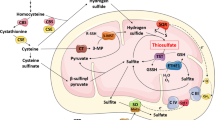
The available information on the mechanisms of involvement of vitamin B1 (thiamine) in vital processes in neural cells is analyzed. It is concluded that, together with the coenzymatic functions performed by this vitamin, the latter realizes “noncoenzymatic” functions. These functions play significant roles in cellular processes in various cells, and this is an indisputable fact. Some features of the structural/functional organization of neurons are responsible for the importance of thiamine-dependent processes for the maintenance of the functional activity of these cells. The accumulated data on the release of thiamine from neurons in the course of their excitation, as well as on the high dynamics of B1 metabolism in nerve cells related to rapid changes between states of excitation and inhibition, allow researchers to formulate an idea on the existence of a rapidly exchanged pool of thiamine ant its biologically active derivatives (“mobile thiamine pool”) in such cells. It is hypothesized that circulation of this pool between the main part of the intracellular space and presynaptic compartments of the synaptic structures is associated with changes in the membrane potential of nerve cells and modifications of cellular metabolism. This is confirmed by the data on coupling of the regulation of the mitochondrial multienzyme pyruvate dehydrogenase complex with the functioning of the excitable membranes. It is suggested that the interaction of thiamine with cytoskeletal proteins plays an important role in vitally important nonenzymatic processes realized by this vitamin in neural cells. The possible role of thiamine deficiency in the development of neurodegenerative diseases, such as Alzheimer’s disease, Parkinson’s disease, and Wernicke’s encephalopathy, is discussed.
Similar content being viewed by others
References
D. Lonsdale, “A review of the biochemistry, metabolism and clinical benefits of thiamin(e) and its derivatives,” Evid. Based Complement. Altern. Med., 3, No. 1, 49-59 (2006).
S. Manzetti, J. Zhang, and van der D. Spoel, “Thiamine function, metabolism, uptake, and transport,” Biochemistry, 53, No. 5, 821-835 (2014).
R. F. Butterworth, J. J. Kril, and C. G. Harper,“Thiamine-dependent enzyme changes in the brains of alcoholics: relationship to the Wernicke-Korsakoff syndrome,” Alcohol Clin. Exp. Res., 17, No. 5, 1084-1088 (1993).
K. V. Lu’o’ng and L. T. Nguyên, “Role of thiamine in Alzheimer’s disease,” Am. J. Alzheimers Dis. Other Demen., 26, No. 8, 588-598 (2011).
J. P. Blass, P. Gleason, D. Brush, et al., “Thiamine and Alzheimer’s disease. A pilot study,” Arch. Neurol., 45, No. 8, 833-835 (1988).
X. Pan, G. Fei, J. Lu, et al., “Measurement of blood thiamine metabolites for Alzheimer’s disease diagnosis,” EBioMedicine, 26, No. 3, 155-162 (2015).
K. V. Luong and L. T. Nguyên, “Thiamine and Parkinson’s disease,” J. Neurol. Sci., 316, Nos. 1/2, 1-8 (2012).
K. V. Luong and L. T. Nguyên, “The beneficial role of thiamine in Parkinson disease,” CNS Neurosci. Ther., 19, No. 7, 461-468 (2011).
A. Plaitakis, W. O. Whetsell, and M. D. Yahr, “Subacute necrotizing encephalomyelopathy (Leigh’s disease): clinical and genetic considerations of its adult form,” Trans. Am. Neurol. Ass., 102, No. 1, 32-35 (1977).
J. R. Cooper and J. H. Pincus, “Roles of thiamine triphosphate in subacute necrotizing encephalomyelopathy,” J. Agric. Food Chem., 20, No. 3, 490-493 (1972).
Yu. M. Ostrovsky, “On the mechanism of coenzymic and noncoenzymic action of thiamine,” J. Vitaminol., 14, Suppl., 98-102 (1968).
R. H. Haas, “Thiamine and the brain,” Annu. Rev. Nutr., 8, 483-515 (1988).
Yu. M. Parkhomenko, G. V. Donchenko, and Z. S. Protasova, “Neuroactivity of thiamine: Facts and hypotheses,” Ukr. Biokhim. Zh., 68, No. 2, 3-14 (1996).
