Abstract
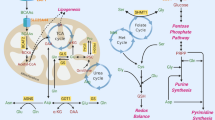
Cancer metabolism is characterized by increased macromolecular syntheses through coordinated increases in energy and substrate metabolism. The observation that cancer cells produce lactate in an environment of oxygen sufficiency (aerobic glycolysis) is a central theme of cancer metabolism known as the Warburg effect. Aerobic glycolysis in cancer metabolism is accompanied by increased pentose cycle and anaplerotic activities producing energy and substrates for macromolecular synthesis. How these processes are coordinated is poorly understood. Recent advances have focused on molecular regulation of cancer metabolism by oncogenes and tumor suppressor genes which regulate numerous enzymatic steps of central glucose metabolism. In the past decade, new insights in cancer metabolism have emerged through the application of stable isotopes particularly from 13C carbon tracing. Such studies have provided new evidence for system-wide changes in cancer metabolism in response to chemotherapy. Interestingly, experiments using metabolic inhibitors on individual biochemical pathways all demonstrate similar system-wide effects on cancer metabolism as in targeted therapies. Since biochemical reactions in the Warburg effect place competing demands on available precursors, high energy phosphates and reducing equivalents, the cancer metabolic system must fulfill the condition of balance of flux (homeostasis). In this review, the functions of the pentose cycle and of the tricarboxylic acid (TCA) cycle in cancer metabolism are analyzed from the balance of flux point of view. Anticancer treatments that target molecular signaling pathways or inhibit metabolism alter the invasive or proliferative behavior of the cancer cells by their effects on the balance of flux (homeostasis) of the cancer metabolic phenotype.






Similar content being viewed by others
Notes
In a cellular system, biochemical reactions are constrained by physical and chemical factors such as compartmentalization or tunneling; Km and Vmax of Michaelis–Menten kinetics; cofactor concentrations, and conservation of mass and energy (balance of flux).
The concept of balance of flux is applicable to reactions in one functional or physical compartment, as well as in all the compartments of the cellular metabolic network as a whole.
Glucose is the ultimate source of energy and carbon for amino acids and fatty acid synthesis. As a general rule in metabolism, energy is conserved (stored) in the form of fatty acids, and carbon source is conserved or regenerated through anaplerosis. Thus, glucose is generally not used fot ATP production, which is consistent with the occurrence of hypoglycemia in fatty acid oxidation abnormality and the development of diabetes under high fat dietary condition.
Futile cycle refers to a pair of reversible reactions in which the net result is the consumption of ATP or NADPH. A typical example of a pair of reactions is the combined action of glucokinase and glucose-6-phosphatase. It should be noted that these reactions do not take place contemporaneously, and reaction in either direction potentially affects its “downstream” reactions.
[1, 2-13C2]-OAA is converted to [1, 2-13C2]-glutamate and [3, 4-13C2]-OAA to [3-13C]-glutamate. The carbon 2–5 fragment has only singly labeled mass isotopomer from recycling.
The relation between m1/m2 ratio (r) and relative anaplerotic flux (Y) is given by the equation:
$${\text{Y}} = \left( { 1- {\text{r}}} \right)/{\text{r}}$$
References
Boren, J., et al. (2001). Gleevec (STI571) influences metabolic enzyme activities and glucose carbon flow toward nucleic acid and fatty acid synthesis in myeloid tumor cells. Journal of Biological Chemistry, 276, 37747–37753.
Boren, J., et al. (2003). The stable isotope-based dynamic metabolic profile of butyrate-induced HT29 cell differentiation. Journal of Biological Chemistry, 278, 28395–28402. doi:10.1074/jbc.M302932200.
Boros, L. G., Torday, J. S., Lim, S., Bassilian, S., Cascante, M., & Lee, W. N. (2000). Transforming growth factor beta2 promotes glucose carbon incorporation into nucleic acid ribose through the nonoxidative pentose cycle in lung epithelial carcinoma cells. Cancer Research, 60, 1183–1185.
Boros, L. G., Bassilian, S., Lim, S., & Lee, W. N. (2001). Genistein inhibits nonoxidative ribose synthesis in MIA pancreatic adenocarcinoma cells: a new mechanism of controlling tumor growth. Pancreas, 22, 1–7.
Boros, L. G., Cascante, M., & Lee, W. N. (2002a). Metabolic profiling of cell growth and death in cancer: Applications in drug discovery. Drug Discovery Today, 7, 364–372.
Boros, L. G., Lee, W. N., & Go, V. L. (2002b). A metabolic hypothesis of cell growth and death in pancreatic cancer. Pancreas, 24, 26–33.
