Abstract
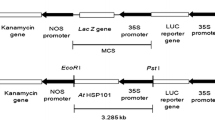
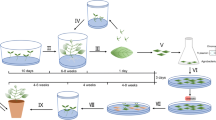
A reliable Agrobacterium-mediated transformation and shoot regeneration protocol was developed for breeding lines of commercially important western-shipper cantaloupe and honeydew melons, ‘F39’ and ‘150’, respectively. Different media were tested to select a shoot regeneration system for each of three elite breeding lines ‘F39’, ‘141’ and ‘TMS’. Murashige & Skoog (MS) basal medium supplemented with 1 mg l−1 benzyladenine (BA), 0.26 mg l−1 abscisic acid (ABA) and 0.8 mg l−1 indole-3-acetic acid (IAA) was used for shoot regeneration from cotyledonary explants in ‘F39’ and ‘150’. Kanamycin sensitivity as well as Timentin™ and Clavamox® were evaluated using wild-type ‘F39’ and ‘150’ cotyledons. Kanamycin concentrations of 200 and 150 mg l−1 were chosen as the threshold levels for ‘F39’ and ‘150’, respectively. No significant differences were found between Timentin™ and Clavamox® in ‘F39’; however, Clavamox® reduced the incidence of vitrification and increased the frequency of shoot elongation in ‘150’. A. tumefaciens strain EHA105, harboring pCNL56 carrying neomycin phosphotransferase II (nptII) and gusA reporter genes, was selected to establish a transformation protocol for ‘F39’ and ‘150’. Putative transformants were evaluated using β-glucuronidase (GUS) histochemical assay, polymerase chain reaction (PCR) and Southern blot analyses. Based on these parameters, the transformation efficiency for cantaloupe ‘F39’ was 0.3% and that for honeydew ‘150’ was 0.5%.








Similar content being viewed by others
Abbreviations
- BA:
-
6-Benzyladenine
- IAA:
-
Indole-3-acetic acid
- ABA:
-
Abscisic acid
- nptII :
-
Neomycin phosphotransferase II
- GUS:
-
β-glucuronidase
- MS:
-
Murashige and Skoog
- PCR:
-
Polymerase chain reaction
References
Afroz A, Chaudhry Z, Rashid U, Ali GM, Nazir F, Iqbal J, Khan MR (2010) Enhanced resistance against bacterial wilt in transgenic tomato (Lycopersicon esculentum) lines expressing the Xa21 gene. Plant Cell Tissue Organ Cult 104:227–237
Akasaka-Kennedy Y, Tomita K, Ezura H (2004) Efficient plant regeneration and Agrobacterium-mediated transformation via somatic embryogenesis in melon (Cucumis melo L.). Plant Sci 166:763–769
Ayub R, Guis M, Amor MB, Gillot L, Roustan JP, Latché A, Bouzayen M, Pech JC (1996) Expression of ACC oxidase antisense gene inhibits ripening of cantaloupe melon fruits. Nat Biotechnol 14:862–866
Bordas M, Montesinos C, Dabauza M, Salvador A, Roig LA, Serrano R, Moreno V (1997) Transfer of the yeast salt tolerance gene HAL1 to Cucumis melo L. cultivars and in vitro evaluation of salt tolerance. Trans Res 6:41–50
Castelblanque L, Marfa V, Claveria E, Martinez I, Perez-Grau L, Dolcet-Sanjuan R (2008) Improving the genetic transformation efficiency of Cucumis melo subsp. melo ‘Piel de Sapo’ via Agrobacterium. Cucurbitaceae 2008. In: Pitrat M (ed) Proc IXth EUCARPIA Mtg on Gene and Breed of Cucurbitaceae, INRA, Avignon, France, 21–24 May 2008, pp 627–631
Cheng ZM, Schnurr JA, Kapaun JA (1998) Timentin as an alternative antibiotic for suppression of Agrobacterium tumefaciens in genetic transformation. Plant Cell Rep 17:646–649
Chovelon V, Restier V, Dogimont C, Aarrouf J (2008) Histological study of shoot organogenesis in melon (Cucumis melo L.) after genetic transformation. Cucurbitaceae 2008. In: Pitrat M (ed) Proc IXth EUCARPIA mtg on Gene and Breed of Cucurbitaceae, INRA, Avignon, France, 21–24 May 2008, pp 633–637
Clough GH, Hamm PB (1995) Coat protein transgenic resistance to watermelon mosaic and zucchini yellows mosaic-virus in squash and cantaloupe. Plant Dis 79:1107–1109
