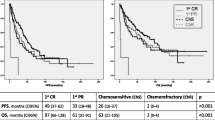
Abstract
Purpose of Review
The treatment landscape of mantle cell (MCL) and peripheral T-cell lymphomas (PTCL) is rapidly changing; however, despite improvement in patients’ survival, they still remain a largely incurable diseases. Treatment choice is dependent on patient factors, prior therapy, remission duration, and candidacy for stem cell transplantation (SCT). There are subsets of high-risk patients who do not benefit substantially from autologous SCT (ASCT) and for whom alternative targeted approaches are being examined. Here, we critically analyze the actual role of ASCT in PTCL and MCL.
Recent Findings
Research in areas of maintenance therapy and minimal residual disease is ongoing to identify MCL patients who may not require ASCT for durable response. Moreover, there are subsets of high-risk MCL patients who do not benefit substantially from ASCT and for whom alternative, targeted approaches are being examined. Much less clear evidence exists regarding the impact of consolidative ASCT in PTCL, mainly for the heterogeneity of these lymphomas: it is still controversial whether patients who achieved a complete response significantly take advantage of this procedure over active surveillance only.
Summary
Several clinical and biologic markers are available to predict prognosis; however, despite improvements in outcomes, standard therapeutic approaches have not been able to overcome high-risk disease features for PTCL and MCL. Thus, the need of ASCT for these diseases is still matter of debate among hematologists.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Broccoli A, Chihara D, Fanale M, Zinzani PL. Standard of care in T cell lymphoma. In: Lenz G, Salles G, editors. Aggressive lymphomas. Cham: Springer; 2019. p. 227–44.
Horwitz S, O’Connor OA, Pro B, Illidge T, Fanale M, Advani R, et al. Brentuximab vedotin with chemotherapy for CD30-positive peripheral T-cell lymphoma (ECHELON-2): a global, double-blind, randomised, phase 3 trial. Lancet. 2019;393:229–40. https://doi.org/10.1016/S0140-6736(18)32984-2.
Martin P, Ghione P, Dreyling. Mantle cell lymphoma–current standards of care and future directions. Cancer Treat Rev. 2017;58:51–60. https://doi.org/10.1016/j.ctrv.2017.05.008.
Adams SV, Newcomb PA, Shustov AR. Racial patterns of peripheral T-cell lymphoma incidence and survival in the United States. J Clin Oncol. 2016;34:963–71. https://doi.org/10.1200/JCO.2015.63.5540.
• Vose J, Armitage J, Weisenburger D. International T-Cell Lymphoma Project. International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes. J Clin Oncol. 2008;26:4124–30. https://doi.org/10.1200/JCO.2008.16.4558The paper states that expert hematopathology reveiw is important for accurate diagnosis of PTCL.
Ellin F, Landström J, Jerkeman M, Relander T. Real-world data on prognostic factors and treatment in peripheral T-cell lymphomas: a study from the Swedish Lymphoma Registry. Blood. 2014;124:1570–7. https://doi.org/10.1182/blood-2014-04-573089.
• Fiore D, Cappelli LV, Broccoli A, Zinzani PL, Chan WC, Inghirami G. Peripheral T cell lymphomas: from the bench to the clinic. Nat Rev Cancer. 2020;20:323–42. https://doi.org/10.1038/s41568-020-0247-0The paper discusses that the overall survival of patients with PTCLs will dramatically increased by combining new targeted agents with immune regulators and chimeric antigen receptor-expreesing natural killer and T cell.
• Swerdlow SH, Campo E, Pileri SA, Harris NL, Stein H, Siebert R, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375–90. https://doi.org/10.1182/blood-2016-01-643569Reference consensus among hematopathologists. geneticists, and clinicians regarding both updated to current entities as well as the addition of a limited number of new provisional entities in lymphoid malignancies.
Briski R, Feldman AL, Bailey NG, Lim MS, Ristow K, Habermann TM, et al. The role of front-line anthracycline-containing chemotherapy regimens in peripheral T-cell lymphomas. Blood Cancer J. 2014;4:e214. https://doi.org/10.1038/bcj.2014.34.
Abramson JS, Feldman T, Kroll-Desrosiers AR, Muffly LS, Winer E, Flowers CR, et al. Peripheral T-cell lymphomas in a large US multicentre cohort: prognostication in the modern era including impact of frontline therapy. Ann Oncol. 2014;25:2211–7. https://doi.org/10.1093/annonc/mdu443.