L. Bettendorff, “A non-cofactor role of thiamine derivatives in excitable cells?” Arch. Physiol. Biochem., 104, No. 6, 745-751 (1996).
Y. Itokawa, “Thiamine and nervous system function: an historical sketch,” Metab. Brain Dis., 11, No. 1, 1-7 (1996).
A. Bâ, “Metabolic and structural role of thiamine in nervous tissues,” Cell. Mol. Neurobiol., 28, No. 7, 923-931 (2008).
I. Bubko, B. M. Gruber, and E. L. Anuszewska, “The role of thiamine in neurodegenerative diseases,” Postepy Hig. Med. Dosw. (Online) [in Polish], 21, No. 69, 1096-1106 (2015).
L. Binet and B. Minz, “Sur les reactions biochimiques des nerf au repos et au cours d’une exitation electrique,” Arch. Int. Physiol., 42, 281-300 (1936).
B. Minz, “Sur la liberation de la vitamin B1 par le trone isole de nerf pneumogastrique soumis a l’exitation electrique,” Comp. Rend. Soc. Biol., 127, No. 6, 1251-1253 (1938).
H. A. Kunz, “Effect of aneurin antimetabolites on a single labeled nerve fiber,” Helv. Physiol. Pharmacol. Acta, 1, No. 14, 411-423 (1956).
S. F. Petropulos, “The action of an antimetabolite of thiamine on single myelinated nerve fibers,” J. Cell Comp. Physiol., 56, No. 1, 7-13 (1960).
M. Sasa, I. Takemoto, and K. Nishino, “The role of thiamine on excitable membrane of crayfish giant axon,” J. Nutr. Sci. Vitaminol., 22, Suppl., 21-24 (1976).
A. V. Romanenko, “Effects of thiamine on neuromuscular transmission in the frog,” Neirofiziologiya, 17, No. 6, 794-800 (1985).
T. Matsuda, H. Iwata, and J. R. Cooper, “Involvement of sulfhydryl groups in the inhibition of brain (Na+,K+)-ATPase by pyrithiamin,” Biochim. Biophys. Acta, 817, No. 1, 17-24 (1985).
J. M. Duclos and P. Haake, “Ring opening of thiamine analogs. The role of ring opening in physiological function,” Biochemistry, 13, No. 26, 5358-5362 (1974).
F. A. Oliveira, D. T. Galan, A. M. Ribeiro, et al., “Thiamine deficiency during pregnancy leads to cerebellar neuronal death in rat offspring: role of voltage-dependent K+ channels,” Brain Res., 1134, No. 1, 79-86 (2007).
L. Bettendorff, H. A. Kolb, and E. Schoffeniels, “Thiamine triphosphate activates an anion channel of large unit conductance in neuroblastoma cells” J. Membr. Biol., 136, No. 3, 281-288 (1993).
R. S. Gupta, “Microtubules, mitochondria and molecular chaperons: a new hypothesis for in vivo assembly of microtubules,” Biochem. Cell Biol., 68, No. 12, 1352-1363 (1990).
K. Berman and R. A. Fishman, “Thiamine phosphate metabolism and possible coenzyme independent functions of thiamine in brain,” J. Neurochem., 24, No. 3, 457-465 (1975).
E. Shoffeniels and D. C. Marginetu, “Thiamine triphosphate as specific operating substance in axonal conduction,” in: The Molecular Bases of Nerve Active Processes, Walter de Gruyter & Co., Berlin, New York (1984-1985), pp. 401-415.
R. V. Čhagovec and A. A. Rybina, “Investigations on the forms and metabolism of thiamine in tissues of the animal organism by means of thiamine S35,” International Symposium B Vitamins, Poznan (1959), pp. 324-338.
Yu. M. Parkhomenko, A, A, Strokina, S. Yu. Pilipchuk, et al., “The presence of two separrate active centers on the thiamine-binding protein of the synaptosome plasma membranes,”Ukr. Biokhim. Zh., 82, No. 1, 34-41 (2010).
Ch. Tanaka, Y. Itokawa, and S. Tanaka, “The axoplasmic transport of thiamine in rat sciatic nerve,” J. Histochem. Cytochem., 21, No. 1, 81-85 (1973).
