Abstract
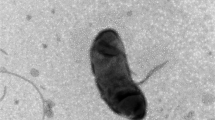
A Gram-reaction negative, strictly aerobic, non-motile, orange colored, and rod-shaped bacterium (designated BS26T) isolated from compost, was characterized by a polyphasic approach to clarify its taxonomic position. Strain BS26T was observed to grow optimally at 25–30°C and at pH 7.0 on R2A and nutrient media. Strain BS26T showed ß-glucosidase activity that was responsible for its ability to transform ginsenoside Rb1 (one of the active components of ginseng) to ginsenoside compound-K (C-K). Phylogenetic analysis based on 16S rRNA gene sequences indicated that strain BS26T belongs to the genus Niabella of family Chitinophagaceae and was most closely related to Niabella soli DSM 19437T (94.5% similarity), N. yanshanensis CCBAU 05354T (94.3%), and N. aurantiaca DSM 17617T (93.8%). The G+C content of genomic DNA was 47.3 mol%. Chemotaxonomic data [predominant isoprenoid quinone-MK-7, major fatty acids–iso-C15:0, iso-C15:1 G, iso-C17:0 3-OH, and summed feature 3 (comprising C16:1 ?7c and/or C16:1 ?6c)] supported the affiliation of strain BS26T to the genus Niabella. However, strain BS26T could be differentiated genotypically and phenotypically from the recognized species of the genus Niabella. The novel isolate therefore represents a novel species, for which the name Niabella ginsenosidivorans sp. nov. is proposed, with the type strain BS26T (=KACC 16620T =JCM 18199T).
Similar content being viewed by others
References
Ahn, J.H., Jo, E.H., Kim, B.Y., Song, J., Kwon, S.W., and Weon, H.Y. 2013. Niabella terrae sp. nov. isolated from greenhouse soil. J. Microbiol. 51, 731–735.
Atlas, R.M. 1993. Handbook of Microbiological Media. CRC Press, Boca Raton, USA.
Buck, J.D. 1982. Nonstaining (KOH) method for determination of Gram reactions of marine bacteria. Appl. Environ. Microbiol. 44, 992–993.
Cappuccino, J.G. and Sherman, N. 2002. Microbiology: a laboratory manual, 6th ed., Pearson Education, Inc., California, USA.
Dai, J., Jiang, F., Wang, Y., Yu, B., Qi, H., Fang, C., and Zheng, C. 2011. Niabella tibetensis sp. nov., isolated from soil, and emended description of the genus Niabella. Int. J. Syst. Evol. Microbiol. 61, 1201–1205.
Du, J., Cui, C.H., Park, S.C., Kim, J.K., Yu, H.S., Jin, F.X., Sun, C., Kim, S.C., and Im, W.T. 2014. Identification and characterization of a ginsenoside-transforming β-glucosidase from Pseudonocardia sp. Gsoil 1536 and its application for enhanced production of minor ginsenoside Rg2(S). PLoS One 9, e96914.
Euzeby, J.P. 1997. List of bacterial names with standing in nomenclature: A folder available on the internet. Int. J. Syst. Microbiol. 47, 590–592.
Felsenstein, J. 1981. Evolutionary trees from DNA sequences: a maximum likelihood approach. J. Mol. Evol. 17, 368–376.
Felsenstein, J. 1985. Confidence limit on phylogenies: an approach using the bootstrap. Evolutio. 39, 783–791.
Fitch, W.M. 1971. Toward defining the course of evolution: minimum change for a specific tree topology. Syst. Zool. 20, 406–416.
Glaeser, S.P., Galatis, H., Martin, K., and Kämpfer, P. 2013. Niabella hirudinis and Niabella drilacis sp. nov., isolated from the medicinal leech Hirudo verbana. Int. J. Syst. Evol. Microbiol. 63, 3487–3493.
Hall, T.A. 1999. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 41, 95–98.
Hiraishi, A., Ueda, Y., Ishihara, J., and Mori, T. 1996. Comparative lipoquinone analysis of influent sewage and activated sludge by high-performance liquid chromatography and photodiode array detection. J. Gen. Appl. Microbiol. 42, 457–469.
Kim, O.S., Cho, Y.J., Lee, K., Yoon, S.H., Kim, M., Na, H., Park, S.C., Jeon, Y.S., Lee, J.H., Yi, H., et al. 2012. Introducing eztaxon-e: A prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int. J. Syst. Evol. Microbiol. 62, 716–721.