Corradini P, Tarella C, Zallio F, Dodero A, Zanni M, Valagussa P, et al. Long-term follow-up of patients with peripheral T-cell lymphomas treated up-front with high-dose chemotherapy followed by autologous stem cell transplantation. Leukemia. 2006;20:1533–8. https://doi.org/10.1038/sj.leu.2404306.
Mercadal S, Briones J, Xicoy B, Escoda L, Estany C, Camós M, et al. Intensive chemotherapy (high-dose CHOP/ESHAP regimen) followed by autologous stem-cell transplantation in previously untreated patients with peripheral T-cell lymphoma. Ann Oncol. 2008;19:958–63. https://doi.org/10.1093/annonc/mdn022.
•• Reimer P, Rüdiger T, Geissinger E, Weissinger F, Nerl C, Schmitz N, et al. Autologous stem-cell transplantation as first-line therapy in peripheral T-cell lymphomas: results of a prospective multicentre study. J Clin Oncol. 2009;27:106–13. https://doi.org/10.1200/JCO.2008.17.4870Clinically study on the role of ASCT in first line for PTCL.
•• Wilhelm M, Smetak M, Reimer P, Geissinger E, Ruediger T, Metzner B, et al. First-line therapy of peripheral T-cell lymphoma: extension and long-term follow-up of a study investigating the role of autologous stem cell transplantation. Blood Cancer J. 2016;6:e452. https://doi.org/10.1038/bcj.2016.63Clinically study on the role of ASCT in first line for PTCL.
•• d’Amore F, Relander T, Lauritzsen GF, Jantunen E, Hagberg H, Anderson H, et al. Up-front autologous stem-cell transplantation in peripheral T-cell lymphoma: NLG-T-01. J Clin Oncol. 2012;30:3093–9. https://doi.org/10.1200/JCO.2011.40.2719Clinically study on the role of ASCT in first line for PTCL.
Park SI, Horwitz SM, Foss FM, Pinter-Brown LC, Carson KR, Rosen ST, et al. The role of autologous stem cell transplantation in patients with nodal peripheral T-cell lymphomas in first complete remission: report from COMPLETE, a prospective, multicenter cohort study. Cancer 2019;125:1507-1517. doi: https://doi.org/10.1002/cncr.31861.
Savage KJ, Horwitz SM, Advani RH, Christensen JH, Domingo-Domenech E, Rossi G, et al. An exploratory analysis of brentuximab vedotin plus CHP (A+CHP) in the frontline treatment of patients with CD30+ peripheral T-cell lymphomas (ECHELON-2): impact of consolidative stem cell transplant. Blood (ASH Annual Meeting Abstracts). 2019;134(suppl 1):464 abstr. https://doi.org/10.1182/blood-2019-122781.
d’Amore F, Gaulard P, Trümper L, Corradini P, W-S Kim LS, et al. ESMO Guidelines Committee. Peripheral T-cell lymphomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2015;26(Suppl 5):v108–15. https://doi.org/10.1093/annonc/mdv201.
El-Asmar J, Reljic T, Ayala E, Hamadani M, Nishihori T, Kumar A, et al. Efficacy of high-dose therapy and autologous hematopoietic cell transplantation in peripheral T cell lymphomas as front-line consolidation or in the relapsed/refractory setting: a systematic review/meta-analysis. Biol Blood Marrow Transplant. 2016;22:802–14. https://doi.org/10.1016/j.bbmt.2015.12.004.
Fossard G, Broussais F, Coelho I, Bailly S, Nicolas-Virelizier E, Toussaint E, et al. Role of up-front autologous stem-cell transplantation in peripheral T-cell lymphoma for patients in response after induction: an analysis of patients from LYSA centers. Ann Oncol. 2018;29:715–23. https://doi.org/10.1093/annonc/mdx787.
Yam C, Landsburg DJ, Nead KT, Lin X, Mato AR, Svoboda J, et al. Autologous stem cell transplantation in first complete remission may not extend progression-free survival in patients with peripheral T cell lymphomas. Am J Hematol. 2016;91:672–6. https://doi.org/10.1002/ajh.24372.
Tang T, Khoo LP, Lim C, Ham JS, Kim SJ, Hong H, et al. Outcomes of patients with peripheral T-cell lymphoma in first complete remission: data from three tertiary Asian cancer centers. Blood Cancer J. 2017;7:653. https://doi.org/10.1038/s41408-017-0030-y.
