Abstract
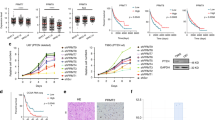
Posttranslational modifications of histones are considered as critical regulators of gene expression, playing significant role in the pathogenesis and progression of tumors. Trimethylation of histone 3 lysine 9 (H3K9me3), a repressed transcription mark, is mainly regulated by the histone lysine N-methyltransferases (HKMTs), SUV39H1 and SETDB1. The present study investigated the implication of these HKMTs in glioma progression. SUV39H1 and SETDB1 expression was upregulated in glioma cell lines (GOS-3, 1321N1, T98G, U87MG) and in glioma tissues compared to normal brain being positively correlated with grade and histological malignancy. Suppression by siRNA of the two HKMTs for 24 and 48 h resulted in significantly reduced proliferation of GOS-3 and T98G glioma cells with siSUV39H1 effects been most prominent. Furthermore, HKMTs knockdown-induced apoptosis with a high rate of apoptotic cells have been observed after siSUV39H1 and siSETDB1 for both cell lines. Additionally, suppression of the two HKMTs reduced cell migration and clonogenic ability of both glioma cell lines. Our results indicate overexpression of SETDB1 and SUV39H1 in gliomas. Treatments that alter HKMT expression affect the proliferative and apoptotic rates in glioma cells as well as their migratory and colony formation capacity. These data suggest that both HKMTs and especially SUV39H1 may serve as novel biomarkers for future therapeutic targeting of these tumors.






Similar content being viewed by others
Abbreviations
- BRAF:
-
Serine/threonine-protein kinase B-raf
- Dnmt:
-
DNA (cytosine-5)-methyltransferase
- DOT1L:
-
Histone-lysine N-methyltransferase, H3 lysine-79 specific
- EZH2:
-
Enhancer of Zeste 2
- G9A:
-
Histone-lysine N-methyltransferase EHMT2
- HCC:
-
Hepatocellular carcinoma
- HKDM:
-
Histone lysine demethylase
- HKMT:
-
Histone lysine N-methyltransferase
- HP1:
-
Heterochromatin protein 1
- IDH1:
-
Isocitrate dehydrogenase 1
- SET9:
-
Histone-lysine N-methyltransferase SETD7
- SETDB1:
-
SET domain bifurcated 1
- SMYD3:
-
SET and MYND domain-containing protein 3
- SUV39H1:
-
Suppressor of variegation 3–9 homolog 1
References
Aagaard, L., Schmid, M., Warburton, P., & Jenuwein, T. (2000). Mitotic phosphorylation of SUV39H1, a novel component of active centromeres, coincides with transient accumulation at mammalian centromeres. Journal of Cell Science, 113, 817–829.
Alimova, I., Venkataraman, S., Harris, P., Marquez, V. E., Northcott, P. A., Dubuc, A., et al. (2012). Targeting the enhancer of zeste homologue 2 in medulloblastoma. International Journal of Cancer, 131, 1800–1809.
Ayyanathan, K., Lechner, M. S., Bell, P., Maul, G. G., Schultz, D. C., Yamada, Y., et al. (2003). Regulated recruitment of HP1 to a euchromatic gene induces mitotically heritable, epigenetic gene silencing: A mammalian cell culture model of gene variegation. Genes & Development, 17, 1855–1869.
Babbio, F., Pistore, C., Curti, L., Castiglioni, I., Kunderfranco, P., Brino, L., et al. (2012). The SRA protein UHRF1 promotes epigenetic crosstalks and is involved in prostate cancer progression. Oncogene, 31, 4878–4887.
Baumgart, S., Glesel, E., Singh, G., Chen, N. M., Reutlinger, K., Zhang, J., et al. (2012). Restricted heterochromatin formation links NFATc2 repressor activity with growth promotion in pancreatic cancer. Gastroenterology, 142, 388–398.
Bennani-Baiti, I. M. (2011). Epigenetic and epigenomic mechanisms shape sarcoma and other mesenchymal tumor pathogenesis. Epigenomics, 3, 715–732.
Bracken, A. P., Pasini, D., Capra, M., Prosperini, E., Colli, E., & Helin, K. (2003). EZH2 is downstream of the pRB-E2F pathway, essential for proliferation and amplified in cancer. The EMBO journal, 22, 5323–5335.
Braig, M., Lee, S., Loddenkemper, C., Rudolph, C., Peters, A. H., Schlegelberger, B., et al. (2005). Oncogene-induced senescence as an initial barrier in lymphoma development. Nature, 436, 660–665.