Kim, J.K., Choi, T.E., Liu, Q.M., Park, H.Y., Yi, T.H., Yoon, M.H., Kim, S.C., and Im, W.T. 2013a. Mucilaginibacter ginsenosidivorax sp. nov., with ginsenoside converting activity isolated from sediment. J. Microbiol. 51, 394–399.
Kim, J.K., Cui, C.H., Liu, Q., Yoon, M.H., Kim, S.C., and Im, W.T. 2013b. Mass production of the ginsenoside Rg3(S) through the combinative use of two glycoside hydrolases. Food Chem. 141, 1369–1377.
Kim, B.Y., Weon, H.Y., Yoo, S.H., Hong, S.B., Kwon, S.W., Stackebrandt, E., and Go, S.J. 2007. Niabella aurantiaca gen. nov., sp. nov., isolated from a greenhouse soil in Korea. Int. J. Syst. Evol. Microbiol. 57, 538–541.
Kimura, M. 1983. The Neutral Theory of Molecular Evolution. Cambridge: Cambridge University Press, Cambridge, New York, USA.
Mesbah, M., Premachandran, U., and Whitman, W. 1989. Precise measurement of the G+C content of deoxyribonucleic acid by high performance liquid chromatography. Int. J. Syst. Bacteriol. 39, 159–167.
Moore, D.D. and Dowhan, D. 1995. Preparation and analysis of DNA, pp. 2–11. In Ausubel, F.W., Brent, R., Kingston, R.E., Moore, D.D., Seidman, J.G., Smith, J.A., and Struhl, K. (eds.), Current Protocols in Molecular Biology. Wiley, New York, USA.
Pham, V.H. and Kim, J. 2014. Niabella thaonhiensis sp. nov., isolated from the forest soil of Kyonggi University in Korea. Curr. Microbiol. 69, 176–181.
Reichenbach, H. 1992. The order Cytophagales. The Prokaryotes. A Handbook on the Biology of Bacteria: Ecophysiology, Isolation, Identification, Applications, 2nd edn, pp. 3631–3675. In Balows, A., Trüper, H.G., Dworkin, M., Harder, W., and Schleifer, K.H. (eds.). Springer, New York, USA.
Saitou, N. and Nei, M. 1987. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4, 406–425.
Sasser, M. 1990. Identification of bacteria by gas chromatography of cellular fatty acids. MIDI Technical Note 101. MIDI Inc., Newark, DE, USA.
Stackebrandt, E. and Goebel, B.M. 1994. Taxonomic note: A place for DNA-DNA reassociation and 16S rRNA sequence analysis in the present species definition in bacteriology. Int. J. Syst. Bacteriol. 44, 846–849.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., and Kumar, S. 2013. MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol. Biol. Evol. 30, 2725–2729.
Ten, L.N., Im, W.T., Kim, M.K., Kang, M.S., and Lee, S.T. 2004. Development of a plate technique for screening of polysaccharidedegrading microorganisms by using a mixture of insoluble chromogenic substrates. J. Microbiol. Method. 56, 375–382.
Thompson, J.D., Gibson, T.J., Plewniak, F., Jeanmougin, F., and Higgins, D.G. 1997. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 25, 4876–4882.
Wang, H., Zhang, Y.Z., Man, C.X., Chen, W.F., Sui, X.H., Li, Y., Zhang, X.X., and Chen, W.X. 2009. Niabella yanshanensis sp. nov., isolated from the soybean rhizosphere. Int. J. Syst. Evol. Microbiol. 59, 2854–2856.
Weon, H.Y., Kim, B.Y., Joa, J.H., Kwon, S.W., Kim, W.G., and Koo, B.S. 2008. Niabella soli sp. nov., isolated from soil from Jeju Island, Korea. Int. J. Syst. Evol. Microbiol. 58, 467–469.
Weon, H.Y., Yoo, S.H., Kim, B.Y., Son, J.A., Kim, Y.J., and Kwon, S.W. 2009. Niabella ginsengisoli sp. nov., isolated from soil cultivated with Korean ginseng. Int. J. Syst. Evol. Microbiol. 59, 1282–1285.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yi, KJ., Im, WT., Kim, DW. et al. Niabella ginsenosidivorans sp. nov., isolated from compost. J Microbiol. 53, 762–766 (2015). https://doi.org/10.1007/s12275-015-5463-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-015-5463-z