Abstract
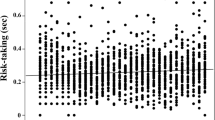
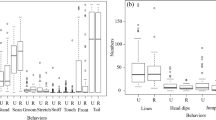
Females of the malaria mosquito, Anopheles gambiae, predominantly obtain blood meals within human dwellings. Being highly anthropophilic, human skin odor offers a reliable, host-specific cue, but the challenge posed by pervasive human odor found indoors from used clothing, bedding etc. remains unclear. Anopheles gambiae spends much of its adult life indoors, constantly exposed to human odor even when dwellings are unoccupied. In landing assays, we found that female mosquitoes respond very weakly to human skin odor alone, suggesting that, alone, it is an ineffective landing cue. Landing, however, was dramatically increased by addition of carbon dioxide at a range of concentrations above ambient. Indeed, this effect was seen even when carbon dioxide was just 0.015 % above ambient within the assay cage. The synergistic effect of added carbon dioxide quickly waned, thereby facilitating a highly adaptive “sit-and-wait” ambush strategy, wherein females ignore persistent human odor until a living human is present. Unexpectedly, landing rates in the presence of added carbon dioxide were almost as robust during daytime, when An. gambiae has previously been assumed inactive, possibly facilitating opportunistic feeding at times of day when human dwellings are occupied intermittently. We suggest earlier studies that showed strong upwind flight behavior toward human odor alone could, in fact, have been demonstrating orientation toward a human dwelling rather than toward a living human. This new interpretation of how human odors mediate upwind orientation and landing in An. gambiae is discussed.



Similar content being viewed by others
References
Bruce TJA, Pickett JA (2011) Perception of plant volatile blends by herbivorous insects - finding the right mix. Phytochemistry 72:1605–1611. doi:10.1016/j.phytochem.2011.04.011
Cardé RT, Gibson G (2010) Host finding by female mosquitoes: mechanisms of orientation to host odours and other cues. In: Takken W, Knols BGJ (eds) Olfaction in vector-host interactions. Wageningen Academic Publishers, Wageningen, pp 115–142. doi:10.3920/978-90-8686-698-4
Dekker T, Geier M, Cardé RT (2005) Carbon dioxide instantly sensitizes female yellow fever mosquitoes to human skin odours. J Exp Biol 208:2963–2972. doi:10.1242/jeb.01736
Development Core Team (2012) R: a language and environment for statistical computing, 2.15.0 edn. R Foundation for Statistical Computing, Vienna
Faye O, Konate L, Mouchet J, Fontenille D, Sy N, Hebrard G, Herve JP (1997) Indoor resting by outdoor biting females of Anopheles gambiae complex (Diptera: Culicidae) in the sahel of northern Senegal. J Med Entomol 34:285–289
Gillies MT (1954) Studies of house leaving and outside resting of Anopheles gambiae Giles and Anopheles funestus Giles in East Africa. I.—The outside resting population. Bull Entomol Res 45:361–373. doi:10.1017/S0007485300027188
Gillies MT (1980) The role of carbon dioxide in host-finding by mosquitos (Diptera: Culicidae): a review. Bull Entomol Res 70:525–32. doi:10.1017/S0007485300007811
Haddow AJ (1942) The mosquito fauna and climate of native huts at Kisumu, Kenya. Bull Entomol Res 33:91–142. doi:10.1017/S0007485300026389
Healy TP, Copland MJW (1995) Activation of Anopheles gambiae mosquitoes by carbon dioxide and human breath. Med Vet Entomol 9:331–336. doi:10.1111/j.1365-2915.1995.tb00143.x
Jones MDR, Gubbins SJ (1978) Changes in circadian flight activity of mosquito Anopheles gambiae in relation to insemination, feeding and oviposition. Physiol Entomol 3:213–220. doi:10.1111/j.1365-3032.1978.tb00151.x
Klowden MJ (2007) Making generalizations about vectors: is there a physiology of “the mosquito”? Entomol Res 37:1–13. doi:10.1111/j.1748-5967.2007.00044.x
Krober T, Kessler S, Frei J, Bourquin M, Guerin PM (2010) An in vitro assay for testing mosquito repellents employing a warm body and carbon dioxide as a behavioral activator. J Am Mosq Control Assoc 26:381–386. doi:10.2987/10-6044.1
