V. (2005). Carboxymethylcellulose and poly(vinyl alcohol) used as film support for lipases immobilization. Process Biochemistry, 40, 2677–2682.
Pronk, W., Kerkhof, P. J. A. M., Van Helden, C., & van Rıet, K. (1988). The hydrolysis of triglycerides by immobilized lipase in a hydrophilic membrane reactor. Biotechnology and Bioengineering, 32, 512–518.
Xu, H., Li, M., & He, B. (1995). Immobilization of Candida cylindracea lipase on methyl acrylate-divinyl benzene copolymer and its derivatives. Enzyme and Microbial Technology, 17, 194–199.
Montero, S., Blanco, A., Virto, M., Ladenta, L. C., Agud, I., Solozabal, R., Lascaray, J. M., Renobales, M., Llama, M. J., & Serra, J. L. (1993). Immobilization of Candida rugosa lipase and some properties of the immobilized enzyme. Enzyme and Microbial Technology, 15, 239–247.
Guo, Y., & Bustin, R. M. (1998). FTIR spectroscopy and reflectance of modern charcoals and decayed fungal woods: Implications for studies of intertinite in coals. International Journal of Coal Geology, 37, 29–53.
Figueiredo, J. L., Pereira, M. F. R., Freitas, M. M. A., & Crfao, J. J. M. (1999). Modification of surface chemistry of activated carbons. Carbon, 37, 1379–1389.
Lu, C. F., Nadarajah, A., & Chittur, K. K. (1994). A comprehensive model for protein adsorption to surfaces. Journal of Colloid and Interface Science, 168, 152–161.
Giacomelli, C. E., Bremer, M. G., & Norde, W. J. (1999). ATR-FTIR study of IgG adsorbed on different silica surfaces. Journal of Colloid and Interface Science, 220, 13–23.
Ai, Q., Yang, D., Li, Y., Shi, J., Wang, X., & Jiang, Z. (2014). Highly efficient covalent immobilization of catalase on titanate nanotubes. Biochemical Engineering Journal, 83, 8–15.
Rahimi-Gorji, M., Gorji, T. B., & Gorji-Bandpy, M. (2016). Details of regional particle deposition and airflow structures in a realistic model of human tracheobronchial airways: two-phase flow simulation. Computers in Biology and Medicine, 74, 1–17.
Rahimi-Gorji, M., Pourmehran, O., Gorji-Bandpy, M., & Gorji, T. B. (2015). CFD simulation of airflow behavior and particle transport and deposition in different breathing conditions through the realistic model of human airways. Journal of Molecular Liquids, 209, 121–133.
Hussanan, A., Khan, I., Gorji, M. R., & Khan, W. A. (2019). CNTS-water-based nanofluid over a stretching sheet. BioNanoScience. https://doi.org/10.1007/s12668-018-0592-6.
Sureshkumar Raju, S., Ganesh Kumar, K., Rahimi-Gorji, M., & Khan, I. Darcy–Forchheimer flow and heat transfer augmentation of a viscoelastic fluid over an incessant moving needle in the presence of viscous dissipation. Microsystem Technologies. https://doi.org/10.1007/s00542-019-04340-3.
Ahmed, N., Shah, N. A., Ahmad, B., Shah, S. I. A., Ulhaq, S., & Rahimi-Gorji, M. Transient MHD convective flow of fractional nanofluid between vertical plates. Journal of Applied and Computational Mechanics. https://doi.org/10.22055/JACM.2018.26947.1364.
Ho, Y. S., & McKay, G. (1999). Pseudo-second order model for sorption processes. Process Biochemistry, 34, 451–465.
Doğan, M., & Alkan, M. (2003). Adsorption kinetics of methyl violet onto perlite. Chemosphere, 50, 517–528.
Mall, I. D., & Upadhyay, S. N. (1995). Treatment of methyl violet bearing wastewater from paper mill effluent using low-cost adsorbents. Journal of Indian Pulp and Paper Technical Association, 7(1), 51–57.
Kannan, N., & Sundaram, M. (2001). Kinetics and mechanism of removal of methylene blue by adsorption on various carbons—a comparative study. Dyes and Pigments, 51, 5–40.
Laidler, K. J., & Meiser, J. M. (1999). Physical chemistry (p. 852). New York, NY: Houghton Mifflin.
Singh, D. (2000). Studies of the adsorption thermodynamics of oxamyl on fly ash. Adsorption Science and Technology, 18(8), 741–748.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed Consent
None.
Funding Statement
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Demirbaş, Ö., Çalımlı, M.H., Demirkan, B. et al. The Kinetic Parameters of Adsorption of Enzymes Using Carbon-Based Materials Obtained from Different Food Wastes. BioNanoSci. 9, 749–757 (2019). https://doi.org/10.1007/s12668-019-00635-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12668-019-00635-x