A. Aiuchi, M. Matsunaga, T. Daimatsu, et al., “Effect of glucose and pyruvate metabolism on membrane potential in synaptosomes,” Biochim. Biophys. Acta, 771, No. 20, 228-234 (1984).
M. Browing, W. Benneff, P. Kelley, et al., “Evidence that the 40000 Mr phosphoprotein influenced by high frequency synaptic stimulation is the alpha subunit of pyruvate dehydrogenase,” Brain. Res., 218, No. 1, 255-266 (1981).
Yu. M. Parkhomenko. I. Yu. Chernysh, T. Ya. Churilova, et al., “Effects of thiaminephosphates on the activity of regulatory enzymes of the pyruvate dehydrogenase complex,” Ukr. Biokhim. Zh., 59, No. 5, 49-54 (1987).
J. R. Cooper, J. H. Pincus, Y. Itokawa, et al., “Enzyme inhibiting factor in subacute necrotizing enchephalomyelophathy,” Neurology, 19, No. 6, 841-845 (1969).
H. O. Nghiêm, L. Bettendorff, and J. P. Changeux, “Specific phosphorylation of Torpedo 43K rapsyn by endogenous kinase(s) with thiamine triphosphate as the phosphate donor,” FASEB J., 14, No. 3, 543-554 (2000).
L. Bettendorff, B. Lakaye, G. Kohn, et al., “Thiamine triphosphate: a ubiquitous molecule in search of a physiological role,” Metab. Brain Dis., 29, No. 4, 1069-1082 (2014).
F. Hucho, D. D. Randall., T. E. Roche, et al., “Keto acid dehydrogenase complexes. XVII. Kinetic and regulatory properties of pyruvate dehydrogenase kinase and pyruvate dehydrogenase phosphatase from bovine kidney and heart,” Arch. Biochem. Biophys., 151, No. 1, 328-340 (1972).
A. G. Vitreschak, D. A. Rodionov, and A. A. Mironov, “Riboswitches: the oldest mechanism for the regulation of gene expression?” Trends Gen., 20, No. 1, 44-50 (2004).
Li Sanshu and R. Ronald, “Breaker Eukaryotic TPP riboswitch regulation of alternative splicing involving long-distance base pairing,” Nucleic Acids Res., 41, No. 5, 3022-3031 (2013).
F. Pavlik, A. Bischoff, and I. Bitsch, “Peripheral nerve changes in thiamine deficiency and starvation,” Acta Neuropathol., 39, 211-218 (1977).
S. S. Jhala and A. S. Hazell, “Modeling neurodegenerative disease pathophysiology in thiamine deficiency: consequences of impaired oxidative metabolism,” Neurochem. Int., 58, No. 3, 248-260 (2011).
Yu. M. Parkhomenko, I. Yu. Chernysh, Z. C. Protasova, et al., “Interrelations between the thiamine content, activity of thiamine diphosphate-dependent enzymes, and level of reduced glutathione in the rat liver,” Ukr. Biokhim. Zh., 62, No. 6, 52-58 (1990).
G. E. Gibson and H. Zhang, “Interactions of oxidative stress with thiamine homeostasis promote neurodegeneration,” Neurochem. Int., 40, No. 6, 493-504 (2002).
Yu. M. Ostrovsky, Thiamine, Minsk, Belarus (1971).
Yu. M. Ostrovsky, S, V, Zabrodskaya, and D. A. Oparin, “A noncoenzymatic mechanism of inhibition of pyruvate dehydrogenase by phosphorylated thiamine analogs,” Dokl. AN SSSR, 295, No. 5, 1247-1249 (1987).
M. G. Velichko, R. V. Trebukhina, Yu. M. Ostrovsky, et al., “Effects of oxythiamine on metabolism of pyruvate and lactate in rat tissues,” Vopr. Med. Khim., 25, No. 2, 166-170 (1979).
S. Chornyy, J. Parkhomenko and N. Chorna, “Thiamine deficiency caused by thiamine antagonists triggers upregulation of apoptosis inducing factor gene expression and leads to caspase 3-mediated apoptosis in neuronally differentiated rat PC-12 cells,” Acta Biochim. Pol., 54, No. 2, 315-322 (2007).
