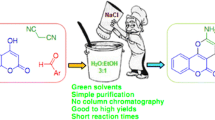
Abstract
Solvent-free one-pot synthesis of 2-amino-4H-chromene scaffold is described in a very simple, efficient, and environmentally benign method using sodium carbonate as a cheap and non-toxic catalyst with up to excellent yields.
Similar content being viewed by others
References
Ellis GP (1997) In: Weissberger A, Taylor EC (eds) The chemistry of heterocyclic compounds. chromenes, chromanes and chromones, chap XI, Alkylchromones. Wiley, New York, pp 581–631
Hafez EAA, Elnagdi MH, Elagemey AGA, El-Taweel FMAA (1987) Nitriles in heterocyclic synthesis—novel synthesis of benzo[c]-coumarin and of benzo[c]pyrano[3,2-c] quinoline derivatives. Heterocycles 26: 903–907. doi:10.3987/R-1987-04-0903
Poupaert J, Carato P, Colacino E (2005) 2(3H)-benzoxazolone and bioisosters as “privileged scaffold” in the design of pharmacological probes. Curr Med Chem 12: 877–885. doi:10.2174/0929867053507388
Triggle DJ (2003) 1,4-Dihydropyridines as calcium channel ligands and privileged structures. Cell Mol Neurobiol 23: 293–303. doi:10.1023/A:1023632419813
Kidwai M, Saxena S, Khan MKR, Thukral SS (2005) Aqua mediated synthesis of substituted 2-amino-4H-chromenes and in vitro study as antibacterial agents. Bioorg Med Chem Lett 15: 4295–4298. doi:10.1016/j.bmcl.2005.06.041
Mohr SJ, Chirigos MA, Fuhrman FS, Pryor JW (1975) Pyran copolymer as an effective adjuvant to chemotherapy against a murine leukemia and solid tumor. Cancer Res 35(12): 3750–3754
Gao M, Miller KD, Hutchins GD, Zheng Q-H (2010) Synthesis of carbon-11-labeled 4-aryl-4H-chromens as new PET agents for imaging of apoptosis in cancer. Appl Radiat Isot 68: 110–116. doi:10.1016/j.apradiso.2009.09.067
Weber L (2002) Multi-component reactions and evolutionary chemistry. Drug Discov Today 7: 143–147. doi:10.1016/S1359-6446(02)00010-7
Doemling A (2002) Recent advances in isocyanide-based multicomponent chemistry. Curr Opin Chem Biol 6: 306–313. doi:10.1016/S1367-5931(02)00328-9
Thompson LA (2000) Recent applications of polymer-supported reagents and scavengers in combinatorial, parallel, or multistep synthesis. Curr Opin Chem Biol 4: 324–337. doi:10.1016/S1367-5931(00)00096-X
Nefzi A, Ostresh JM, Houghten RA (1997) The current status of heterocyclic combinatorial libraries. Chem Rev 97: 449–472. doi:10.1021/cr960010b
Elinson MN, Dorofeev AS, Miloserdov FM, Ilovaisky AI, Feducovich SK, Belyakov PA, Nikishin GI (2008) Catalysis of salicylaldehydes and two different C_H acids with electricity: first example of an efficient multicomponent approach to the design of functionalized medicinally privileged 2-Amino-4H-Chromene scaffolds. Adv Synth Catal 350: 591–601. doi:10.1002/adsc.200700493
Elagamay AGA, El-Taweel FMAA (1990) Nitriles in heterocyclic synthesis: synthesis of condensed pyrans. Indian J Chem B 29: 885–886
Anderson DR, Hegde S, Reinhard E, Gomez L, Vernier WF, Lee L, Liu S, Sambandam A, Snider P A, Masih L (2005) Aminocyanopyridine inhibitors of mitogen activated protein kinase-activated protein kinase 2 (MK-2). Bioorg Med Chem Lett 15: 1587–1590. doi:10.1016/j.bmcl.2005.01.067
Kemnitzer W, Kasibhatla S, Jiang S, Zhang H, Zhao J, Jia S, Xu L, Crogan-Grundy C, Denis R, Barriault N, Vaillancourt L, Charron S, Dodd J, Attardo G, Labrecque D, Lamothe S, Gourdeau H, Tseng B, Drewe J, Cai S X (2005) Discovery of 4-aryl-4H-chromenes as a new series of apoptosis inducers using a cell- and caspase-based high-throughput screening assay. 2. Structure-activity relationships of the 7- and 5-, 6-, 8-positions. Bioorg Med Chem Lett 15: 4745–4751. doi:10.1016/j.bmcl.2005.07.066
Kemnitzer W, Drewe J, Jiang S, Zhang H, Wang Y, Zhao J, Jia S, Herich J, Labreque D, Storer R, Meerovitch K, Bouffard D, Rej R, Denis R, Blais C, Lamothe S, Attardo G, Gourdeau H, Tseng B, Kasibhatla S, Cai S X (2004) Discovery of 4-aryl-4H-chromenes as a new series of apoptosis inducers using a cell- and caspase-based high-throughput screening assay. 1. Structure-activity relationships of the 4-aryl group. J Med Chem 47: 6299–6310. doi:10.1021/jm049640t
Ballini R, Bosica G, Conforti ML, Maggi R, Mazzacanni A, Righi P, Sartori G (2001) Three-component process for the synthesis of 2-amino-2-chrome in aqueous media. Tetrahedron 57: 1395–1398. doi:10.1016/S0040-4020(00)01121-2
Jin TS, Xiao JC, Wang SJ, Li TS (2004) Ultrasound-assisted synthesis of 2-amino-2-chromenes with cetyltrimethylammonium bromide in aqueous media. Ultrason Sonochem 11: 393–397. doi:10.1016/j.ultsonch.2003.10.002
