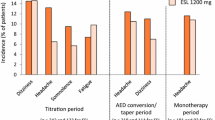
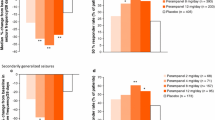
Abstract
Although the majority of people with epilepsy have a good prognosis and their seizures can be well controlled with pharmacotherapy, up to one-third of patients can develop drug-resistant epilepsy, especially those patients with partial seizures. This unmet need has driven considerable efforts over the last few decades aimed at developing and testing newer antiepileptic agents to improve seizure control. One of the most promising antiepileptic drugs of the new generation is zonisamide, a benzisoxazole derivative chemically unrelated to other anticonvulsant agents. In this article, the authors present the results of a systematic literature review summarizing the current evidence on the efficacy and tolerability of zonisamide for the treatment of partial seizures. Of particular interest within this updated review are the recent data on the use of zonisamide as monotherapy, as they might open new therapeutic avenues.

Similar content being viewed by others
References
World Health Organization. The world health report 2001—mental health: new understanding, new hope. World Health Organization; 2001.
Fisher RS, van Emde Boas W, Blume W, et al. Epileptic seizures and epilepsy: definitions proposed by the International League Against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE). Epilepsia. 2005;46:470–2.
Shorvon SD. The etiologic classification of epilepsy. Epilepsia. 2011;52:1052–7.
Berg AT, Bercovic SF, Brodie MJ, et al. Revised terminology and concepts for organization of seizures and epilepsies: report of the ILAE Commission on Classification and Terminology, 2005–2009. Epilepsia. 2010;51:676–85.
Leonardi M, Ustun TB. The global burden of epilepsy. Epilepsia. 2002;43:21–5.
Mitchell JW, Seri S, Cavanna AE. Pharmacotherapeutic and non-pharmacological options for refractory and difficult-to-treat seizures. J Cent Nerv Syst Dis. 2012;19:105–15.
Nunes VD, Sawyer L, Sarri G, Cross H. Diagnosis and management of the epilepsies in adults and children: summary of updated NICE guidance. BMJ. 2012;344:e281.
Brodie MJ, Barry SJ, Bamagous GA, Norrie JD, Kwan P. Patterns of treatment response in newly diagnosed epilepsy. Neurology. 2012;78:1548–54.
Kwan P, Arzimanoglou A, Berg AT, et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia. 2010;51:1069–77.
Leppik IE. Three new drugs for epilepsy: levetiracetam, oxcarbazepine and zonisamide. J Child Neurol. 2002;17:53–7.
Schulze-Bonhage A. Zonisamide in the treatment of epilepsy. Expert Opin Pharmacother. 2010;11:115–26.
Kito M, Maehara M, Watanabe K. Mechanisms of T-type calcium channel blockade by zonisamide. Seizure. 1996;5:115–9.
Mula M. Profile of once-daily zonisamide as monotherapy for treatment of partial seizures in adults. Drug Des Dev Ther. 2013;7:397–402.
Masuda Y, Ishizaki M, Shimizu M. Zonisamide: pharmacology and clinical efficacy in epilepsy. CNS Drug Rev. 1998;4:341–60.
Mula M, Cavanna AE, Monaco F. Psychopharmacology of topiramate: from epilepsy to bipolar disorder. Neuropsychiatr Dis Treat. 2006;2:475–88.
Rosler TW, Arias-Carrion O, Hoglinger GU. Zonisamide: aspects in neuroprotection. Exp Neurol. 2010;224:336–9.
Kochak GM, Page JG, Buchanan RA, Peters R, Padgett CS. Steady-state pharmacokinetics of zonisamide, an antiepileptic agent for treatment of refractory complex partial seizures. J Clin Pharamacol. 1998;38:166–71.
Eisai Pharmaceuticals Inc. Zonegran® (zonisamide) capsules package insert. Eisai Pharmaceuticals Inc; 2006.
Wilfong AA, Willmore LJ. Zonisamide—a review of experience and use in partial seizures. Neuropsychiatr Dis Treat. 2006;2:269–80.
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. J Clin Epidemiol. 2009;62:1–34.
Schmidt D, Jacob R, Loiseau P, et al. Zonisamide for add-on treatment of refractory partial epilepsy: a European double-blind trial. Epilepsy Res. 1993;15:67–73.
Faught E, Ayala R, Montouris GG, Leppik IE. Randomized controlled trial of zonisamide for the treatment of refractory partial-onset seizures. Neurology. 2001;57:1774–9.
Sackellares JC, Ramsay RE, Wilder BJ, Browne TR, Shellenberger MK. Randomized, controlled clinical trial of zonisamide adjunctive treatment for refractory partial seizures. Epilepsia. 2004;45:610–7.
