Abstract
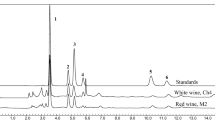
An analytical method for simultaneous determination of total polyphenol content (TPC) and antioxidant activity (AA) of wines (white and red wines) and oenological tannins, using a flow injection system with sequential diode array and electrochemical amperometry detectors (DAD-ECD), was proposed. The signal at 280 nm provided aggregate data for TPC. The anodic peak related to wine phenolic oxidation was scanned using pulsed integrated amperometry over the potential of 800 mV vs. Ag/AgCl, to obtain AA. Serial dilutions avoided the poisoning at the glassy carbon (GC) electrode and the linear response obtained with both detectors was compared with spectrophotometric assays commonly used in oenology laboratory. Intraday and interday analytical repetitions showed a good repeatability and reproducibility (relative standard deviation RSD < 6% for both detectors), and the satisfactory relationship between the proposed coupled flow injection/DAD-ECD and the classic UV methods (R2TPC = 0.9967; R2DPPH = 0.9621) confirmed the efficacy of flow injection analysis with a coupled detection system, for the reliable quality control of wine and wine-related products.



Similar content being viewed by others
References
Aid T, Kaljurand M, Vaher M (2015) Colorimetric determination of total phenolic contents in ionic liquid extracts by paper microzones and digital camera. Anal Methods 7:3193–3199. https://doi.org/10.1039/c5ay00194c
Arce L, Tena MT, Rios A, Valcàrcel M (1998) Determination of trans-resveratrol and other polyphenols in wines by a continuous flow sample clean-up system followed by capillary electrophoresis separation. Anal Chim Acta 359:27–38. https://doi.org/10.1016/S0003-2670(97)00668-5
Arnous A, Makris DP, Kefalas P (2002) Correlation of pigment and flavanol content with antioxidant properties in selected aged regional wines from Greece. J Food Compos Anal 15:655–665. https://doi.org/10.1006/jfca.2002.1070
Arteaga JF, Ruiz-Montoya M, Palma A, Alonso-Garrido G, Pintado S, Rodríguez-Mellado JM (2012) Comparison of the simple cyclic voltammetry (CV) and DPPH assays for the determination of antioxidant capacity of active principles. Molecules 17:5126–5138. https://doi.org/10.3390/molecules17055126
Bland JM, Altman DG (1999) Measuring agreement in method comparison studies. Stat Methods Med Res 8:135–160. https://doi.org/10.1177/096228029900800204
Bobo-García G, Davidov-Pardo G, Arroqui C, Vírseda P, Marín-Arroyo MR, Navarro M (2014) Intra-laboratory validation of microplate methods for total phenolic content and antioxidant activity on polyphenolic extracts, and comparison with conventional spectrophotometric methods. J Sci Food Agric 95:204–209. https://doi.org/10.1002/jsfa.6706
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol 28:25–30. https://doi.org/10.1016/S0023-6438(95)80008-5
Chen W, Li W, Fu P, Yeh A (2011) Reactivity of DPPH in the oxidation of catechol and catechin. In J Chem Kinet 43:147–153. https://doi.org/10.1002/kin.20542
Danilewicz JC (2013) Reactions involving iron in mediating catechol oxidation in model wine. Am J Enol Vitic 64:316–324. https://doi.org/10.5344/ajev.2013.12137
Danilewicz JC (2015) The Folin-Ciocalteu, FRAP, and DPPH• assays for measuring polyphenol concentration in white wine. Am J Enol Vitic 66:463–470. https://doi.org/10.5344/ajev.2015.15025
Dicu T, Postescu ID, Tatomir C, Tămaş M, Dinu A, Cosma C (2010) A novel method to calculate the antioxidant parameters of the redox reaction between polyphenolic compounds and the stable DPPH radical. Ital J Food Sci 22:330–336
Foti M, Ruberto G (2001) Kinetic solvent effects on phenolic antioxidants determined by spectrophotometric measurements. J Agric Food Chem 49:342–348. https://doi.org/10.1021/jf0006527
Gizdavic-Nikolaidis M, Travas-Sejdic J, Bowmaker GA, Cooney RP, Thompson C, Kilmartin PA (2004) The antioxidant activity of conducting polymers in biomedical applications. Curr Appl Phys 4:347–350. https://doi.org/10.1016/j.cap.2003.11.045
Gòmez-Alonso S, Garcìa-Romero E, Hermosìn-Gutiérrez I (2007) HPLC analysis of diverse grape and wine phenolics using direct injection and multidetection by DAD and fluorescence. J Food Compos Anal 20:618–626. https://doi.org/10.1016/j.jfca.2007.03.002
Harbertson JF, Spayd S (2006) Measuring phenolics in the winery. Am J Enol Vitic 57:280–288