L. C. Vedder, J. M. Hall, K. R. Jabrouin, et al., “Interactions between chronic ethanol consumption and thiamine deficiency on neural plasticity, spatial memory, and cognitive flexibility,” Alcohol Clin. Exp. Res., 39, No. 11, 2143-2153 (2015).
X. Wang, B. Wang, Z. Fanb, et al., “Thiamine deficiency induced endoplasmic reticulum stress in neurons,” Neuroscience, 144, No. 3, 1045-1056 (2007).
A. Ba, “Comparative effects of alcohol and thiamine deficiency on the developing central nervous system,” Brain Res., 225, No. 1, 235-242 (2011).
L. Qin and F. T. Crews, “Focal thalamic degeneration from ethanol and thiamine deficiency is associated with neuroimmune gene induction, microglial activation, and lack of monocarboxylic acid transporters,” Alcoholism: Clin. Exp. Res., 38, No. 3, 657-671 (2014).
M. L. Semenova, “Transgenic animals: What are they?,” Soros. Obrazovat. Zh., 7, No. 4, 13-20 (2001).
M. J. Lindhurst, G. Fiermonte, S. Song, et al., “Knockout of Slc25a19 causes mitochondrial thiamine pyrophosphate depletion, embryonic lethality, CNS malformations, and anemia,” Proc. Natl. Acad. Sci. USA, 103, No. 43, 15927-15932 (2006).
A. J. Cooper, K. F. Sheu, J. R. Burke, et al., “Pathogenesis of inclusion bodies in (CAG)n/Qn-expansion diseases with special reference to the role of tissue transglutaminase and to selective vulnerability,” J. Neurochem., 72, No. 3, 889-899 (1999).
A. I. Voskoboyev and I. P. Chernikevich, Biosynthesis, Degradation, and Transport of Thiamine Phosphates, Nauka i Tekhnka, Minsk (1987).
Yu. Parkhomenko, A. Vovk, Z. Protasova, et al., “Thiamine derivatives used to investigate the modulator role of thiamine in the biosynthesis of acetylcholine from pyruvate,” in: The 8th Intern. Conf. Thiamine: From Catalysis to Pathology (Liege, May 23–26), Liege (2014), p. 30.
A. V. Maltsev, N. V. Davidenko, V. K. Uteshev, et al., “Intensive protein synthesis in neurons and phosphorylation of precursor protein and beta-amyloid and tau protein factors are the triggers of amyloidosis and Alzheimer’s disease neurons,” Biochem., Suppl. Ser. B, Biomed. Chem., 59, No. 2, 144-170 (2013).
F. Mouton-Liger, C. Paquet, J. Dumurgier, et al., “Increased cerebrospinal fluid levels of double-stranded RNA-dependent protein kinase in Alzheimer’s disease,” Biol. Psychiat., 71, No. 9, 829-835 (2012).
A. Bose, F. Mouton-Liger, C. Paquet, et al., “Modulation of tau phosphorylation by the kinase PKR implication in Alzheimer’s disease,” Brain Pathol., 21, No. 2, 189-200 (2011).
X. Wang, B. Fan, J. Luo, et al., “Activation of doublestranded RNA-activated protein kinase by mild impairment of oxidative metabolism in neurouns,” J. Neurochem., 103, No. 6, 2380-2390 (2007).
F. Mouton-Liger, A. S. Rebillat, S. Gourmaud, et al., “PKR downregulation prevents neurodegeneration and β-amyloid production in a thiamine-deficient model,” Cell Death Dis., 6, No. 1, 1-8 (2015).
N. Y. Calingasan, S. E. Gandy, H. Baker, et al., “Novel neuritic clusters with accumulations of amyloid precursor protein and amyloid precursor-like protein 2 immunoreactivity in brain regions damages by thiamine deficiency,” Am. J. Pathol., 149, No. 3, 1063-1071 (1996).
S. S. Karuppagounder, X. Hui, D. Pechman, et al., “Translocation of Amyloid precursor protein C-terminal fragment (s) to the nucleus precedes neuronal death due to thiamine deficiency induced mild impairment of oxidative metabolism,” Neurochem. Res., 33, No. 7, 1365-1372 (2008).