Brodie MJ, Duncan R, Vespignani H, Solyom A, Bitenskyy V, Lucas C. Dose-dependent safety and efficacy of zonisamide: a randomized, double-blind, placebo-controlled study in patients with refractory partial seizures. Epilepsia. 2005;46:31–41.
Lu Y, Zhanqin X, Yu W, et al. Efficacy and safety of adjunctive zonisamide in adult patients with refractory partial-onset epilepsy. Clin Drug Investig. 2011;31:221–9.
Guerrini R, Rosati A, Segieth J, Pellacani S, Bradshaw K, Giorgi L. A randomized phase III trial of adjunctive zonisamide in pediatric patients with partial epilepsy. Epilepsia. 2013;54:1473–80.
Baulac M, Brodie MJ, Patten A, Segieth J, Giorgi L. Efficacy and tolerability of zonisamide versus controlled-release carbamazepine for newly diagnosed partial epilepsy: a phase 3, randomised, double-blind, non-inferiority trial. Lancet Neurol. 2012;11:579–88.
Wroe SJ, Yeates AB, Marshall A. Long-term safety and efficacy of zonisamide in patients with refractory partial-onset epilepsy. Acta Neurol Scand. 2008;118:87–93.
Hasegawa H. Utilization of zonisamide in patients with chronic pain or epilepsy refractory to other treatments: a retrospective, open label, uncontrolled study in a VA hospital. Curr Med Res Opin. 2004;20:577–80.
Dupont S, Striano S, Trinka E, et al. Flexible dosing of adjunctive zonisamide in the treatment of adult partial-onset seizures: a non-comparative, open-label study (ZEUS). Acta Neurol Scand. 2010;121:141–8.
Catarino CB, Bartolini E, Bell GS, Yuen AWC, Duncan JS, Sander JW. The long-term retention of zonisamide in a large cohort of people with epilepsy at a tertiary referral centre. Epilepsy Res. 2011;96:39–44.
Helmstaedter C, Stefan H, Witt JA. Quality of life in patients with partial-onset seizures under adjunctive therapy with zonisamide: results from a prospective non-interventional surveillance study. Epileptic Disord. 2011;13:263–76.
Heo K, Lee BI, Yi SD, et al. Short-term efficacy and safety of zonisamide as adjunctive treatment for refractory partial seizures: a multicenter open-label single-arm trial in Korean patients. Seizure. 2012;21:188–93.
Kluger G, Zsoter A, Holthausen H. Long-term use of zonisamide in refractory childhood-onset epilepsy. Eur J Paediatr Neurol. 2008;12:19–23.
Coppola G, Grosso S, Verrotti A, et al. Zonisamide in children and young adults with refractory epilepsy: an open-label, multicenter Italian study. Epilepsy Res. 2009;83:112–6.
Shinnar S, Pellock JM, Conry JA. Open-label, long-term safety study of zonisamide administered to children and adolescents with epilepsy. Eur J Paediatr Neurol. 2009;13:3–9.
Tan HJ, Martland TR, Appleton RE, Kneen R. Effectiveness and tolerability of zonisamide in children with epilepsy: a retrospective review. Seizure. 2010;19:31–5.
Mandelbaum DE, Bunch M, Kugler SL, Venkatasubramanian A, Wollack JB. Broad-spectrum efficacy of zonisamide at 12 months in children with intractable epilepsy. J Child Neurol. 2005;20:594–7.
Lee YJ, Kang HC, Seo JH, Lee JS, Kim HD. Efficacy and tolerability of adjunctive therapy with zonisamide in childhood intractable epilepsy. Brain Dev. 2010;32:208–12.
Park S, Kim S, Hwang Y, Lee H, Suh C, Kwon S. Long-term efficacy and safety of zonisamide monotherapy in epilepsy patients. J Clin Neurol. 2007;3:175–80.
Seki T, Kumagai N, Maezawa M. Effects of zonisamide monotherapy in children with epilepsy. Seizure. 2004;13:26–32.
Kothare SV, Kaleyias J, Mostofi N, et al. Efficacy and safety of zonisamide monotherapy in a cohort of children with epilepsy. Paediatr Neurol. 2006;35:351–4.
Eun S, Kim HD, Eun B, et al. Comparative trial of low- and high-dose zonisamide as monotherapy for childhood epilepsy. Seizure. 2011;20:558–63.
Wilfong AA. Zonisamide monotherapy for epilepsy in children and young adults. J Paediatr Neurol. 2005;32:77–80.
Iinuma K, Haginoya K. Clinical efficacy of zonisamide in childhood epilepsy after long-term treatment: a postmarketing, multi-institutional survey. Seizure. 2004;13:34–9.
Kim HL, Aldridge J, Rho JM. Clinical experience with zonisamide monotherapy and adjunctive therapy in children with epilepsy at a tertiary care referral center. J Child Neurol. 2005;20:212–9.