Carbone, R., Botrugno, O. A., Ronzoni, S., Insinga, A., Di Croce, L., Pelicci, P. G., et al. (2006). Recruitment of the histone methyltransferase SUV39H1 and its role in the oncogenic properties of the leukemia-associated PML-retinoic acid receptor fusion protein. Molecular and Cellular Biology, 26, 1288–1296.
Ceol, C. J., Houvras, Y., Jane-Valbuena, J., Bilodeau, S., Orlando, D. A., Battisti, V., et al. (2011). The histone methyltransferase SETDB1 is recurrently amplified in melanoma and accelerates its onset. Nature, 471, 513–517.
Chaib, H., Nebbioso, A., Prebet, T., Castellano, R., Garbit, S., Restouin, A., et al. (2012). Anti-leukemia activity of chaetocin via death receptor-dependent apoptosis and dual modulation of the histone methyl-transferase SUV39H1. Leukemia, 26, 662–674.
Chernov, A. V., Sounni, N. E., Remacle, A. G., & Strongin, A. Y. (2009). Epigenetic control of the invasion-promoting MT1-MMP/MMP-2/TIMP-2 axis in cancer cells. The Journal of Biological Chemistry, 284, 12727–12734.
Collett, K., Eide, G. E., Arnes, J., Stefansson, I. M., Eide, J., Braaten, A., et al. (2006). Expression of enhancer of zeste homologue 2 is significantly associated with increased tumor cell proliferation and is a marker of aggressive breast cancer. Clinical Cancer Research, 12, 1168–1174.
Dong, C., Wu, Y., Wang, Y., Wang, C., Kang, T., Rychahou, P. G., et al. (2013). Interaction with Suv39H1 is critical for Snail-mediated E-cadherin repression in breast cancer. Oncogene, 32, 1351–1362.
Fan, D. N., Tsang, F. H., Tam, A. H., Au, S. L., Wong, C. C., Wei, L., et al. (2013). Histone lysine methyltransferase, suppressor of variegation 3-9 homolog 1, promotes hepatocellular carcinoma progression and is negatively regulated by microRNA-125b. Hepatology, 57, 637–647.
Fanelli, M., Caprodossi, S., Ricci-Vitiani, L., Porcellini, A., Tomassoni-Ardori, F., Amatori, S., et al. (2008). Loss of pericentromeric DNA methylation pattern in human glioblastoma is associated with altered DNA methyltransferases expression and involves the stem cell compartment. Oncogene, 27, 358–365.
Farmaki, E., Mkrtchian, S., Papazian, I., Papavassiliou, A. G., & Kiaris, H. (2011). ERp29 regulates response to doxorubicin by a PERK-mediated mechanism. Biochimica et Biophysica Acta, 1813, 1165–1171.
Firestein, R., Cui, X., Huie, P., & Cleary, M. L. (2000). Set domain-dependent regulation of transcriptional silencing and growth control by SUV39H1, a mammalian ortholog of Drosophila Su(var)3-9. Molecular and Cellular Biology, 20, 4900–4909.
Horbinski, C. (2013). To BRAF or not to BRAF: Is that even a question anymore? Journal of Neuropathology and Experimental Neurology, 72, 2–7.
Hou, P., Liu, D., Dong, J., & Xing, M. (2012). The BRAF(V600E) causes widespread alterations in gene methylation in the genome of melanoma cells. Cell Cycle, 11, 286–295.
Kang, M. Y., Lee, B. B., Kim, Y. H., Chang, D. K., Kyu Park, S., Chun, H. K., et al. (2007). Association of the SUV39H1 histone methyltransferase with the DNA methyltransferase 1 at mRNA expression level in primary colorectal cancer. International Journal of Cancer, 121, 2192–2197.
Karpinski, P., Ramsey, D., Grzebieniak, Z., Sasiadek, M. M., & Blin, N. (2008). The CpG island methylator phenotype correlates with long-range epigenetic silencing in colorectal cancer. Molecular Cancer Research, 6, 585–591.
Kassambara, A., Klein, B., & Moreaux, J. (2009). MMSET is overexpressed in cancers: Link with tumor aggressiveness. Biochemical and Biophysical Research Communications, 379, 840–845.
Kim, H. A., Koo, B. K., Cho, J. H., Kim, Y. Y., Seong, J., Chang, H. J., et al. (2012). Notch1 counteracts WNT/beta-catenin signaling through chromatin modification in colorectal cancer. The Journal of Clinical Investigation, 122, 3248–3259.