G. E. Gibson, J. A. Hirsch, R. T. Cirio1, et al., “Abnormal thiamine-dependent processes in Alzheimer’s disease. Lessons from diabetes,” Mol. Cell. Neurosci., 55, No. 1, 17-25 ( 2013).
Q. Zhang, G. Yang, W. Li, et al., “Thiamine deficiency increases β-secretase activity and accumulation of β-amyloid peptides,” Neurobiol. Aging, 32, No. 1, 42-53 (2011).
J. Zhao, X. Sun, Z. Yu, et al., “Exposure to pyrithiamine increases β-amyloid accumulation, tau hyperphosphorylation, and glycogen synthase kinase-3 activity in the brain,” Neurotoxicol. Res., 19, No. 4, 575-583 (2011).
A. Schallier, I. Smolders, D. Van Dam, et al., “Regionand age-specific changes in glutamate transport in the AβPP23 mouse model for Alzheimer’s disease,” J. Alzheimer’s Dis., 24, No. 2, 287-300 (2011).
M. Hoshi, A. Takashima, K. Noguchi, et al., “Regulation of mitochondrial pyruvate dehydrogenase activity by tau protein kinase I/glycogen synthase kinase 3 beta in brain,” Proc. Natl. Acad. Sci. USA, 93, No. 7, 2719-2723 (1996).
A. Takashima, K. Noguchi, K. Sato, et al., “Tau protein kinase I is essential for amyloid beta-protein-induced neurotoxicity,” Proc. Natl. Acad. Sci. USA, 90, No. 16, 7789-7793 (1993).
M. Hoshi, M. Sato, S, Kondo, et al. “Different localization of tau protein kinase I/glycogen synthase kinase-3 beta from glycogen synthase kinase-3 alpha in cerebellum mitochondria,” J. Biochem., 118, No. 4, 683-685 (1995).
J. E. Lawson, S. H. Park, A. R. Mattison, et al., “Cloning, expression, and properties of the regulatory subunit of bovine pyruvate dehydrogenase phosphatase,” J. Biol. Chem., 272, No. 50, 31625-31629 (1997).
X. Pan, N. Gong, J. Zhao, et al., “Powerful beneficial effects of benfothiamine on cognitive impairment and beta-amyloid deposition in amyloid precursor protein/presenilin-1 transgenic mice,” Brain, 133, No. 5, 1342-1351 (2010).
K. Meador, D. Loring, M. Nichols, et al., “Preliminary findings of high-dose thiamine in dementia of Alzheimer’s type,” J. Geriat. Psychiat. Neurol., 6, No. 4, 222-229 (1993).
F. J. Jiménez-Jiménez, J. A. Molina, A. Hernánz, et al., “Cerebrospinal fluid levels of thiamine in patients with Parkinson’s disease,” Neurosci. Lett., 271, No. 1, 33-36 (1999).
U. Laforenza, C. Patrini, M. Poloni, et al., “Thiamine mono- and pyrophosphatase activities from brain homogenate of Guamanian amyotrophic lateral sclerosis and parkinsonism-dementia patients,” J. Neurol. Sci., 109, No. 2, 156-161 (1992).
M. Favier, C. Carcenac, G. Drui, et al., “High-frequency stimulation of the subthalamic nucleus modifies the expression of vesicular glutamate transporters in basal ganglia in a rat model of Parkinson’s disease,” BMC Neurosci., 14, No. 152, 1-13 (2013).
K. G. McLure, M. Nakagi, and M. B. Kastan, “NAD+ modulates p53 DNA binding specificity and function,” Mol. Cell Biol., 24, No. 22, 9958-9967 (2004).
S. A. Chornyy, Effects of Thiamine Antagonists on Signaling Pathways of Apoptosis in Cultured Nerve Cells, Abstr. Thesis Cand. Biol., Kyiv (2010).
T. Tanaka, D. Yamamoto, T. Sato, et al., “Adenosine thiamine triphosphate (AThTP) inhibits Poly(ADPRibose) polymerase-1 (PARP-1) activity,” J. Nutr. Sci. Vitaminol., 57, No. 2, 192-196 (2011).