Haslam E (1998) Practical polyphenolics: from structure to molecular recognition and physiological action. Cambridge University Press, Cambridge
Ivanova V, Dörnyei Á, Márk L, Vojnoski B, Stafilov T, Stefova M, Kilár F (2011) Polyphenolic content of Vranec wines produced by different vinification conditions. Food Chem 124:316–325. https://doi.org/10.1016/j.foodchem.2010.06.039
Ivanova-Petropulos V, Ricci A, Nedelkovski D, Dimovska V, Parpinello GP, Versari A (2015) Targeted analysis of bioactive phenolic compounds and antioxidant activity of Macedonian red wines. Food Chem 171:412–420. https://doi.org/10.1016/j.foodchem.2014.09.014
Kilmartin PA, Zou H, Waterhouse AL (2001) A cyclic voltammetry method suitable for characterizing antioxidant properties of wine and wine phenolics. J Agric Food Chem 49:1957–1965. https://doi.org/10.1021/jf001044u
Litwinienko G, Ingold KU (2003) Abnormal solvent effects on hydrogen atom abstractions. 1. The reactions of phenols with 2,2-diphenyl-1-picrylhydrazyl (dpph•) in alcohols. J Org Chem 68:3433–3438. https://doi.org/10.1021/jo026917t
Lorrain B, Ky I, Pechamat L, Teissedre PL (2013) Evolution of analysis of polyphenols from grapes, wines, and extracts. Molecules 18:1076–1100. https://doi.org/10.3390/molecules18011076
Luque de Castro MD, González-Rodríguez J, Pérez-Juan P (2005) Analytical methods in wineries: is it time to change? Food Rev Intl 21:231–265
Magalhães LM, Ramos II, Reis S, Segundo MA (2014) Antioxidant profile of commercial oenological tannins determined by multiple chemical assays. Aust J Grape Wine Res 20:72–79. https://doi.org/10.1111/ajgw.12058
Makhotkina O, Kilmartin PA (2010) The use of cyclic voltammetry for wine analysis: determination of polyphenols and free sulfur dioxide. Anal Chim Acta 668:155–165. https://doi.org/10.1016/j.aca.2010.03.064
Mannino S, Brenna O, Buratti S, Cosio MS (1998) A new method for the evaluation of the ‘antioxidant power’ of wines. Electroanalysis 10:908–912. https://doi.org/10.1002/(SICI)1521-4109(199810)10:13<908::AID-ELAN908>3.0.CO;2-L
Muselík J, García-Alonso M, Martín-López MP, Žemlička M, Rivas-Gonzalo JC (2007) Measurement of antioxidant activity of wine catechins, procyanidins, anthocyanins and pyranoanthocyanins. Int J Mol Sci 8:797–809. https://doi.org/10.3390/i8080797
Ribéreau-Gayon P (1970) Le dosage des composés phénoliques totaux dans les vins rouges. Chim Anal 52:627–631
Rivero-Pérez MD, Muñiz P, González-Sanjosé ML (2007) Antioxidant profile of red wines evaluated by total antioxidant capacity, scavenger activity, and biomarkers of oxidative stress methodologies. J Agric Food Chem 55:5476–5483. https://doi.org/10.1021/jf070306q
Šeruga M, Novak I, Jakobek L (2011) Determination of polyphenols content and antioxidant activity of some red wines by differential pulse voltammetry, HPLC and spectrophotometric methods. Food Chem 124:1208–1216. https://doi.org/10.1016/j.foodchem.2010.07.047
Singleton V, Esau P (1969) Phenolic substances in grape and wine and their significance. Academic Press, New York. https://doi.org/10.1136/pgmj.45.530.776
Tenore GC, Basile A, Novellino E (2011) Antioxidant and antimicrobial properties of polyphenolic fractions from selected Moroccan red wines. J Food Sci 76:C1342–C1348. https://doi.org/10.1111/j.1750-3841.2011.02426.x
Acknowledgments
Authors gratefully acknowledge the HTS Enologia and Laffort companies for having supplied food-grade tannin samples.
Funding
Authors A.R. and N.T. had PhD grants by the University of Bologna (Italy) and Penta EU program, respectively. Author V.I.P. was funded by the ERASMUS Mundus Action 2 Project while carrying out this experiment.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Arianna Ricci declares that she has no conflict of interest. Nemanja Teslic declares that he has no conflict of interest. Violeta-Ivanova Petropolus declares that she has no conflict of interest. Giuseppina Paola Parpinello declares that she has no conflict of interest. Andrea Versari declares that she has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Ricci, A., Teslic, N., Petropolus, VI. et al. Fast Analysis of Total Polyphenol Content and Antioxidant Activity in Wines and Oenological Tannins Using a Flow Injection System with Tandem Diode Array and Electrochemical Detections. Food Anal. Methods 12, 347–354 (2019). https://doi.org/10.1007/s12161-018-1366-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-018-1366-z