Teras LR, DeSantis CE, Cerhan JR, Morton LM, Jemal A, Flowers CR. US lymphoid malignancy statistics by World Health Organizationsubtypes. CA Cancer J Clin. 2016;66:443–59.
Epperla N, Hamadani M, Fenske TS, Costa LJ. Incidence and survival trends in mantle cell lymphoma. Br J Haematol. 2018;181(5):703–6. https://doi.org/10.1111/bjh.14699.
Ladha A, Zhao J, Epner EM, Pu JJ. Mantle cell lymphoma and its management: where are we now? Exp Hematol Oncol. 2019;8(2). https://doi.org/10.1186/s40164-019-0126-0.
Jain P, Wang M. Mantle cell lymphoma: 2019 update on the diagnosis, pathogenesis, prognostication, and management. Am J Hematol. 2019;94(6):710–25. https://doi.org/10.1002/ajh.25487.
Gerson JN, Handorf E, Villa D, Gerrie AS, Chapani P, Li S, et al. Survival outcomes of younger patients with mantle cell lymphoma treated in the rituximab era. J Clin Oncol. 2019;37(6):471–80.
Staton AD, Langston AA. Autologous stem cell transplant: still the standard for fit patients with mantle cell lymphoma. Clin Lymphoma Myeloma Leuk. 2017;17S:S96–9. https://doi.org/10.1016/j.clml.2017.02.021.
Hermine O, Hoster E, Walewski J, Bosly A, Stilgenbauer S, Thieblemont C, et al. Addition of high-dosecytarabine to immunochemotherapy before autologous stem-cell transplantation in patients aged 65 years or younger with mantle cell lymphoma (MCL younger): a randomised, open-label, phase 3 trial of the European mantle cell lymphoma network. Lancet. 2016;388(10044):565–75.
Armand P, Redd R, Bsat J, Mayuram S, Giardino A, Fisher DC, et al. A phase 2 study of rituximab-Bendamustine and rituximab-Cytarabine for transplant-eligible patients with mantle cell lymphoma. Br J Haematol. 2016;173(1):89–95.
•• Drandi D, Alcantara M, Benmaad I, Söhlbrandt A, Lhermitte L, Zaccaria G, et al. Droplet digital PCR quantification of mantle cell lymphoma follow-up samples from four prospective trials of the European MCL Network. Hemasphere. 2020;4(2):e347. https://doi.org/10.1097/HS9.0000000000000347The papaer describes the increasing investigation of MRD in MCL.
• Le Gouill S, Thieblemont C, Oberic L, Moreau A, Bouabdallah K, Dartigeas C, et al. Rituximab after autologous stem-cell transplantation in mantle-cell lymphoma. N Engl J Med. 2017;377(13):1250–60 This article reviews the of rituximab after ASCT in MCL.
Rule S. The modern approach to mantle cell lymphoma. Hematol Oncol. 2019;37:66–9. https://doi.org/10.1002/hon.2596.
Vose JM. Mantle cell lymphoma: 2017 update on diagnosis, risk-stratification, and clinical management. Am J Hematol. 2017;92:806–13. https://doi.org/10.1002/ajh.24797.
Le Gouill S, Thieblemont C, Oberic L, Moreau A, Bouabdallah K, Gyan E, et al. Rituximab maintenance after autologous stem cell transplantation prolongs survival in younger patients with mantle cell lymphoma: final results of the randomized phase 3 LyMa trial of the LYSA/Goelams group. Blood. 2016;128:145.
Romaguera JE, Fayad L, Rodriguez MA, Broglio KR, Hagemeister FB, Pro B, et al. High rate of durable remissions after treatment of newly diagnosed aggressive mantle-cell lymphoma with rituximab plus hyper-CVAD alternating with rituximab plus high dose methotrexate and cytarabine. J Clin Oncol. 2005;23(28):7013–23.
Hoster E, Rosenwald A, Berger F, Bernd HW, Hartmann S, Loddenkemper C, et al. Prognostic value of Ki-67 index, cytology, and growth pattern in mantle cell lymphoma: results from randomized trials of the European Mantle Cell Lymphoma Network. J Clin Oncol. 2016;34(12):1386–94.