Lacey ES, Cardé RT (2011) Activation, orientation and landing of female Culex quinquefasciatus in response to carbon dioxide and odour from human feet: 3D flight analysis in a wind tunnel. Med Vet Entomol 25:94–103. doi:10.1111/j.1365-2915.2010.00921.x
Lacey ES, Ray A, Cardé RT (2014) Close encounters: contributions of carbon dioxide and human skin odour to finding and landing on a host in Aedes aegypti. Physiol Entomol 39:60–68. doi:10.1111/phen.12048
Logan JG, Stanczyk NM, Hassanali A, Kemei J, Santana AEG, Ribeiro KAL, Pickett JA, Mordue J (2010) Arm-in-cage testing of natural human-derived mosquito repellents. Malar J 9:239. doi:10.1186/1475-2875-9-239
Lorenz LM et al (2013) Taxis assays measure directional movement of mosquitoes to olfactory cues. Parasite Vector 6:131. doi:10.1186/1756-3305-6-131
Manda H, Gouagna LC, Foster WA, Jackson RJ, Beier JC, Githure JI, Hassanali A (2007) Effect of discriminative plant-sugar feeding on the survival and fecundity of Anopheles gambiae. Malar J 6:113. doi:10.1186/1475-2875-6-113
McMeniman CJ, Corfas RA, Matthews BJ, Ritchie SA, Vosshall LB (2014) Multimodal integration of carbon dioxide and other sensory cues drives mosquito attraction to humans. Cell 156:1060–1071. doi:10.1016/j.cell.2013.12.044
Menger DJ, Van Loon JJA, Takken W (2014) Assessing the efficacy of candidate mosquito repellents against the background of an attractive source that mimics a human host. Med Vet Entomol 28:407–413. doi:10.1111/mve.12061
Moiroux N, Gomez MB, Pennetier C, Elanga E, Djènontin A, Chandre F, Djègbé I, Guis H, Corbel V (2012) Changes in Anopheles funestus biting behavior following universal coverage of long-lasting insecticidal nets in Benin. J Infect Dis 206:1622–1629. doi:10.1093/infdis/jis565
Molina R, Benito A, Blanca F, Roche J, Otunga B, Alvar J (1996) The anophelines of Equatorial Guinea. Ethology and susceptibility studies. Res Rev Parasitol 56:105–110
Müller GC, Beier JC, Traore SF, Toure MB, Traore MM, Bah S, Doumbia S, Schlein Y (2010) Field experiments of Anopheles gambiae attraction to local fruits/seedpods and flowering plants in Mali to optimize strategies for malaria vector control in Africa using attractive toxic sugar bait methods. Malar J 9:262. doi:10.1186/1475-2875-9-262
Muturi E, Muriu S, Shililu J, Mwangangi JM, Jacob BG, Mbogo C, Githure J, Novak RJ (2008) Blood-feeding patterns of Culex quinquefasciatus and other culicines and implications for disease transmission in Mwea rice scheme, Kenya. Parasitol Res 102:1329–1335. doi:10.1007/s00436-008-0914-7
Njie M, Dilger E, Lindsay SW, Kirby MJ (2009) Importance of eaves to house entry by Anopheline, but not Culicine, mosquitoes. J Med Entomol 46:505–510. doi:10.1603/033.046.0314
Njiru BN, Mukabana WR, Takken W, Knols BGJ (2006) Trapping of the malaria vector Anopheles gambiae with odour-baited MM-X traps in semi-field conditions in western Kenya. Malar J 5:1–8. doi:10.1186/1475-2875-5-39
Paaijmans KP, Thomas MB (2011) The influence of mosquito resting behaviour and associated microclimate for malaria risk. Malar J 10:183. doi:10.1186/1475-2875-10-183
Pates HV, Takken W, Stuke K, Curtis CF (2001) Differential behaviour of Anopheles gambiae sensu stricto (Diptera : Culicidae) to human and cow odours in the laboratory. Bull Entomol Res 91:289–296. doi:10.1079/ber200198
Qiu YT, Smallegange RC, Van Loon JJA, Ter Braak CJF, Takken W (2006) Interindividual variation in the attractiveness of human odours to the malaria mosquito Anopheles gambiae s. s. Med Vet Entomol 20:280–287. doi:10.1111/j.1365-2915.2006.00627.x
Qiu YT et al (2007) Attractiveness of MM-X traps baited with human or synthetic odor to mosquitoes (Diptera : Culicidae) in the Gambia. J Med Entomol 44:970–983. doi:10.1603/0022-2585(2007)44[970:aomtbw]2.0.co;2
Reddy MR, Overgaard HJ, Abaga S, Reddy VP, Caccone A, Kiszewski AE, Slotman MA (2011) Outdoor host seeking behaviour of Anopheles gambiae mosquitoes following initiation of malaria vector control on Bioko Island, Equatorial Guinea. Malar J 10:184. doi:10.1186/1475-2875-10-184
