Abstract
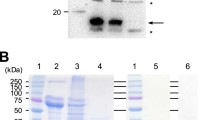
Three forms of recombinant protein complexes comprising the human prorenin (hPro) and (pro)renin receptor (hPRR) (hPRR/prorenin) were successfully expressed in the silkworm larvae using Bombyx mori nucleopolyhedrovirus (BmNPV) bacmids. They were localized in the fat body cells and formed a prorenin-bound hPRR complex. The expressed levels of hPro and hPRR were similar judging from Western blotting. The hPRR/prorenin complex containing 40 μg of hPRR (yield, 43%) and 30 μg of hPro (yield, 34%) was purified from 15 silkworm larvae by a series of purification using anti-FLAG and Strep-Tactin affinity chromatography. The renin activity of the purified hPRR/prorenin complex was 3.8-fold that of the mixture of hPRR and hPro expressed individually in vitro judging from the renin assay. These results show that the unstable transmembrane protein, hPRR, was coexpressed stably with ligand, hPro, and formed a stable protein, hPRR/prorenin complex that showed a high catalytic active form.





Similar content being viewed by others
References
Wallrabe, H., Elangovan, M., Burchard, A., Periasamy, A., & Barroso, M. (2003). Confocal FRET microscopy to measure clustering of ligand–receptor complexes in endocytic membranes. Biophysical Journal, 85, 559–571.
Fenge, C., Jansson, I., Fröberg, T., Jönsson, M., Lüllau, E., Sygowski, L., et al. (2002). Process development for functional membrane receptor production in mammalian cells. Cytotechnology, 38, 109–117.
Kato, T., Park, E. Y., Murata, T., & Usui, T. (2004). Efficient production of human (1, 3-N-acetylglucosaminyltransferase 2 fused with green fluorescent protein in insect cell. Biochemical Engineering Journal, 19, 15–23.
Massotte, D. (2003). G protein-coupled receptor overexpression with the baculovirus–insect cell system: A tool for structural and functional studies. Biochimica et Biophysica Acta, 1610, 77–89.
Motohashi, T., Shimojima, T., Fukagawa, T., Maenaka, K., & Park, E. Y. (2005). Efficient large-scale protein production of larvae and pupae of silkworm nuclear polyhedrovirus (BmNPV) bacmid system. Biochemical and Biophysical Research Communications, 326, 564–569.
Suzuki, F., Hayakawa, M., Nakagawa, T., Nasir, U. M., Ebihara, A., Iwasawa, A., et al. (2003). Human prorenin has “gate and handle” regions for its non-proteolytic activation. The Journal of Biological Chemistry, 278, 22217–22222.
Nguyen, G., Delarue, F., Burckle, C., Bouzhir, L., Giller, T., & Sraer, J. -D. (2002). Pivotal role of the renin/prorenin receptor in angiotensin II production and cellular responses to renin. The Journal of Clinical Investigation, 109, 1417–1427.
Burcklé, C. A., Danser, A. H. J., Müller, D. N., Garrelds, I. M., Gasc, J. M., Popova, E., et al. (2006). Elevated blood pressure and heart rate in human renin receptor transgenic rats. Hypertension, 47, 552–556.
Danser, A. H., & Deinum, J. (2005). Renin, prorenin and the putative (pro)renin receptor. Hypertension, 46, 1069–1076.
Ichihara, A., Hayashi, M., Kaneshiro, Y., Suzuki, F., Nakagawa, T., Tada, Y., et al. (2004). Inhibition of diabetic nephropathy by a decoy peptide corresponding to the “handle” region for nonproteolytic activation of prorenin. The Journal of Clinical Investigation, 114, 1128–1135.
Du, D., Kato, T., Nabi, A. N., Suzuki, F., & Park, E. Y. (2008). Expression of functional human (pro)renin receptor in silkworm larvae using BmNPV bacmid. Biotechnology and Applied Biochemistry, 49, 195–202.
Kato, T., Kageshima, A., Suzuki, F., & Park, E. Y. (2008). Expression and purification of human (pro)renin receptor in insect cells using baculovirus expression system. Protein Expression and Purification, 58, 242–248.
Hiyoshi, M., Kageshima, A., Kato, T., & Park, E. Y. (2007). Construction of cysteine protease deficient Bombyx mori nuclear polyhedrosis bacmid and its application on improved expression of fusion protein. Journal of Virological Methods, 144, 91–97.
Suzuki, F., Hatano, Y., Nakagawa, T., Terazawa, K., Gotoh, A., Nasir, U. M., et al. (1999). Non-proteolytic activation of human prorenin by anti-prorenin prosegment (pf#1: 1P–15P) antiserum. Bioscience, Biotechnology, and Biochemistry, 63, 550–554.
Batenburg, W. W., Krop, M., Garrelds, I. M., de Vries, R., de Bruin, R. J., Burcklé, C. A., et al. (2007). Prorenin is the endogenous agonist of the (pro)renin receptor. Binding kinetics of renin and prorenin in rat vascular smooth muscle cells overexpressing the human (pro)renin receptor. Journal of Hypertension, 25, 2441–2453.
Kaneshiro, Y., Ichihara, A., Takemitsu, T., Sakoda, M., Suzuki, F., Nakagawa, T., et al. (2006). Increased expression of cyclooxygenase-2 in the renal cortex of human prorenin receptor gene-transgenic rats. Kidney International, 70, 641–646.
Huang, Y., Wongamorntham, S., Kasting, J., McQuillan, D., Owens, R. T., Yu, L., et al. (2006). Renin increases mesangial cell transforming growth factor-β1 and matrix proteins through receptor-mediated, angiotensin II-independent mechanisms. Kidney International, 69, 105–113.
Du, D., Kato, T., Suzuki F., & Park, E. Y. (2009). Binding affinity of full-length and extracellular domains of recombinant human (pro)renin receptors to human renin when expressed in the fat body and hemolymph of silkworm larvae. Journal of Bioscience and Bioengineering, in press.
Mendrola, J. M., Berger, M. B., King, M. C., & Lemmon, M. A. (2002). The single transmembrane domains of ErbB receptors self-associate in cell membranes. The Journal of Biological Chemistry, 277, 4704–4712.
Chiang, W. C., & Knowles, A. F. (2008). Transmembrane domain interactions affects the stability of the extracellular domain of the human NTPDase 2. Archives of Biochemistry and Biophysics, 472, 89–99.
Acknowledgments
This work was supported by the Program for Promotion of Basic Research Activities for Innovative Biosciences (PROBRAIN), Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Du, D., Kato, T., Suzuki, F. et al. Expression of Protein Complex Comprising the Human Prorenin and (Pro)Renin Receptor in Silkworm Larvae Using Bombyx mori Nucleopolyhedrovirus (BmNPV) Bacmids for Improving Biological Function. Mol Biotechnol 43, 154–161 (2009). https://doi.org/10.1007/s12033-009-9183-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-009-9183-7