Bernstein S, Epner E, Unger JM, Leblanc M, Cebula E, Burack R, et al. A phase II multicenter trial of hyperCVAD MTX/Ara-C and rituximab in patients with previously untreated mantle cell lymphoma: SWOG 0213. Ann Oncol. 2013;24:1587–93.
Dreyling M, Lenz G, Hoster E, Van Hoof A, Gisselbrecht C, Schmits R, et al. Early consolidation by myeloablative radiochemotherapy followed by autologous stem cell transplantation in first remission significantly prolongs progression-free survival in mantle-cell lymphoma: results of a prospective randomized trial of the European MCL Network. Blood. 2005;105:2677–84.
Geisler CH, Kolstad A, Laurell A, Andersen NS, Pedersen LB, Jerkeman M, et al. Long-term progression-free survival of MCL after intensive front-line immunochemotherapy with in vivo-purged stem cell rescue: a randomized phase 2 multicenter study by the Nordic Lymphoma Group. Blood. 2008;112:2687–93.
• Eskelund CW, Gronbaek K, Hansen JW, Westman M, Kolstad A, Pedersen LB, et al. TP53 mutations identify younger mantle cell lymphoma patients who do not benefit from intensive chemoimmunotherapy. Blood. 2017;130:1903–10 The article suggests that patients with MCL should be stratified according to TP53 status.
Wang ML, Rule S, Martin P, Goy A, Auer R, Kahl BS, et al. Targeting BTK with ibrutinib in relapsed or refractory mantle-cell lymphoma. N Engl J Med. 2013;369:507–16.
• Wang M, Rule S, Zinzani PL, Goy A, Casasnovas O, Smith SD, et al. Acalabrutinib in relapsed or refractory mantle cell lymphoma (ACE-LY-004): a single-arm, multicentre, phase 2 trial. Lancet. 2018;391:659–67 The paper reports on the pahase II study suggesting an important role for acalabrutinib in the treatment of MCL.
Cheah CY, Seymour JF, Wang ML. Mantle cell lymphoma. J Clin Oncol. 2016;34:1256–69.
Martin P, Maddocks K, Leonard JP, Ruan J, Goy A, Wagner-Johnston N, et al. Postibrutinib outcomes in patients with mantle cell lymphoma. Blood. 2016;127:1559–63.
Jain P, Kanagal-Shamanna R, Zhang S, Ahmed M, Ghorab A, Zhang L, et al. Long-term outcomes and mutation profiling of patients with mantle cell lymphoma (MCL) who discontinued ibrutinib. Br J Haematol. 2018;183:578–87.
Epperla N, Hamadani M, Cashen AF, Ahn KW, Oak E, Kanate AS, et al. Predictive factors and outcomes for ibrutinib therapy in relapsed/refractory mantle cell lymphoma—a “real world” study. Hematol Oncol. 2017;35:528–35.
• Wang M, Munoz J, Goy A, Locke FL, Jacobson CA, Hill BT, et al. KTE-X19 CAR T-cell therapy in relapsed or refractory mantle-cell lymphoma. N Engl J Med. 2020;382:1331–42. https://doi.org/10.1056/NEJMoa1914347The paper describes the remarkable clinical outcome of anti-CD19 chimeric antigen receptor T cells in treating MCL.
• Stirrups R. CAR T-cell therapy for relapsed or refractory mantle-cell lymphoma. Lancet Oncol. 2020;21:e239. https://doi.org/10.1016/S1470-2045(20)30231-XThe paper comments on the especially impressive results of CAR-T cell therapy study for MCL.
• Jain P, Zhao S, Kanagal-Shamanna R, Navsaria L, Ok CY, Zhang S, et al. Efficacy of venetoclax in high risk relapsed mantle cell lymphoma (MCL)-outcomes and mutation profile from venetoclax resistant MCL patients. Am J Hematol. 2020. https://doi.org/10.1002/ajh.25796Study indicating that venetoclax resistance in MCL is predominantly associated with non-BCL2 gene mutations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Alessandro Broccoli, Lisa Argnani, and Pier Luigi Zinzani declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on B Cell NHL, T Cell NHL, and Hodgkin Lymphoma
Rights and permissions
About this article
Cite this article
Broccoli, A., Argnani, L. & Zinzani, P.L. Invited Review: Will Consolidation with ASCT Be a Thing of the Past for MCL and PTCL?. Curr Hematol Malig Rep 16, 82–88 (2021). https://doi.org/10.1007/s11899-021-00609-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-021-00609-5