Boros, L. G., Torday, J. S., Lee, W. N., & Rehan, V. K. (2002c). Oxygen-induced metabolic changes and transdifferentiation in immature fetal rat lung lipofibroblasts. Molecular Genetics and Metabolism, 77, 230–236.
Boros, L. G., Steinkamp, M. P., Fleming, J. C., Lee, W. N., Cascante, M., & Neufeld, E. J. (2003). Defective RNA ribose synthesis in fibroblasts from patients with thiamine-responsive megaloblastic anemia (TRMA). Blood, 102, 3556–3561. doi:10.1182/blood-2003-05-1537.
Chaturvedi, A., et al. (2013). Mutant IDH1 promotes leukemogenesis in vivo and can be specifically targeted in human AML. Blood, 122, 2877–2887. doi:10.1182/blood-2013-03-491571.
Chen, C., et al. (2013). Cancer-associated IDH2 mutants drive an acute myeloid leukemia that is susceptible to Brd4 inhibition. Genes & Development, 27, 1974–1985. doi:10.1101/gad.226613.113.
Comin-Anduix, B., et al. (2001). The effect of thiamine supplementation on tumour proliferation. A metabolic control analysis study. European Journal of Biochemistry, 268, 4177–4182.
D’Alessandro, A., & Zolla, L. (2012). Metabolomics and cancer drug discovery: Let the cells do the talking. Drug Discovery Today, 17, 3–9. doi:10.1016/j.drudis.2011.09.017.
Edwards, J. S., Ramakrishna, R., & Palsson, B. O. (2002). Characterizing the metabolic phenotype: A phenotype phase plane analysis. Biotechnology and Bioengineering, 77, 27–36.
Faubert, B., et al. (2013). AMPK is a negative regulator of the Warburg effect and suppresses tumor growth in vivo. Cell Metabolism, 17, 113–124. doi:10.1016/j.cmet.2012.12.001.
Feng, Z., & Levine, A. J. (2010). The regulation of energy metabolism and the IGF-1/mTOR pathways by the p53 protein. Trends in Cell Biology, 20, 427–434. doi:10.1016/j.tcb.2010.03.004.
Ferreira, L. M. (2010). Cancer metabolism: the Warburg effect today. Experimental and Molecular Pathology, 89, 372–380. doi:10.1016/j.yexmp.2010.08.006.
Harris, D. M., Li, L., Chen, M., Lagunero, F. T., Go, V. L., & Boros, L. G. (2012). Diverse mechanisms of growth inhibition by luteolin, resveratrol, and quercetin in MIA PaCa-2 cells: A comparative glucose tracer study with the fatty acid synthase inhibitor C75. Metabolomics, 8, 201–210. doi:10.1007/s11306-011-0300-9.
Katz, J., & McGarry, J. D. (1984). The glucose paradox. Is glucose a substrate for liver metabolism? Journal of Clinical Investigation, 74, 1901–1909. doi:10.1172/jci111610.
Katz, J., Lee, W. N., Wals, P. A., & Bergner, E. A. (1989). Studies of glycogen synthesis and the Krebs cycle by mass isotopomer analysis with [U-13C]glucose in rats. Journal of Biological Chemistry, 264, 12994–13004.
Katz, J., Wals, P., & Lee, W. N. (1993). Isotopomer studies of gluconeogenesis and the Krebs cycle with 13C-labeled lactate. Journal of Biological Chemistry, 268, 25509–25521.
Kim, J.-W., Gardner, L. B., & Dang, C. V. (2005). Oncogenic alterations of metabolism and the Warburg effect. Drug Discovery Today: Disease Mechanisms, 2, 233–238. doi:10.1016/j.ddmec.2005.04.001.
Kurland, I. J., Alcivar, A., Bassilian, S., & Lee, W. N. (2000). Loss of [13C]glycerol carbon via the pentose cycle. Implications for gluconeogenesis measurement by mass isotoper distribution analysis. Journal of Biological Chemistry, 275, 36787–36793. doi:10.1074/jbc.M004739200.
Lee, W. N. (1993). Appendix. Analysis of tricarboxylic acid cycle using mass isotopomer ratios. Journal of Biological Chemistry, 268, 25522–25526.
Lee, W. N. (2006). Characterizing phenotype with tracer based metabolomics. Metabolomics, 2, 31–39. doi:10.1007/s11306-006-0017-3.
Lee, W. N., Boros, L. G., Puigjaner, J., Bassilian, S., Lim, S., & Cascante, M. (1998). Mass isotopomer study of the nonoxidative pathways of the pentose cycle with [1,2-13C2]glucose. American Journal of Physiology, 274, E843–E851.