Kleer, C. G., Cao, Q., Varambally, S., Shen, R., Ota, I., Tomlins, S. A., et al. (2003). EZH2 is a marker of aggressive breast cancer and promotes neoplastic transformation of breast epithelial cells. Proceedings of the National Academy of Sciences of the United States of America, 100, 11606–11611.
Kondo, Y., Shen, L., Ahmed, S., Boumber, Y., Sekido, Y., Haddad, B. R., et al. (2008). Downregulation of histone H3 lysine 9 methyltransferase G9a induces centrosome disruption and chromosome instability in cancer cells. PLoS One, 3, e2037.
Kreth, S., Thon, N., Eigenbrod, S., Lutz, J., Ledderose, C., Egensperger, R., et al. (2011). O-methylguanine-DNA methyltransferase (MGMT) mRNA expression predicts outcome in malignant glioma independent of MGMT promoter methylation. PLoS One, 6, e17156.
Lakshmikuttyamma, A., Scott, S. A., DeCoteau, J. F., & Geyer, C. R. (2010). Reexpression of epigenetically silenced AML tumor suppressor genes by SUV39H1 inhibition. Oncogene, 29, 576–588.
Lee, C. H., Hung, H. W., Hung, P. H., & Shieh, Y. S. (2010). Epidermal growth factor receptor regulates beta-catenin location, stability, and transcriptional activity in oral cancer. Molecular cancer, 9, 64.
Lee, J., Son, M. J., Woolard, K., Donin, N. M., Li, A., Cheng, C. H., et al. (2008). Epigenetic-mediated dysfunction of the bone morphogenetic protein pathway inhibits differentiation of glioblastoma-initiating cells. Cancer Cell, 13, 69–80.
Li, H., Rauch, T., Chen, Z. X., Szabo, P. E., Riggs, A. D., & Pfeifer, G. P. (2006). The histone methyltransferase SETDB1 and the DNA methyltransferase DNMT3A interact directly and localize to promoters silenced in cancer cells. The Journal of Biological Chemistry, 281, 19489–19500.
Lindgren, D., Sjodahl, G., Lauss, M., Staaf, J., Chebil, G., Lovgren, K., et al. (2012). Integrated genomic and gene expression profiling identifies two major genomic circuits in urothelial carcinoma. PLoS One, 7, e38863.
Loenarz, C., & Schofield, C. J. (2008). Expanding chemical biology of 2-oxoglutarate oxygenases. Nature Chemical Biology, 4, 152–156.
Lu, C., Ward, P. S., Kapoor, G. S., Rohle, D., Turcan, S., Abdel-Wahab, O., et al. (2012). IDH mutation impairs histone demethylation and results in a block to cell differentiation. Nature, 483, 474–478.
Macgregor, S., Montgomery, G. W., Liu, J. Z., Zhao, Z. Z., Henders, A. K., Stark, M., et al. (2011). Genome-wide association study identifies a new melanoma susceptibility locus at 1q21.3. Nature Genetics, 43, 1114–1118.
Martin, C., & Zhang, Y. (2005). The diverse functions of histone lysine methylation. Nature Reviews Molecular Cell Biology, 6, 838–849.
Northcott, P. A., Nakahara, Y., Wu, X., Feuk, L., Ellison, D. W., Croul, S., et al. (2009). Multiple recurrent genetic events converge on control of histone lysine methylation in medulloblastoma. Nature Genetics, 41, 465–472.
Orzan, F., Pellegatta, S., Poliani, P. L., Pisati, F., Caldera, V., Menghi, F., et al. (2011). Enhancer of Zeste 2 (EZH2) is up-regulated in malignant gliomas and in glioma stem-like cells. Neuropathology and Applied Neurobiology, 37, 381–394.
Patani, N., Jiang, W. G., Newbold, R. F., & Mokbel, K. (2011). Histone-modifier gene expression profiles are associated with pathological and clinical outcomes in human breast cancer. Anticancer Research, 31, 4115–4125.
Peters, A. H., O’Carroll, D., Scherthan, H., Mechtler, K., Sauer, S., Schofer, C., et al. (2001). Loss of the Suv39h histone methyltransferases impairs mammalian heterochromatin and genome stability. Cell, 107, 323–337.
Piperi, C., Themistocleous, M. S., Papavassiliou, G. A., Farmaki, E., Levidou, G., Korkolopoulou, P., et al. (2010). High incidence of MGMT and RARbeta promoter methylation in primary glioblastomas: Association with histopathological characteristics, inflammatory mediators and clinical outcome. Molecular Medicine, 16, 1–9.
