Abstract
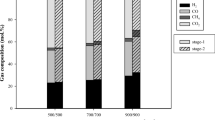
The fixed-gas drag force from a model calculation method that stabilizes the agitation capabilities of different gas ratios was used to explore the influence of temperature and hydrogen concentration on fluidizing duration, metallization ratio, utilization rate of reduction gas, and sticking behavior. Different hydrogen concentrations from 5vol% to 100vol% at 1073 and 1273 K were used while the drag force with the flow of N2 and H2 (N2: 2 L·min−1; H2: 2 L·min−1) at 1073 K was chosen as the standard drag force. The metallization ratio, mean reduction rate, and utilization rate of reduction gas were observed to generally increase with increasing hydrogen concentration. Faster reduction rates and higher metallization ratios were obtained when the reduction temperature decreased from 1273 to 1073 K. A numerical relation among particle diameter, particle drag force, and fluidization state was plotted in a diagram by this model.
Similar content being viewed by others
Abbreviations
- C d :
-
Drag coefficient
- d p :
-
Particle diameter, m
- d pmax :
-
The maximum fluidized particle size, m
- F d :
-
Particle drag force, N
- F dn :
-
Standard particle drag force, N
- M R :
-
Metallization ratio
- M Fe :
-
Metallic iron content
- M RR :
-
Mean reduction rate, s−1
- M R(t) :
-
Metallization ratio at time t
- m :
-
The feeding quantity of Fe2O3, g
- M i , M j :
-
Molar mass of gas components i and j, respectively, kg·mol−1
- R :
-
Fluidized bed inside the diameter, m
- Re :
-
Reynolds number
- T Fe :
-
Total iron content
- t :
-
Reduction time, s
- U g :
-
Gas velocity, m·s−1
- U mf :
-
The minimum fluidization velocity, m·s−1
- V mol :
-
Molar volume, m3·mol−1
- \(V_{H_2 } ,V_{N_2 }\) :
-
The gas flow rate of H2 and N2, respectively, m−3·s−1
- α :
-
Constant
- \(\eta _{H_2 }\) :
-
Hydrogen average utilization rate
- φ ij :
-
Coefficient, the ratio of viscosity to molecular mass of gas components i and j
- ρ g :
-
Gas density, kg·m−3
- ρ p :
-
True density of fluidized particles, kg·m−3
- µg :
-
Gas dynamic viscosity, Pa·s
- µ i , µ j :
-
Dynamic viscosity of gas components i and j, respectively, Pa·s
References
A. Hasanbeigi, M. Arens, and L. Price, Alternative emerging ironmaking technologies for energy-efficiency and carbon dioxide emissions reduction: a technical review, Renewable Sustainable Energy Rev., 33(2014), p. 645.
T. Harada and H. Tanaka, Future steelmaking model by direct reduction technologies, ISIJ Int., 51(2011), No. 8, p. 1301.
J.H. Shao, Z.C. Guo, and H.Q. Tang, Effect of coating MgO on sticking behavior during reduction of iron ore concentrate fines in fluidized bed, Steel Res. Int., 84(2013), p. 111.
J.H. Shao, Z.C. Guo, and H.Q. Tang, Influence of reducing atmosphere on the sticking during reduction of iron ore fines in a fluidized bed, J. Univ. Sci. Technol. Beijing, 35(2013), No. 3, p. 273.
S. Hayashi and Y. Iguchi, Factors affecting the sticking of fine iron ores during fluidized bed reduction, ISIJ Int., 32(1992), No. 9, p. 962.
R. Turton and O. Levenspiel, A short note on the drag correlation for spheres, Powder Technol., 47(1986), No. 1, p. 83.
Y.W. Zhong, Z. Wang, Z.C. Guo, and Q. Tang, Prediction of defluidization behavior of iron powder in a fluidized bed at elevated temperatures: theoretical model and experimental verification, Powder Technol., 249(2013), p. 175.
Y. Hu, R.D. Lü, and G.J. Liu, Inoganic Chemistry, Higher Education Press, Beijing, 2006, p. 12.
Y.C. Li, Introduction of Fluidized Process Engineering, Science Press, Beijing, 2008, p. 33.
W.M. Rohsenow, J.R. Hartnett, and Y.I. Cho, Handbook of Heat Transfer, McGraw-Hill Professional, New York, 1998, p. 231.
R.S. Xie, Chemical Process Nomography: Physical Property Data Calculation, Chemistry Industry Press, Beijing, 1983, p. 200.
S.P. Matthew, T.R. Cho, and P.C. Hayes, Mechanisms of porous iron growth on wustite and magnetite during gaseous reduction, Metall. Trans. B, 21(1990), No. 4, p. 733.
S.P. Matthew and P.C. Hayes, Microstructural changes occurring during the gaseous reduction of magnetite, Metall. Trans. B, 21(1990), No. 1, p. 153.
Y.W. Zhong, X.Z. Gong, Z. Wang, and Z.C. Guo, Effect of iron surfaces with nano/micro structures on the sticking of Fe2O3 in fluidized bed reduction, J. Univ. Sci. Technol. Beijing, 33(2011), No. 4, p. 406.
B. Zhang, X.Z. Gong, Z. Wang, and Z.C. Guo, Relation between sticking and metallic iron precipitation on the surface of Fe2O3 particles reduced by CO in the fluidized bed, ISIJ Int., 51(2011), No. 9, p. 1403.
R. Nicolle and A. Rist, The mechanism of whisker growth in the reduction of wüstite, Metall. Mater. Trans. B, 10(1979), No. 3, p. 429.
M. Chang, C. Lutgard, and J.H. De, Whisker growth in reduction of oxides, Metall. Mater. Trans. B, 15(1984), No. 4, p. 685.
S. El Moujahid and A. Rist, The nucleation of iron on dense wustite: a morphological study, Metall. Mater. Trans. B, 19(1988), No. 5, p. 787.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, L., Gao, H., Yu, Jt. et al. Influence of hydrogen concentration on Fe2O3 particle reduction in fluidized beds under constant drag force. Int J Miner Metall Mater 22, 12–20 (2015). https://doi.org/10.1007/s12613-015-1037-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-015-1037-7