Compton ME, Gray DJ, Gaba VP (2004) Genetic transformation of watermelon. In: Curtis IS (ed) Transgenic crops of the world-essential protocols. Kluwer Academic Publishers, Dordrecht, pp 425–433
Costa MS, Miguel C, Oliveira MM (2006) An improved selection strategy and the use of acetosyringone in shoot induction medium increase almond transformation efficiency by 100-fold. Plant Cell Tissue Organ Cult 85:205–209
Dan Y, Charles L, Armstrong Dong J, Feng X, Fry JE, Keithly GE, Martinell BJ, Roberts GA, Smith LA, Tan LJ, Duncan DR (2009) Lipoic acid—a unique plant transformation enhancer. In Vitro Cell Dev Biol Plant 45:630–638
Dong JZ, Yang MZ, Jia SR, Chua NH (1991) Transformation of melon (Cucumis melo L.) and expression from the cauliflower mosaic virus-35 s promoter in transgenic melon plants. Biotechnology 9:858–863
Dutt M, Vasconcellos M, Grosser JW (2011) Effects of antioxidants on Agrobacterium-mediated transformation and accelerated production of transgenic plants of Mexican lime (Citrus aurantifolia Swingle). Plant Cell Tissue Organ Cult Online First™, 6 May 2011
Fan JD, He QW, Wang XF, Yu XY (2007) Antisense acid Invertase (anti-MAI1) gene alters soluble sugar composition and size in transgenic muskmelon fruits. Acta Hort Sinica 34:677–682
Fang G, Grumet R (1990) Agrobacterium tumefaciens mediated transformation and regeneration of muskmelon plants. Plant Cell Rep 9:160–164
Ficcadenti N, Rotino GL (1995) Genotype and medium affect shoot regeneration of melon. Plant Cell Tissue Organ Cult 40:293–295
Fuchs M, McFerson JR, Tricoli DM, McMaster JR, Deng RZ, Boeshore ML, Reynolds JF, Russell PF, Quemada HD, Gonsalves D (1997) Cantaloupe line CZW-30 containing coat protein genes of cucumber mosaic virus, zucchini yellow mosaic virus, and watermelon mosaic virus-2 is resistant to these three viruses in the field. Mol Breed 3:279–290
Fullner KJ, Nester EW (1996) Temperature affects the T-DNA transfer machinery of Agrobacterium tumefaciens. J Bacteriol 178:1498–1504
Gaba V, Elman C, Watad AA, Gray DJ (1996) Ancymidol hastens in vitro bud development in melon. HortScience 31:1223–1224
Galperin M, Patlis L, Ovadia A, Wolf D, Zelcer A, Kenigsbuch D (2003) A melon genotype with superior competence for regeneration and transformation. Plant Breed 122:66–69
Gonsalves C, Xue B, Yepes M, Fuchs M, Ling KS, Namba S, Chee P, Slightom JL, Gonsalves D (1994) Transferring cucumber mosaic virus-white leaf strain coat protein gene into Cucumis melo L. and evaluating transgenic plants for protection against infections. J Am Soc Hort Sci 119:345–355
Gould JH, Magallens-Cedeno M (1998) Adaptation of cotton shoot apex culture to Agrobacterium-mediated transformation. Plant Mol Biol Rep 16:1–10
Gray DJ, McColley DW, Compton ME (1993) High-frequency somatic embryogenesis from quiescent seed cotyledons of Cucumis melo cultivars. J Am Soc Hort Sci 118:425–432
Guis M, Latché A, Pech JC, Roustan JP (1997) An efficient method for production of diploid cantaloupe charentais melon (Cumcumis melo L. var cantalupensis) by somatic embryogenesis. Scientia Hort 69:199–206
Guis M, Amor MB, Latché A, Pech JC, Roustan JP (2000) A reliable system for the transformation of cantaloupe charentais melon (Cucumis melo L. var. cantalupensis) leading to a majority of diploid regenerants. Scientia Hort 84:91–99
Hao J, Niu Y, Yang B, Gao F, Zhang L, Wang J, Hasi A (2011) Transformation of a marker-free and vector-free antisense ACC oxidase gene cassette into melon via the pollen-tube pathway. Biotechnol Lett 33:55–61