Pogribny, I. P., Ross, S. A., Tryndyak, V. P., Pogribna, M., Poirier, L. A., & Karpinets, T. V. (2006). Histone H3 lysine 9 and H4 lysine 20 trimethylation and the expression of Suv4-20h2 and Suv-39h1 histone methyltransferases in hepatocarcinogenesis induced by methyl deficiency in rats. Carcinogenesis, 27, 1180–1186.
Portela, A., & Esteller, M. (2010). Epigenetic modifications and human disease. Nature Biotechnology, 28, 1057–1068.
Rai, K., Nadauld, L. D., Chidester, S., Manos, E. J., James, S. R., Karpf, A. R., et al. (2006). Zebra fish Dnmt1 and Suv39h1 regulate organ-specific terminal differentiation during development. Molecular and Cellular Biology, 26, 7077–7085.
Rea, S., Eisenhaber, F., O’Carroll, D., Strahl, B. D., Sun, Z. W., Schmid, M., et al. (2000). Regulation of chromatin structure by site-specific histone H3 methyltransferases. Nature, 406, 593–599.
Sarraf, S. A., & Stancheva, I. (2004). Methyl-CpG binding protein MBD1 couples histone H3 methylation at lysine 9 by SETDB1 to DNA replication and chromatin assembly. Molecular Cell, 15, 595–605.
Schultz, D. C., Ayyanathan, K., Negorev, D., Maul, G. G., & Rauscher, F. J, 3rd. (2002). SETDB1: A novel KAP-1-associated histone H3, lysine 9-specific methyltransferase that contributes to HP1-mediated silencing of euchromatic genes by KRAB zinc-finger proteins. Genes & Development, 16, 919–932.
Spyropoulou, A., Piperi, C., Adamopoulos, C., & Papavassiliou, A. G. (2013). Deregulated chromatin remodeling in the pathobiology of brain tumors. NeuroMolecular Medicine, 15, 1–24.
Turcan, S., Rohle, D., Goenka, A., Walsh, L. A., Fang, F., Yilmaz, E., et al. (2012). IDH1 mutation is sufficient to establish the glioma hypermethylator phenotype. Nature, 483, 479–483.
Varambally, S., Dhanasekaran, S. M., Zhou, M., Barrette, T. R., Kumar-Sinha, C., Sanda, M. G., et al. (2002). The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature, 419, 624–629.
Venneti, S., Felicella, M. M., Coyne, T., Phillips, J. J., Gorovets, D., Huse, J. T., et al. (2013). Histone 3 lysine 9 trimethylation is differentially associated with isocitrate dehydrogenase mutations in oligodendrogliomas and high-grade astrocytomas. Journal of Neuropathology and Experimental Neurology, 72, 298–306.
Wang, G. G., Allis, C. D., & Chi, P. (2007). Chromatin remodeling and cancer, part II: ATP-dependent chromatin remodeling. Trends in Molecular Medicine, 13, 373–380.
Wang, H., An, W., Cao, R., Xia, L., Erdjument-Bromage, H., Chatton, B., et al. (2003). mAM facilitates conversion by ESET of dimethyl to trimethyl lysine 9 of histone H3 to cause transcriptional repression. Molecular Cell, 12, 475–487.
Watanabe, H., Soejima, K., Yasuda, H., Kawada, I., Nakachi, I., Yoda, S., et al. (2008). Deregulation of histone lysine methyltransferases contributes to oncogenic transformation of human bronchoepithelial cells. Cancer Cell International, 8, 15.
Weikert, S., Christoph, F., Kollermann, J., Muller, M., Schrader, M., Miller, K., et al. (2005). Expression levels of the EZH2 polycomb transcriptional repressor correlate with aggressiveness and invasive potential of bladder carcinomas. International Journal of Molecular Medicine, 16, 349–353.
Yan, H., Parsons, D. W., Jin, G., McLendon, R., Rasheed, B. A., Yuan, W., et al. (2009). IDH1 and IDH2 mutations in gliomas. The New England Journal of Medicine, 360, 765–773.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Anastasia Spyropoulou and Antonios Gargalionis contributed equally to this work.
Rights and permissions
About this article
Cite this article
Spyropoulou, A., Gargalionis, A., Dalagiorgou, G. et al. Role of Histone Lysine Methyltransferases SUV39H1 and SETDB1 in Gliomagenesis: Modulation of Cell Proliferation, Migration, and Colony Formation. Neuromol Med 16, 70–82 (2014). https://doi.org/10.1007/s12017-013-8254-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-013-8254-x