Ballini R, Bigi F, Conforti ML (2000) Multicomponent reactions under clay catalysis. Catal Today 60: 305–309. doi:10.1016/S0920-5861(00)00347-3
Wang X, Shi D, Tu S (2004) Synthesis of 2-Aminochromene Derivatives Catalyzed by KF/Al2O3. Synth Commun 34: 509–514. doi:10.1081/SCC-120027291
Sunil Kumar B, Srinivasulu N, Udupi R, Rajitha B, Thirupathi Reddy Y, Narsimha Reddy P, Kumar P (2006) Efficient synthesis of benzo[g]-and benzo[h]chromene derivatives by one-pot three-component condensation of aromatic aldehydes with active methylene compounds and naphthols. Russ J Org Chem 42: 1813–1815. doi:10.1134/S1070428006120098
Shestopalov AM, Emelianova YM, Nesterov VN (2002) Molecular and crystal structure of 2-amino-3-cyano-6-hydroxy-4-phenyl-4H-benzo[f]chromene. Russ Chem Bull 51: 2238–2243. doi:10.1023/A:1022135402451
Maggi R, Ballini R, Sartori G (2004) Basic alumina catalysed synthesis of substituted 2-amino-2-chromenes via three-component reaction. Tetrahedron Lett. 45: 2297–2299. doi:10.1016/j.tetlet.2004.01.115
Kumar D, Reddy VB, Mishra BK, Rana GR, Mallikarjuna N, Nadagouda R, Varma S (2007) Nanosized magnesium oxide as catalyst for the rapid and green synthesis of substituted 2-amino-2-chromenes. Tetrahedron 63: 3093–3097. doi:10.1016/j.tet.2007.02.019
Heravi MM, Bakhtiari Kh, Zadsirjan V, Bamoharram FF, Heravi MO (2007) Aqua mediated synthesis of substituted 2-amino-4H-chromenes catalyzed by green and reusable Preyssler heteropolyacid. Bioorg Med Chem Lett 17: 4262–4265. doi:10.1016/j.bmcl.2007.05.023
Gong K, Wang HL, Fang Liu Z-L (2008) Basic ionic liquid as catalyst for the rapid and green synthesis of substituted 2-amino-2-chromenes in aqueous media. Catal Commun 9: 650–653. doi:10.1016/j.catcom.2007.07.010
Ren YM, Cai Ch (2008) Convenient and efficient method for synthesis of substituted 2-amino-2-chromenes using catalytic amount of iodine and K2CO3 in aqueous medium. Catal Commun 9: 1017–1020. doi:10.1016/j.catcom.2007.10.002
Balalaie S, Ramezanpour S, Bararjanian M, Gross JH (2008) DABCO-Catalyzed efficient synthesis of naphthopyran derivatives via one-pot three-component condensation reaction at room temperature. Synth Commun 38: 1078–1089. doi:10.1080/00397910701862865
Mashkouri S, Naimi-Jamal MR (2009) Mechanochemical solvent-free and catalyst-free one-pot synthesis of pyrano[2,3-d]pyrimidine-2,4(1H,3H)-diones with quantitative yields. Molecules 14(1): 474–479. doi:10.3390/molecules14010474
Kaupp G, Naimi-Jamal MR, Schmeyers J (2003) Solvent-free Knoevenagel condensations and Michael additions in the solid state and in the melt with quantitative yield. Tetrahedron 59: 3753–3760. doi:10.1016/S0040-4020(03)00554-4
Sharifi A, Mirzaei M, Naimi-Jamal MR (2007) A facile solvent-free one-pot three-component Mannich reaction of aldehydes, amines and terminal alkynes catalysed by CuCl2. J Chem Res 2: 129–132. doi:10.3184/030823407X191994
Sharifi A, Mohsenzadeh F, Naimi-Jamal MR (2000) Solvent-free preparation of monoacylaminals assisted by microwave irradiation. J Chem Res 8: 394–396. doi:10.3184/030823400103167813
Naimi-Jamal MR, Mirzaei M, Bolourtchian M, Sharifi A (2006) One-pot solvent-free preparation of 2-phenyl-1,3,2-aryldioxaborins on acidic alumina. Synth Commun 36(18): 2711–2717. doi:10.1080/00397910600764741
Sharifi A, Mirzaei M, Naimi-Jamal MR (2006) Modified Glaser reaction of terminal alkynes on KF/Alumina. Monatshefte fur Chemie 137(2): 213–217. doi:10.1007/s00706-005-0416-6
Sharifi A, Farhangian H, Mohsenzadeh F, Naimi-Jamal MR (2002) Microwave assisted Mannich reaction of terminal alkynes on alumina. Monatshefte fur Chemie 133(2): 199–204. doi:10.1007/s706-002-8251-8
Abd-El-Aziz AS, El-Agrody AM, Bedair AH, Corkery TC, Ata A (2004) Synthesis of hydroxyquinoline derivatives, aminohydroxychromene, aminocoumarin and their anti-bacterial activities. Heterocycles 63(8): 1793–1812. doi:10.3987/COM-04-10089
Abdolmohammadi S, Balalaie S (2007) Novel and efficient catalysts for the one-pot synthesis of 3,4-dihydropyrano[c]chromene derivatives in aqueous media. Tetrahedron Lett 48: 3299–3303. doi:10.1016/j.tetlet.2007.02.135
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Naimi-Jamal, M.R., Mashkouri, S. & Sharifi, A. An efficient, multicomponent approach for solvent-free synthesis of 2-amino-4H-chromene scaffold. Mol Divers 14, 473–477 (2010). https://doi.org/10.1007/s11030-010-9246-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-010-9246-5