Hoekema A, Hirsch PR, Hooykaas PJJ, Schilperoort RA (1983) A binary plant vector strategy based on separation of vir- and T-region of the Agrobacterium tumefaciens Ti-plasmid. Nature 303:179–180
Hood EE, Gelvin SB, Melchers LS, Hoekema A (1993) New Agrobacterium helper plasmids for gene transfer to plant. Transgenic Res 2:208–218
Hussain AF, Anfoka GH, Hassawi DS (2008) Transformation of tomato with TYLCV gene silencing construct using optimized Agrobacterium-mediated protocol. Biotechnology 7:537–543
Ieamkhang S, Chatchawankanphanich O (2005) Augmentin® as an alternative antibiotic for growth suppression of Agrobacterium for tomato (Lycopersicon esculentum) transformation. Plant Cell Tissue Organ Cult 82:213–220
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: β-glucuonidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 13:3901–3907
Kathiravan K, Vengedesan G, Singer S, Steinitz B, Paris HS, Gaba V (2006) Adventitious regeneration in vitro occurs across a wide spectrum of squash (Cucurbita pepo) genotypes. Plant Cell Tissue Organ Cult 85:285–295
Keng CL, Hoong LK (2005) In vitro plantlets regeneration from nodal segments of muskmelon (Cucumis melo L.). Biotechnology 4:354–357
Kintzios SE, Taravira N (1997) Effect of genotype and light intensity on somatic embryogenesis and plant regeneration in melon (Cucumis melo L.). Plant Breed 116:359–362
Kintzios S, Stavropoulou Er, Skamneli S (2004) Accumulation of selected macronutrients and carbohydrates in melon tissue cultures: association with pathways of in vitro dedifferentiation and differentiation (organogenesis, somatic embryogenesis). Plant Sci 167:655–664
Kumar V, Campbell LM, Rathore KS (2011) Rapid recovery- and characterization of transformants following Agrobacterium-mediated T-DNA transfer to sorghum. Plant Cell Tissue Organ Cult 104:137–146
Larkin PJ, Scowcroft WR (1981) Somaclonal variation—a novel source of variability from cell cultures for plant improvement. Theor Appl Genet 60:197–214
Li X, Liu C, Ritchie SW, Peng J, Gelvin SB, Hodges TK (1992) Factors influencing Agrobacterium-mediated transient expression of gusA in rice. Plant Mol Biol 20:1037–1048
Mollel MHN, Goyvaerts EMA (2004) Preliminary examination of factors affecting Agrobacterium tumefaciens-mediated transformation of marula, Sclerocarya birrea subsp. caffra (Anacardiaceae). Plant Cell Tissue Organ Cult 79:321–328
Moore GA (1995) Phenotypic stability of transgenic citrus. HortScience 30:903–904 (Abstract)
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nakagawa H, Saijyo T, Yamauchi N, Shigyo M, Kako S, Ito A (2001) Effects of sugars and abscisic acid on somatic embryogenesis from melon (Cucumis melo L.) expanded cotyledon. Scientia Horti 90:85–92
Nuňez-Palenius HG, Cantliffe DJ, Huber DJ, Ciardi J, Klee HJ (2006) Transformation of a muskmelon ‘Galia’ hybrid parental line (Cucumis melo L. var. reticulatus Ser.) with an antisense ACC oxidase gene. Plant Cell Rep 25:198–205
Nuňez-Palenius HG, Grumet R, Lester G, Cantliffe D (2008) Melon fruits: genetic diversity, physiology, and biotechnology features. Crit Rev Biotechnol 28:13–55
Olhoft PM, Somer DA (2001) l-Cysteine increases Agrobacterium-mediated T-DNA delivery into soybean cotyledonary-node cells. Plant Cell Rep 20:706–711
Oridate T, Oosawa K (1986) Somatic embryogenesis and plant regeneration from suspension callus culture in melon. Jpn J Breed 36:424–428
Oridate T, Atsumi H, Ito S, Araki H (1992) Genetic differences in somatic embryogenesis from seeds in melon (Cucumis melo L.). Plant Cell Tissue Organ Cult 29:27–30