Y. Bando, R. Onuki, T. Katayama, et al., “Double-strand RNA dependent protein kinase (PKR) is involved in the extrastriatal degeneration in Parkinson’s disease and Huntington’s disease,” Neurochem. Int., 46, No. 1, 11-18 (2005).
A. Costantini, M. I. Pala, L. Compagnoni, and M. Colangeli, “High-dose thiamine as initial treatment for Parkinson’s disease,” BMJ Case Reports (2013); doi:10.1136/bcr-2013-009289.
A. Costantini and R. Fancellu, “An open-label pilot study with high-dose thiamine in Parkinson’s disease,” Neural Regen. Res., 11, No. 3, 406-407 (2016).
A. Costantini, M. I. Pala, E. Grossi, et al., “Long-term treatment with high-dose thiamine in Parkinson disease: an open-label pilot study,” J. Altern. Complement Med., 21, No. 12, 740-747 (2015).
L. C. Heap, O. E. Pratt, R. J. Ward, et al., “Individual susceptibility to Wernicke-Korsakoff syndrome and alcoholism-induced cognitive deficit: impaired thiamine utilization found in alcoholics and alcohol abusers,” Psychiat. Genet., 12, No. 4, 217-224 (2002).
Y. M. Parkhomenko, P. A. Kudryavtsev, S.Y. Pylypchuk, et al., “Chronic alcoholism in rats induces a compensatory response, preserving brain thiamine diphosphate, but the brain 2-oxo acid dehydrogenases are inactivated despite unchanged coenzyme levels,” J. Neurochem., 117, No. 6, 1055-1065 (2011).
K. O. Bueno, L. de Souza Resende, A. F. Ribeiro, et al., “Spatial cognitive deficits in an animal model of Wernicke-Korsakoff syndrome are related to changes in thalamic VDAC protein concentrations,” Neuroscience, 294, No. 1, 29-37 (2015).
D. W. Frijlink, J. J. Tilanus, and G. Roks, “Elevated cerebrospinal fluid tau in Wernicke encephalopathy,” BMJ Case Rep. (2012), doi: 10.1136/bcr-2012-006661.
Yu. M. Parkhomenko., G. V. Donchenko, S. A. Chornyy, et al., “Thiamine metabolism in neurons and their vital capacity upon the action of ethanol and acetaldehyde,“ Neurophysiology, 46, No. 1, 1-9 (2014).
A. S. Hazell, K. V. Rao, N. C. Danbolt, et al., “Selective down-regulation of the astrocyte glutamate transporters GLT-1 and GLAST within the medial thalamus in experimental Wernicke’s encephalopathy,” J. Neurochem., 78, No. 3, 560-568 (2001).
N. Latt and G. Dore, “Thiamine in the treatment of Wernicke encephalopathy in patients with alcohol use disorders,” Int. Med. J., 44, No. 9, 911-915 (2014). 94. E. Rees and L. R. Gowing, “Supplementary thiamine is still important in Alcohol dependence,” Alcohol Alcoholism, 48, No. 1, 88-92 (2013).
R. Nardone, Y. Höller, M. Storti, et al., “Thiamine deficiency induced neurochemical, neuroanatomical, and neuropsychological alterations: a reappraisal,” Sci. World J., 2013, 2013:309143, doi:10.1155/2013/309143. eCollection2013.
S. Afadlal R. Labetoulle, and A. S. Hazell, “Role of astrocytes in thiamine deficiency,” Metab. Brain Dis., 29, No. 4, 1061-1068 (2014).
S. A. Chornyy and Y. M. Parkhomenko, “Comparative characteristics of the action of thiamine antagonists as apoptosis inducers in different types of nerve cells,” Ukr. Biokhim. Zh., 80, No. 5, 76-84 (2008).
R. K. Sheean, C. L. Lau, Y. S. Shin, et al., “Links between L-glutamate transporters, Na+/K+-ATPase and cytoskeleton in astrocytes: evidence following inhibition with rottlerin,” Neuroscience, 254, 335-346 (2013).
B. H. Shin, S. H. Choi, E. Y. Cho, et al., “Thiamine attenuates hypoxia-induced cell death in cultured neonatal rat cardiomyocytes,” Mol. Cells, 18, No. 2, 133-140 (2004).