Lee, W. N., Edmond, J., Bassilian, S., & Morrow, J. W. (1996). Mass isotopomer study of glutamine oxidation and synthesis in primary culture of astrocytes. Developmental Neuroscience, 18, 469–477.
Lee, W. N., & Go, V. L. (2005). Nutrient-gene interaction: Tracer-based metabolomics. Journal of Nutrition, 135, 3027S–3032S.
Lee, W. N., Guo, P., Lim, S., Bassilian, S., Lee, S. T., Boren, J., et al. (2004). Metabolic sensitivity of pancreatic tumour cell apoptosis to glycogen phosphorylase inhibitor treatment. British Journal of Cancer, 91, 2094–2100. doi:10.1038/sj.bjc.6602243.
Lee, W. N., Wahjudi, P. N., Xu, J., & Go, V. L. (2010). Tracer-based metabolomics: concepts and practices. Clinical Biochemistry, 43, 1269–1277. doi:10.1016/j.clinbiochem.2010.07.027.
Levine, A. J., & Puzio-Kuter, A. M. (2010). The control of the metabolic switch in cancers by oncogenes and tumor suppressor genes. Science, 330, 1340–1344. doi:10.1126/science.1193494.
Lu, Q.-Y., Zhang, L., Yee, J., Go, V.-L., & Lee, W.-N. (2014). Metabolic consequences of LDHA inhibition by epigallocatechin gallate and oxamate in MIA PaCa-2 pancreatic cancer cells. Metabolomics. doi:10.1007/s11306-014-0672-8.
Ma, D., et al. (2012). Inhibition of glycogen phosphorylation induces changes in cellular proteome and signaling pathways in MIA pancreatic cancer cells. Pancreas, 41, 397–408. doi:10.1097/MPA.0b013e318236f022.
Malloy, C. R., Sherry, A. D., & Jeffrey, F. M. (1990). Analysis of tricarboxylic acid cycle of the heart using 13C isotope isomers. American Journal of Physiology, 259, H987–H995.
Marin, S., et al. (2004). Dynamic profiling of the glucose metabolic network in fasted rat hepatocytes using [1,2-13C2]glucose. Biochemical Journal, 381, 287–294. doi:10.1042/bj20031737.
Orth, J. D., Thiele, I., & Palsson, B. O. (2010). What is flux balance analysis? Nature Biotechnology, 28, 245–248. doi:10.1038/nbt.1614.
Rais, B., et al. (1999). Oxythiamine and dehydroepiandrosterone induce a G1 phase cycle arrest in Ehrlich’s tumor cells through inhibition of the pentose cycle. FEBS Letters, 456, 113–118.
Ramos-Montoya, A., et al. (2006). Pentose phosphate cycle oxidative and nonoxidative balance: A new vulnerable target for overcoming drug resistance in cancer. International Journal of Cancer, 119, 2733–2741. doi:10.1002/ijc.22227.
Reed, J. L., & Palsson, B. O. (2003). Thirteen years of building constraint-based in silico models of Escherichia coli. Journal of Bacteriology, 185, 2692–2699.
Rehan, V. K., et al. (2007). In utero nicotine exposure alters fetal rat lung alveolar type II cell proliferation, differentiation, and metabolism. American Journal of Physiology. Lung Cellular and Molecular Physiology, 292, L323–L333. doi:10.1152/ajplung.00071.2006.
Reitman, Z. J., et al. (2014). Cancer-associated isocitrate dehydrogenase 1 (IDH1) R132H mutation and d-2-hydroxyglutarate stimulate glutamine metabolism under hypoxia. Journal of Biological Chemistry, 289, 23318–23328. doi:10.1074/jbc.M114.575183.
Schilling, C. H., & Palsson, B. O. (1998). The underlying pathway structure of biochemical reaction networks. Proceedings of the National Academy of Science USA, 95, 4193–4198.
Schilling, C. H., Letscher, D., & Palsson, B. O. (2000). Theory for the systemic definition of metabolic pathways and their use in interpreting metabolic function from a pathway-oriented perspective. Journal of Theoretical Biology, 203, 229–248. doi:10.1006/jtbi.2000.1073.
Selivanov, V. A., et al. (2004). An optimized algorithm for flux estimation from isotopomer distribution in glucose metabolites. Bioinformatics, 20, 3387–3397. doi:10.1093/bioinformatics/bth412.