Wilensky AJ, Friel PN, Ojemann LM, Dodrill CB, McCormick KB, Levy RH. Zonisamide in epilepsy: a pilot study. Epilepsia. 1985;26:212–20.
Faught E. Review of United States and European clinical trials of zonisamide in the treatment of refractory partial-onset seizures. Seizure. 2004;13:59–65.
Park S, Hwang Y, Lee H, Suh C, Kwon S, Lee B. Long-term cognitive and mood effects of zonisamide monotherapy in epilepsy patients. Epilepsy Behav. 2008;12:102–8.
Cavanna AE, Ali F, Rickards HE, McCorry D. Behavioural and cognitive effects of anti-epileptic drugs. Discov Med. 2010;9:138–44.
Eddy CM, Rickards HE, Cavanna AE. The cognitive impact of antiepileptic drugs. Ther Adv Neurol Disord. 2011;4:385–407.
Eddy CM, Rickards H, Cavanna AE. Behavioral adverse effects of antiepileptic drugs in epilepsy. J Clin Psychopharmacol. 2012;32:362–75.
Piedad J, Rickards H, Besag F, Cavanna AE. Beneficial and adverse psychotropic effects of antiepileptic drugs in patients with epilepsy: a summary of prevalence, underlying mechanisms and data limitations. CNS Drugs. 2012;26:319–35.
Cavanna AE, Seri S. Psychiatric adverse effects of zonisamide in patients with epilepsy and mental disorder comorbidities. Epilepsy Behav. 2013;29:281–4.
Frampton JE, Scott LJ. Zonisamide—a review of its use in the management of partial seizures in epilepsy. CNS Drugs. 2005;19:347–67.
Brodie MJ. Zonisamide as adjunctive therapy for refractory partial seizures. Epilepsy Res. 2006;68:11–6.
Baulac M, Leppik IE. Efficacy and safety of adjunctive zonisamide therapy for refractory partial seizures. Epilepsy Res. 2007;75:75–83.
Levy RH, Ragueneau-Majlessi I, Garnett WR, et al. Lack of a clinically significant effect of zonisamide on phenytoin steady-state pharmacokinetics in patients with epilepsy. J Clin Pharmacol. 2004;44:1230–4.
Ragueneau-Majlessi I, Levy RH, Brodie M, Smith D, Shah J, Grundy JS. Lack of pharmacokinetic interactions between steady-state zonisamide and valproic acid in patients with epilepsy. Clin Pharmacokinet. 2005;44:517–23.
Levy RH, Ragueneau-Majlessi I, Brodie MJ, Smith DF, Shah J, Pan WJ. Lack of clinically significant pharmacokinetic interactions between zonisamide and lamotrigine at steady state in patients with epilepsy. Ther Drug Monit. 2005;27:193–8.
Griffith SG, Dai Y. Effect of zonisamide on the pharmacokinetics and pharmacodynamics of a combination ethinyl estradiol-norethindrone oral contraceptive in healthy women. Clin Ther. 2004;26:2056–65.
Chadwick DW, Marson AG. Zonisamide add-on for drug-resistant partial epilepsy. Cochrane Database Syst Rev. 2005;4:CD001416.
Vossler DG, Conry JA, Murphy JV, ZNS-502/505 PME Study Group. Zonisamide for the treatment of myoclonic seizures in progressive myoclonic epilepsy: an open-label study. Epileptic Disord. 2008;10:31–4.
Italiano D, Pezzella M, Coppola A, et al. A pilot open-label trial of zonisamide in Unverricht–Lundborg disease. Mov Disord. 2011;26:341–3.
Kelemen A, Rásonyl G, Neuwirth M, et al. Our clinical experience with zonisamide in resistant generalized epilepsy syndromes. Ideggyogy Sz. 2011;64:187–92.
Acknowledgments
No funding or sponsorship was received for this study or publication of this article. All named authors meet the ICMJE criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published.
Conflict of interest
Andrea E. Cavanna has received Board Membership fees and research grants from Eisai Pharmaceuticals and lectureship grants from Eisai Pharmaceuticals and Janssen-Cilag. Stefano Seri has received unrestricted educational grants from Eisai Pharmaceuticals, UCB Pharma, and Beacon Pharmaceuticals Limited. Joanna H. Cox has no conflict of interest.
Compliance with ethics guidelines
The analysis in this article is based on previously conducted studies, and does not involve any new studies of human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cox, J.H., Seri, S. & Cavanna, A.E. Zonisamide as a Treatment for Partial Epileptic Seizures: A Systematic Review. Adv Ther 31, 276–288 (2014). https://doi.org/10.1007/s12325-014-0104-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-014-0104-1