P. H. Frederikse, I. P. Farnsworth, and J. S. Zigler, “Thiamine deficiency in vivo produces fiber cell degeneration in mouse lenses,” Biochem. Biophys. Res. Commun., 258, No. 3, 703-707 (1999).
D. Ye. Bobkov, I. V. Kropachyova, and G. P. Pinayev, “Multimolecular complexes that contain a P65 subunit of the NF-kB factor and cytoskeletal proteins in A431 cells,” Biol. Membr., 27, No. 1, 133-137 (2010).
R. Comming and R. D. Burgoyne, “Compartmentalization of neuronal cytoskeletal proteins,” Biosci. Rep., 3, No. 11, 997-1006 (1983).
V. A. Berezin and G. M. Shevchenko, “Neurospecific proteins of the cytoskeleton,” Ukr. Biokhim. Zh., 59, No. 1, 105-115 (1987).
A. Murai and E. Katsura, “Thiamine triphosphatase activity of myosin and accelerating effect of thiamine di- and triphosphates on superprecipitation of actomyosin,” J. Nutr. Sci. Vitaminol., 21, No. 3, 169-183 (1975).
I. A. Romero, R. J. Rist, A. Aleshaiker, et al., “Metabolic and permeability changes caused by thiamine deficiency in immortalized rat brain microvessel endothelial cells,” Brain Res., 756, Nos. 1/2, 133-140 (1997).
Z. J. Ke, X. Wang, Z. Fan, and J. Lou, “Ethanol promotes thiamine deficiency-induces neuronal death: involvement of double-stranded RNA-activated protein kinase,” Alcohol Clin. Exp. Res., 33, No. 6, 1097-1103 (2009).
L. S. Resende, A. M. Ribeiro, D. Wernerb, et al. “Thiamine deficiency degrades the link between spatial behavior and hippocampal synapsin I and phosphorylated synapsin I protein levels,” Behav. Brain Res., 232, No. 2, 421-425 (2012).
K. M. Cullen and G. M. Halliday, “Mechanisms of cell death in cholinergic basal forebrain neurons in chronic alcoholics,” Metab. Brain Dis., 10, No. 1, 81-91 (1995).
R. Perez-Pineiro, T. C. Bjorndahl, M. V. Berjanski, et al., “The prion protein binds thiamine,” FEBS J., 278, No. 21, 4002-4014 (2011).
N. S. Pagadala, T. C. Bjorndah, N. Blinov, et al., “Molecular docking of thiamine reveals similarity in binding properties between the prion protein and other thiamine-binding proteins,” J. Mol. Model., 19, No. 2, 5225-5235 (2013).
R. Mendoza, M. M. Anderson, and J. Overbaugh, “A putative thiamine transport protein is a receptor for feline leukemia virus subgroup A,” J. Virol., 80, No.7, 3378-3385 (2006).
R. Mendoza, A. D. Miller, and J. Overbaugh, “Disruption of thiamine uptake and growth of cells by feline leukemia virus subgroup A,” J. Virol., 87, No. 5, 2412-2419 (2013).
S. Shoji, K. Furuishi, S. Misumi, et al., “Thiamine disulfide as a potent inhibitor of human immunodeficiency virus (type-1) production,” Biochem. Biophys. Res. Commun., 205, No. 1, 967-975 (1994).
S. Shoji, K. Furuishi, A. Ogata, et al., “An allosteric drug, O,O’-bismyristoyl thiamine disulfide, suppresses HIV-1 replication through prevention of nuclear translocation of both HIV-1 Tat and NF-kappa B,” Biochem. Biophys. Res. Commun., 249, No. 3, 745-753 (1998).
V. Bunik, Y. Parkhomenko, T. Kaehne, et al. “Thiamine and thiazole binding proteome includes DJ-1, amyloid beta and several membrane proteins,” in: Proceeding of the 11th International Conference on Alzheimer’s and Parkinson’s Diseases (Florence, Italy, March 6-10), Florence (2013), Abstract-No: A-459-0001-00462.
G. Mkrtchyan, V. Aleshin, Yu. Parkhomenko, et al., “Molecular mechanisms of the non-coenzyme action of thiamine in brain: biochemical, structural and pathway analysis,” Sci. Rep. 2015, 5, 1258, 1-26, doi: 10.1038/srep 12583 (2015).
