Abstract
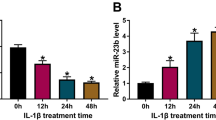
Osteoarthritis (OA) is characterized by degradation of articular cartilage. MiRNAs are involved in the regulation of chondrogenesis and OA. We aimed to investigate effects and mechanisms of miR-19b-3p in regulating chondrocytes viability, cartilage degradation and inflammatory response. Primary chondrocytes were isolated from cartilages in control subjects and patients with OA. Murine ATDC5 cells were pre-conditioned with IL-1β in vitro. Expressions and interaction of miR-19b-3p with G protein-coupled receptor kinase 6 (GRK6), and their effects on inflammation, chondrocytes viability and cartilage degradation were determined after miR-19b-3p mimic or GRK6 siRNA transfection. MiR-19b-3p was significantly decreased in OA chondrocytes and IL-1β-stimulated ATDC5 cells, in paralleled with the elevated type-II-collagen, aggrecan, MMP13 and GRK6 expression. MiR-19b-3p mimic dramatically increased the viability of chondrocytes and suppressed cell apoptosis. It also increased type-II-collagen, aggrecan expression and glycosaminoglycan (sGAG) content, and decreased the expression of MMP-1 and MMP-13 that controlled by IL-1β. Overexpression of miR-19b-3p inhibited the production of IL-6 and IL-8 in ATDC5 cells. However, the protective effects of miR-19b-3p mimic on IL-1β induced cell death; IL-8 production and sGAG decrease were greatly discounted by GRK6 lentiviral vectors. Luciferase reporter assay confirmed that GRK6 gene was a direct target ofmiR-19b-3p. GRK6 siRNA transfection antagonized the IL-1β-induced chondrocytes injury, extracellular matrix degradation and inflammatory response. MiR-19b-3p mimic and GRK6 siRNA showed comparable inhibitory effect on IL-1β-provoked NF-κB as reflected by the expression of p-p65. NF-κB translocation inhibition with PS1154 reversed the effects of IL-1β on IL-8 and sGAG. Collectively, miR-19b-3p attenuated OA by targeting GRK6-NF-κB pathway.





Similar content being viewed by others
References
Muniraj N, Stamp LK, Badiei A, Hegde A, Cameron V, Bhatia M (2017) Hydrogen sulfide acts as a pro-inflammatory mediator in rheumatic disease. Int J Rheum Dis 20:182–189
Yang X, Guan Y, Tian S, Wang Y, Sun K, Chen Q (2016) Mechanical and IL-1β responsive mir-365 contributes to osteoarthritis development by targeting histone deacetylase 4. Int J Mol Sci 17:436
Abouheif MM, Nakasa T, Shibuya H, Niimoto T, Kongcharoensombat W, Ochi M (2010) Silencing microRNA-34a inhibits chondrocyte apoptosis in a rat osteoarthritis model in vitro. Rheumatology 49:2054–2060
Boehme KA, Rolauffs B (2018) Onset and progression of human osteoarthritis-can growth factors, inflammatory cytokines, or differential miRNA expression concomitantly induce proliferation, ECM degradation, and inflammation in articular cartilage? Int J Mol Sci 19:E2282
Yang X, Zhang Q, Gao Z, Yu C, Zhang L (2018) Down-regulation of miR-150 alleviates inflammatory injury induced by interleukin 1 via targeting kruppel-like factor 2 in human chondrogenic cells. Cell Physiol Biochem 47:2579–2588
Malemud CJ (2018) MicroRNAs and osteoarthritis. Cells 7:E92
Kong R, Gao J, Si Y, Zhao D (2017) Combination of circulating miR-19b-3p, miR-122-5p and miR-486-5p expressions correlates with risk and disease severity of knee osteoarthritis. Am J Transl Res 9:2852–2864
Zhou Y, Li RJ, Li M, Liu X, Zhu HY, Ju Z, Miao X, Xu GY (2016) Overexpression of GRK6 attenuates neuropathic pain via suppression of CXCR8 in rat dorsal root ganglion. Mol Pain 12:1744806916646381
Sun B, Gao Y, Dong L, Wu X, Wei H, Wen H, Deng X, Zhang F (2013) Expression of G-protein-coupled receptor kinase 6 (GRK6) after acute spinal cord injury in adult rat. J Mol Histol 44:259–270
Managò F, Espinoza S, Salahpour A, Sotnikova TD, Caron MG, Premont RT, Gainetdinov RR (2012) The role of GRK6 in animal models of Parkinson’s Disease and L-DOPA treatment. Sci Rep 2:301
Aigner T, Fundel K, Saas J, Gebhard PM, Haag J, Weiss T, Zien A, Obermayr F, Zimmer R and Bartnik E (2006) Large-scale gene expression profiling reveals major pathogenetic pathways of cartilage degeneration in osteoarthritis. Arthritis Rheum 54:3533–3544
Xu B, Li YY, Ma J, Pei FX (2016) Roles of microRNA and signaling pathway in osteoarthritis pathogenesis. J Zhejiang Univ B 17:200–208