Ribbands CR (1946) Moonlight and house-hauting habits of female anophelines in West Africa. Bull Entomol Res 36:395–417
Rund SSC, Bonar NA, Champion MM, Ghazi JP, Houk CM, Leming MT, Syed Z, Duffield GE (2013) Daily rhythms in antennal protein and olfactory sensitivity in the malaria mosquito Anopheles gambiae. Sci Rep 3:2494. doi:10.1038/srep02494
Smallegange RC, Schmied WH, van Roey KJ, Verhulst NO, Spitzen J, Mukabana WR, Takken W (2010) Sugar-fermenting yeast as an organic source of carbon dioxide to attract the malaria mosquito Anopheles gambiae. Malar J 9:292. doi:10.1186/1475-2875-9-292
Smallegange RC, van Gemert GJ, van de Vegte-Bolmer M, Gezan S, Takken W, Sauerwein RW, Logan JG (2013) Malaria infected mosquitoes xxpress enhanced attraction to human odor. PLoS One 8 doi:10.1371/journal.pone.0063602
Snow WF (1987) Studies of house-entering habits of mosquitos in the Gabia, West Africa: experiments with prefabricated huts with varied wall apertures. Med Vet Entomol 1:9–21. doi:10.1111/j.1365-2915.1987.tb00318.x
Spitzen J, Smallegange RC, Takken W (2008) Effect of human odours and positioning of CO2 release point on trap catches of the malaria mosquito Anopheles gambiae sensu stricto in an olfactometer. Physiol Entomol 33:116–122. doi:10.1111/j.1365-3032.2008.00612.x
Spitzen J et al (2013) A 3D analysis of flight behavior of Anopheles gambiae sensu stricto malaria mosquitoes in response to human odor and heat. PLoS ONE 8:e62995. doi:10.1371/journal.pone.0062995
Takken W, Knols BGJ (1999) Odor-mediated behavior of Afrotropical malaria mosquitoes. Annu Rev Entomol 44:131–157. doi:10.1146/annurev.ento.44.1.131
Takken W, Verhulst NO (2013) Host preferences of blood-feeding mosquitoes. Annu Rev Entomol 58:433–453. doi:10.1146/annurev-ento-120811-153618
Takken W, Dekker T, Wijnholds YG (1997) Odor-mediated flight behavior of Anopheles gambiae Giles Sensu Stricto and A.stephensi Liston in response to CO2, acetone, and 1-octen-3-ol (Diptera: Culicidae). J Insect Behav 10:395–407. doi:10.1007/bf02765606
Tauxe GM, MacWilliam D, Boyle SM, Guda T, Ray A (2013) Targeting a dual detector of skin and CO2 to modify mosquito host seeking. Cell 155:1365–1379. doi:10.1016/j.cell.2013.11.013
Thomson RCM (1948) Studies on Anopheles gambiae and A. melas in and around Lagos. Bull Entomol Res 38:527–558
Trpis M, Hausermann W (1978) Genetics of house-entering behavior in East-African populations of Aedes aegypti (L) (Diptera:Culicidae) and its relevance to speciation. Bull Ent Res 68:521–532
Tuno N, Kjaerandsen J, Badu K, Kruppa T (2010) Blood-feeding behavior of Anopheles gambiae and Anopheles melas in Ghana, Western Africa. J Med Entomol 47:28–31. doi:10.1603/033.047.0104
Turissini DA, Gamez S, White BJ (2014) Genome-wide patterns of polymorphism in an inbred line of the African malaria mosquito Anopheles gambiae. Genome Biol Evol. doi:10.1093/gbe/evu243
Turner SL, Li N, Guda T, Githure J, Cardé RT, Ray A (2011) Ultra-prolonged activation of CO2-sensing neurons disorients mosquitoes. Nature 474:87–U114. doi:10.1038/nature10081
Webster B, Bruce T, Pickett J, Hardie J (2010) Volatiles functioning as host cues in a blend become nonhost cues when presented alone to the black bean aphid. Anim Behav 79:451–457. doi:10.1016/j.anbehav.2009.11.028
Acknowledgments
This work was funded by an R56AI099778 (National Institute of Allergy and Infectious Diseases) grant to Anandasankar Ray and Ring Cardé. The granting agencies had no role in experimental design or analysis. We are grateful to Anandasankar Ray for his comments and Bradley White for our colony of An. gambiae.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Webster, B., Lacey, E.S. & Cardé, R.T. Waiting with Bated Breath: Opportunistic Orientation to Human Odor in the Malaria Mosquito, Anopheles gambiae, is Modulated by Minute Changes in Carbon Dioxide Concentration. J Chem Ecol 41, 59–66 (2015). https://doi.org/10.1007/s10886-014-0542-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-014-0542-x