Selivanov, V. A., et al. (2005). Rapid simulation and analysis of isotopomer distributions using constraints based on enzyme mechanisms: An example from HT29 cancer cells. Bioinformatics, 21, 3558–3564. doi:10.1093/bioinformatics/bti573.
Turcan, S., et al. (2013). Efficient induction of differentiation and growth inhibition in IDH1 mutant glioma cells by the DNMT Inhibitor Decitabine. Oncotarget, 4, 1729–1736.
Upadhyay, M., Samal, J., Kandpal, M., Singh, O. V., & Vivekanandan, P. (2013). The Warburg effect: Insights from the past decade. Pharmacology & Therapeutics, 137, 318–330. doi:10.1016/j.pharmthera.2012.11.003.
Vaitheesvaran, B., et al. (2014). Role of the tumor suppressor IQGAP2 in metabolic homeostasis: Possible link between diabetes and cancer. Metabolomics, 10, 920–937. doi:10.1007/s11306-014-0639-9.
Vizan, P., et al. (2005). K-ras codon-specific mutations produce distinctive metabolic phenotypes in NIH3T3 mice [corrected] fibroblasts. Cancer Research, 65, 5512–5515. doi:10.1158/0008-5472.can-05-0074.
Wang, J., et al. (2013). Inhibition of transketolase by oxythiamine altered dynamics of protein signals in pancreatic cancer cells. Experimental Hematology & Oncology, 2, 18. doi:10.1186/2162-3619-2-18.
Xu, J., Lee, W. N., Phan, J., Saad, M. F., Reue, K., & Kurland, I. J. (2006). Lipin deficiency impairs diurnal metabolic fuel switching. Diabetes, 55, 3429–3438. doi:10.2337/db06-0260.
Yang, Y., et al. (2013). Metabolic reprogramming for producing energy and reducing power in fumarate hydratase null cells from hereditary leiomyomatosis renal cell carcinoma. PLoS ONE, 8, e72179. doi:10.1371/journal.pone.0072179.
Zhang, H., et al. (2010). Inhibition of protein phosphorylation in MIA pancreatic cancer cells: Confluence of metabolic and signaling pathways. Journal of Proteome Research, 9, 980–989. doi:10.1021/pr9008805.
Zheng, J. (2012). Energy metabolism of cancer: Glycolysis versus oxidative phosphorylation (Review). Oncology Letters, 4, 1151–1157. doi:10.3892/ol.2012.928.
Acknowledgments
This work was supported by the National Institutes of Health (P01AT003960) and the Hirshberg Foundation for Pancreatic Cancer Research, V.B.P was supported by DK58132-01A2 and NIAID Grant U19AI091175-01.
Conflict of interest
The authors have no conflicts of interest to disclose.
Compliance with Ethical Requirements
This article does not contain any studies with human or animal subjects.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.

11306_2014_760_MOESM1_ESM.jpg
Supplemental Figure S1: Glycolytic/gluconeogenic pathways and their regulation. The regulation of glycolysis is accomplished through a series of futile cycles (shown as double arrows) and reversible reactions (shown as single bidirectional arrows). The direction of flux is regulated by multiple connected pathways. In addition, it is controlled by expression and/or activation of enzymes. Since glucose is needed for numerous substrate syntheses, multiple regulatory points (double arrows) are necessary to achieve fine control of glucose flux and its directions. HEX stands for hexokinase; G6Pase, glucose-6-phosphatase; GS glycogen synthase; GP, glycogenphosphorylase; PFK, phosphofructose kinase; GAPDH, glyceraldehyde 3 phosphate dehydrogenase; PGK, phosphoglycerate kinase; PGM, phosphoglycerate mutase; ENO, enolase; PKM, pyruvate kinase isozymes M1/M2 and LDH, lactate dehydrogenase. These key control points are regulated by oncogenes and cancer suppressor genes (D’Alessandro and Zolla 2012). (JPEG 228 kb)

11306_2014_760_MOESM2_ESM.jpg
Supplemental Figure S2: Balance of flux equations for anaplerotic reactions. These reactions allow communication (substrate exchange) between cytosolic and mitochondrial compartments. Not included in the OAA or malate balance are equations for transamination, PEPCK, maleic reaction. Together with reactions from Figure 5, these reactions form the basis for the balance of flux model of TCA cycle compartment. (JPEG 332 kb)
Rights and permissions
About this article
Cite this article
Vaitheesvaran, B., Xu, J., Yee, J. et al. The Warburg effect: a balance of flux analysis. Metabolomics 11, 787–796 (2015). https://doi.org/10.1007/s11306-014-0760-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11306-014-0760-9