Matsukawa T, Sakai T, Yonezawa T, Hiraiwa H, Hamada T, Nakashima M, Ono Y, Ishizuka S, Nakahara H, Lotz MK, Asahara H, Ishiguro N (2013) MicroRNA-125b regulates the expression of aggrecanase-1 (ADAMTS-4) in human osteoarthritic chondrocytes. Arthritis Res Ther 15:R28
Song J, Jin EH, Kim D, Kim KY, Chun CH, Jin EJ (2015) MicroRNA-222 regulates MMP-13 via targeting HDAC-4 during osteoarthritis pathogenesis. BBA Clin 3:79–89
Iliopoulos D, Malizos KN, Oikonomou P, Tsezou A (2008) Integrative microRNA and proteomic approaches identify novel osteoarthritis genes and their collaborative metabolic and inflammatory networks. PLoS ONE 3:e3740
Cuadra VMB, González-Huerta NC, Romero-Córdoba S, Hidalgo-Miranda A, Miranda-Duarte A (2014) Altered expression of circulating microRNA in plasma of patients with primary osteoarthritis and in silico analysis of their pathways. PLoS ONE 9(6):e97690
Kolhe R, Hunter M, Liu S, Jadeja RN, Pundkar C, Mondal AK, Mendhe B, Drewry M, Rojiani MV, Liu Y (2017) Gender-specific differential expression of exosomal miRNA in synovial fluid of patients with osteoarthritis. Sci Rep 7:2029
Kong R, Gao J, Si Y, Zhao D (2017) Combination of circulating miR-19b-3p, miR-122-5p and miR-486-5p expressions correlates with risk and disease severity of knee osteoarthritis. Am J Transl Res 9:2852
Usama A, Zhu B, Jing Y, Wan S, Nie Y, Zheng C, Min C, Chong W, Duan X, Hao Z (2016) MicroRNA-19b-3p modulates Japanese encephalitis virus-mediated inflammation via targeting RNF11. J Virol 90:4780–4795
Zhang Y, Jia J, Yang S, Liu X, Ye S, Tian H (2014) MicroRNA-21 controls the development of osteoarthritis by targeting GDF-5 in chondrocytes. Exp Mol Med 46:e79–e79
Pan L, Liu D, Zhao L, Wang L, Xin M, Li X (2018) Long noncoding RNA MALAT1 alleviates lipopolysaccharide-induced inflammatory injury by upregulating microRNA-19b in murine chondrogenic ATDC5 cells. J Cell Biochem 119:10165–10175
Ohba Y, Nakaya M, Watari K, Nagasaka A, Kurose H (2015) GRK6 phosphorylates IκBα at Ser 32/Ser 36 and enhances TNF-α-induced inflammation. Biochem Biophys Res Commun 461:307–313
Gurevich VV, Gurevich EV (2019) GPCR signaling regulation: the role of GRKs and arrestins. Front Pharmacol 10:125
Eijkelkamp N, Heijnen CJ, Lucas A, Premont RT, Elsenbruch S, Schedlowski M, Kavelaars A (2007) G protein-coupled receptor kinase 6 controls chronicity and severity of dextran sodium sulphate-induced colitis in mice. Gut 56:847–854
Ohba Y, Nakaya M, Watari K, Nagasaka A, Kurose H (2015) GRK6 phosphorylates IκBα at Ser 32/Ser 36 and enhances TNF-α-induced inflammation. Biochem Biophys Res Commun 461:307–313
Marcu KB, Otero M, Olivotto E, Borzi RM, Goldring MB (2010) NF-kappaB signaling: multiple angles to target OA. Curr Drug Targets 11:599–613
Rigoglou S, Papavassiliou AG (2013) The NF-kappaB signalling pathway in osteoarthritis. Int J Biochem Cell Biol 45:2580–2584
Saito T, Tanaka S (2017) Molecular mechanisms underlying osteoarthritis development: notch and NF-kappaB. Arthritis Res Ther 19:94
Kanbe K, Takagishi K, Chen Q (2002) Stimulation of matrix metalloprotease 3 release from human chondrocytes by the interaction of stromal cell-derived factor 1 and CXC chemokine receptor 4. Arthritis Rheumatol 46:130–137
Kanbe K, Takemura T, Takeuchi K, Chen Q, Takagishi K, Inoue K (2004) Synovectomy reduces stromal-cell-derived factor-1 (SDF-1) which is involved in the destruction of cartilage in osteoarthritis and rheumatoid arthritis. Bone Joint J 86:296–300
Funding
This work is supported by Shaanxi Science and Technology Co-ordination & Innovation Project (Grant No. 2015KTCL03-02).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This study was approved by the ethics committee of the Shaanxi Provincial People’s Hospital and adhered to the Helsinki Declaration (2000).
Informed consent
Informed consent was obtained from each patient.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Duan, L., Duan, D., Wei, W. et al. MiR-19b-3p attenuates IL-1β induced extracellular matrix degradation and inflammatory injury in chondrocytes by targeting GRK6. Mol Cell Biochem 459, 205–214 (2019). https://doi.org/10.1007/s11010-019-03563-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-019-03563-2