Orts MC, Garciasogo B, Roche MV, Roig LA, Moreno V (1987) Morphogenetic response of calli derived from primary explants of diverse cultivars of melon. HortScience 22:666
Ostergaard L, Yanofsky M (2004) Establishing gene function by mutagenesis in Arabidopsis thaliana. Plant J 39:682–696
Seo MS, Takahashi S, Kadowaki K, Kawamukai M, Takahara M, Takamizo T (2011) Expression of CoQ10-producing ddsA transgene by efficient Agrobacterium-mediated transformation in Panicum meyerianum. Plant Cell Tissue Organ Cult Online First™ 10 June 2011
Sharma M, Kothari-Chajer A, Jagga-Chugh S, Kothari SL (2011) Factors influencing Agrobacterium tumefaciens-mediated genetic transformation of Eleusine coracana (L.) Gaertn. Plant Cell Tissue Organ Cult 105:93–104
Shilpa KS, Kumar VD, Sujatha M (2010) Agrobacterium-mediated genetic transformation of safflower (Carthamus tinctorius L.). Plant Cell Tissue Organ Cult 103:387–401
Skroch PW, Nienhuis J (1995) Qualitative and quantitative characterization of RAPD variation among snap bean genotypes. Theor Appl Genet 91:1078–1085
Slater SMH, KellerWA, Scoles G (2011) Agrobacterium-mediated transformation of Eruca sativa. Plant Cell Tissue Organ Cult Online First™ 5 January 2011
Souza FVD, Garcia-Sogo B, da Silva Souza A, San-Juán P, Moreno V (2006) Morphogenetic response of cotyledon and leaf explants of melon (Cucumis melo L.) cv. Amarillo Oro. Braz Arch Biol Technol 49:21–27
Thiruvengadam M, Hsu W, Yang C (2011) Phosphomannose-isomerase as a selectable marker to recover transgenic orchid plants (Oncidium Gower Ramsey). Plant Cell Tissue Organ Cult 104:239–246
Toldi O, Tóth S, Pónyi T, Scott P (2002) An effective and reproducible transformation protocol for the model resurrection plant Craterostigma plantagineum Hochst. Plant Cell Rep 211:63–69
Vallés MP, Lasa JM (1994) Agrobacterium-mediated transformation of commercial melon (Cucumis melo L. cv. Amarillo Oro). Plant Cell Rep 13:145–148
Wu HW, Yu TA, Raja JAJ, Wang HC, Yeh SD (2009) Generation of transgenic oriental melon resistant to Zucchini yellow mosaic virus by an improved cotyledon-cutting method. Plant Cell Rep 28:1053–1064
Yadav RC, Saleh MT, Grumet R (1996) High frequency shoot regeneration from leaf explants of muskmelon. Plant Cell Tissue Organ Cult 45:207–214
Yasmin A, Debener T (2010) Transient gene expression in rose petals via Agrobacterium infiltration. Plant Cell Tissue Organ Cult 102:245–250
Zambre M, Terryn N, De Clercq J, De Buck S, Dillen W, Van Montagu M, Van Der Straeten D, Angenon G (2003) Light strongly promotes gene transfer from Agrobacterium tumefaciens to plant cells. Planta 216:580–586
Zheng Q, Ju B, Liang L, Xiao X (2005) Effects of antioxidants on the plant regeneration and GUS expressive frequency of peanut (Arachis hypogaea) explants by Agrobacterium tumefaciens. Plant Cell Tissue Organ Cult 811:83–89
Acknowledgments
This research was supported by an USDA-CSREES Special Grant “Designing Foods for Health” (2006-34402-17121, 2008-34402-19195 and 2009-34402-19831) through the Vegetable and Fruit Improvement Center, Texas A&M University. We wish to thank Drs. Rebecca Grumet, Keerti Rathore, Sung Hun Park, Sylvain Marcel, Selvakumar Veluchamy, Mehdi Kabbage, Ms. Marianne Arnold, Sue Hammar, and Jungeun Kim for technical advice and help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ren, Y., Bang, H., Curtis, I.S. et al. Agrobacterium-mediated transformation and shoot regeneration in elite breeding lines of western shipper cantaloupe and honeydew melons (Cucumis melo L.). Plant Cell Tiss Organ Cult 108, 147–158 (2012). https://doi.org/10.1007/s11240-011-0024-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-011-0024-6