N. Lev, D. Roncevic, D. Ickowicz, et al., “Role of DJ-1 in Parkinson’s disease,” J. Mol. Neurosci., 29, No. 3, 215-225 (2006).
N. A. Rege and J. S. Hagood, “Thy-1, a versatile modulator of signaling affecting cellular adhesion, proliferation, survival, and cytokine/growth factor responses,” Biochim. Biophys. Acta, 1763, No. 10, 991-999 (2006).
E. Szirmai, “Thiamine (vitamin B 1) and its effects on the uterine musculature and on the pain during delivery (preliminary report),” Zentralbl. Gynakol., 83, No. 1, 554-555 (1961).
E. Fujihira, Y. Tarumoto, M. Ajioka, et al., “Analgesic effect of O-isobutyrylthiamine disulfide on experimentally induced,” Yakugaku Zasshi., 93, No. 3, 388-391 (1973).
H. Quirin, “Pain and vitamin B therapy,” Bibl. Nutr. Dieta, 38, No. 1, 110-111 (1986).
A. Costantini, M. I. Pala, S. Tundo, et al. “High-dose thiamine improves the symptoms of fibromyalgia,” BMJ Case Rep., 2013, 1-4, doi: 10.1136/bcr-2013-009019 (2013).
S. H. Lee, S. Y. Kim, J. H. Kim, et al. “Phosphoproteomic analysis of electroacupuncture analgesia in an Inflammatory pain rat model,” Mol. Med. Rep., 6, No. 1, 157-162 (2012).
H. M. Beere and D. R. Green,”Stress management – heat shock protein-70 and the regulation of apoptosis,” Trends Cell. Biol., 11, No. 1, 6-10 (2001).
J. V. Y. Rodrigues, B. J. Henriques, T. G. Lucas, et al., “Cofactors and metabolites as protein folding helpers in metabolic diseases,” Curr. Med. Chem., 2, No. 22, 2546-2559 (2012).
S. A. Petrov, “Noncoenzymatic effects of thiamine and its metabolites,” Biomed. Khim., 52, No. 4, 335-345 (2006).
S. A. Petrov, “Inhibition of alcohol dehydrogenase by thiochrome,” Ukr. Biokhim. Zh., 64, No. 6, 91-94 (1992).
S. A. Petrov, O. A. Kotenko, and M. El-Absi, “Effects of thiamine and its metabolites on the activity of tissue and purified lactate dehydrogenase,” Ukr. Biokhim. Zh., 63, No. 2, 105-108 (1991).
A. I. Vovk, L. V. Babii, and I. V. Muraviova, “Relative reaction abilities of thiamine monophosphate and thiamine diphosphate in their interaction with alkaline phosphatase,” Ukr. Biokhim. Zh., 74, No. 1, 93-96 (2002).
Yu. M. Ostrovskii, S, V, Zabrodskaya, T. I. Zimatkina, et al., “Selective inhibition of pyruvate dehydrogenase in the murine liver and heart by triphosphorus esters of thiochrome and tetrahydrothiamine,” Biokhimiya, 48, No. 6, 928-931 (1983).
S. A. Strumilo, Yu. V. Kiselevskii, N. I. Taranda, et al., “Interaction of the pyruvate dehydrogenase complex from the cardial muscle with thiamine diphosphate and its derivatives,” Vopr. Med. Khim., 35, No. 2, 102-105 (1989).
X. Zhang, I. Hernandez, D. Rei, et al., “Diaminothiazoles modify tau phosphorylation and improve the tauopathy in mouse models,” J. Biol. Chem., 288, No. 3, 22042-22056 (2013).
Li Chien-Ming, Z. Wang, Y. Lu et al., “Biological activity of 4-substituted methoxybenzoyl-aryl-thiazole: an active microtubule inhibitor,” Cancer Res., 71, No. 1, 216-224 (2011).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Parkhomenko, Y.M., Pavlova, A.S. & Mezhenskaya, O.A. Mechanisms Responsible for the High Sensitivity of Neural Cells to Vitamin B1 Deficiency. Neurophysiology 48, 429–448 (2016). https://doi.org/10.1007/s11062-017-9620-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11062-017-9620-3