Abstract
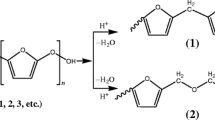
Macromonomers from oxazoline derivates were synthesized by using in conjunction with ethylenically unsaturated modifier. allyl chloride (AC) is chosen as representative modifier for the synthesis of allyl-terminated macromonomers in the presence of an acid exchanged montmorillonite as catalyst. The macromonomers were characterized by FT-IR, 1H-NMR, 13C-NMR and MALDI-TOF mass spectroscopy. The effects of amount of catalyst, amount of initiator, solvent and temperature on yields and intrinsic viscosity were studied. NMR and IR spectra of macromonomers show characteristic chemical shifts absorptions of the ethylenically unsaturated group of the modifier. The MALDI-TOF spectrum shows that the macromonomers synthesized accordingly are terminated with ethylenically unsaturated group of modifier at one end of the polymer chain and a hydroxyl group at the other end.












Similar content being viewed by others
References
Kobayashi, S. SaegusaT. (1986) Encyclopedia of Polymer Science and Engineering; Wiley: New York; Vol. 4, pp 525–537
Saegusa, T. Kobayashi S. (1975).In Macromolecular Science, International Review of Science, Physical Chemistry; Butterworth: London, Series 2, Vol. 8 Chapter 4
Kagiya T, Narisawa S, Maeda T, Fukui K (1966) Ring-opening polymerization of 2-substituted 2-oxazolines. Polym Lett 4(7):441–445
Tomalia, D.A., Killat G. R. (1985) Alkyleneimine Polymers. Encyclopedia of Polymer Science and Engineering, 2nd ed., Vol. 1, Wiley-Interscience, New York, pp 680–739
Saegusa T, Chujo Y, Aoi K, Miyamoto M (1990) One-shot block copolymerization. Macromol Chem Macromol Symp 32(1):1–10
Miyamoto M, Aoi K, Saegusa T (1989) Poly [(acylimino) alkylene] block copolymers having perfluoroalkyl hydrophobic blocks: synthesis and surfactant properties. Macromolecules 22(9):3540–3543
Kagiya T, Matsuda M, Nakato R, Hirata J (1972) Polymerization of 2-oxazolines. IV. The structure and the reactivity of 2-substituted-2-oxazolines and oxazolinium perchlorates. Macromol Sci Chem 6(8):1631–1652
Saegusa T, Kobayashi S, Yamada A (1976) Kinetics and mechanism of the isomerization polymerization of 2-methyl-2-oxazoline by benzyl chloride and bromide initiators. Effect of halogen counteranions. Makromol Chem 177(8):2271–2283
Gherras H, Hachemaoui A, Yahiaoui A, Belbachir M, Belfedal A (2012) Chemical synthesis and characterization of a new soluble conducting polymer. Synth Met 162(19–20):1750–1755
Belmokhtar A, Yahiaoui A, Hachemaoui A, Belbachir M (2013) Synthesis and characterization of Zn phtalocyanine (ZnPc) using maghnite-Zn as clay catalyst. Key Eng Mater 553:111–115
Thomas CL, Hickey J, Stecker G (1950) Chemistry of clay cracking catalysts. Ind Eng Chem 42:866–871
Hojabri F (1971) Gas-phase catalytic alkylation of aromatic hydrocarbons. J Appl Chem Biotechnol 21(3):87–89
Mahdavinia GR, Massoudi A, Baghban A, Massoumi B (2012) Novel Carrageenan-based hydrogel nanocomposites containing laponite RD and their application to remove cationic dye. Iran Polym J 21(9):609–619
Qinghong K, Yuwen T, Yuan H, Le S, Hong L, Lixia L (2012) Thermal stability and flame retardance properties of acrylonitrile-butadiene-styrene/polyvinyl chloride/organophilic Fe-montmorillonite nanocomposites. J Polym Res 19(1):1–10
Ballantine JA, Davies M, Purnell H (1981) Chemical conversions using sheet silicates: novel interlamellar dehydrations of alcohols to ethers and polymers. J Chem Soc Chem Comm 9:427
Yahiaoui A, Belbachir M, Soutif JC, Fontaine L (2005) Synthesis and structural analyses of POLY (1, 2-cyclohexene oxide) over solid acid catalyst. Mater Lett 59:759–767
Chabani M, Yahiaoui A, Hachemaoui A, Belbachir M (2011) New approach for the polymerization of 2-chloroethyl vinyl ether using a maghnite clay as Eco-catalyst. J App Polym Sci 122:1800–1806
Shokoufeh H, Majid SJ, Mohammad EZ (2013) Polyethylene nanocomposite prepared by a metallocene catalyst supported on MMT using a new pretreatment method. J Polym Res 20(129)
Hachemaoui A, Belbachir M (2005) Ring-opening polymerization of lactones catalyzed by Ion- exchanged clay montmorillonite and the application to well-defined block copolymer synthesis with seven-membered cyclic carbonate. Mater Lett 59:3904–3908
Hachemaoui A, Yahiaoui A, Belbachir M (2010) Structural and photooxidation studies of poly (styrene oxide) prepared with maghnite-H + as cationic catalyst. J App Polym Sci 118:3445–3452
Breen C, Madejovà J, Komadel P (1995) Characterisation of moderately acid-treated, size-fractionated montmorillonites using IR and MAS NMR spectroscopy and thermal analysis J. Mater Chem 5:469–474
Madejovà J, Komadel P (1997) Characterization and catalytic activ- ity of acid-treated, size-fractionated smectites. J Phys Chem B 27:5324–5331
Njopwouo D, Roques G, Wandji R (1987) A contribution to the study of the catalytic action of clays on the polymerization of styrene: I. Characterization of polystyrene. Clay Miner 22(2):45
Kadokawa J, Iwasaki Y, Tagaya H (2002) Ring-opening polymerization of lactones catalyzed by ion-exchanged clay montmorillonite. Green Chem 4(1):14–16
Crivello JV, Fan M (1992) Regioselective rhodium-containing catalysts for ring-opening polymerizations and hydrosilylations. J Polym Sci A 30(1):1–11
Inoue S, Aida T (1984) In: ring–opening polymerization, Vol. 1; ivin K J, sagusa T. Ed. Applied Science Publishers, New York, NY
Goethals A (1976) Cyclic oligomers in the cationic polymerization of heterocycles. Pure Appl Chem 48(3):335–341
Okamoto K.MALDI Mass Spectrometry of Synthetic Polymers. R&D Review of Toyota CRDL Vol. 41 No. 3
Acknowledgments
This work was supported by The National Agency for the Development of University Research (ATRST), the Directorate General of Scientific Research and Technological Development (DGRSDT) of Algeria.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghillal, Y., Hachemaoui, A., Benkouider, A.M. et al. Synthesis and caracterization of macromonomers of poly (2-methyl-2-oxazoline)-allyl by an acid exchanged clay as eco-catalyst. J Polym Res 21, 528 (2014). https://doi.org/10.1007/s10965-014-0528-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-014-0528-y