Abstract
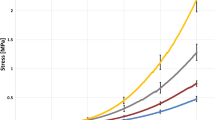
The cornea is a protective transparent connective tissue covering the front of the eye. The standard uniaxial tensile experiments are among the most popular techniques for investigating biomechanical properties of the cornea. This experimental method characterizes the stress–strain response of corneal strips immersed in a bathing solution. In the present study, the important roles of corneal hydration on tensile viscoelastic properties were investigated. The thickness was used as a surrogate for hydration and uniaxial tensile experiments were performed on bovine corneal samples with four different average thickness (hydration), i.e., 1100 μm (4.87 mg water/mg dry tissue), 900 μm (4.13 mg water/mg dry tissue), 700 μm (3.20 mg water/mg dry tissue), and 500 μm (1.95 mg water/mg dry tissue). The samples were immersed in mineral oil in order to prevent their swelling during the experiments. A quasilinear viscoelastic (QLV) model was used to analyze the experimental measurements and determine viscoelastic material constants. It was observed that both maximum and equilibrium (relaxed) stresses were exponentially increased with decreasing tissue thickness (hydration). Furthermore, the QLV model successfully captured the corneal viscoelastic response with an average R 2 value greater than 0.99. Additional experiments were conducted in OBSS in order to confirm that these significant changes in viscoelastic properties were because of corneal hydration and not the bathing solution. The findings of this study suggest that extra care must be taken in interpreting the results of earlier uniaxial tensile testings and their correspondence to the corneal biomechanical properties.






Similar content being viewed by others
References
Abramowitch, S. D., and S. L. Woo. An improved method to analyze the stress relaxation of ligaments following a finite ramp time based on the quasi-linear viscoelastic theory. J. Biomech. Eng. 126:92–97, 2004.
Abramowitch, S. D., S. L. Woo, T. D. Clineff, and R. E. Debski. An evaluation of the quasi-linear viscoelastic properties of the healing medial collateral ligament in a goat model. Ann. Biomed. Eng. 32:329–335, 2004.
Anderson, K., A. El-Sheikh, and T. Newson. Application of structural analysis to the mechanical behaviour of the cornea. J. R. Soc. Lond. Interface 1:3–15, 2004.
Boschetti, F., V. Triacca, L. Spinelli, and A. Pandolfi. Mechanical characterization of porcine corneas. J. Biomech. Eng. 134:031003–031009, 2012.
Boyce, B. L., J. M. Grazier, R. E. Jones, and T. D. Nguyen. Full-field deformation of bovine cornea under constrained inflation conditions. Biomaterials 29:3896–3904, 2008.
Boyce, B. L., R. E. Jones, T. D. Nguyen, and J. M. Grazier. Stress-controlled viscoelastic tensile response of bovine cornea. J. Biomech. 40:2367–2376, 2007.
Carew, E. O., A. Garg, J. E. Barber, and I. Vesely. Stress relaxation preconditioning of porcine aortic valves. Ann. Biomed. Eng. 32:563–572, 2004.
Cheng, X., H. Hatami-Marbini, and P. M. Pinsky. Modeling collagen-proteoglycan structural interactions in the human cornea. In: Computer Models in Biomechanics, edited by G. A. Holzapfel, and E. Kuhl. Heidelberg: Springer, 2013, pp. 11–24.
Doehring, T. C., E. O. Carew, and I. Vesely. The effect of strain rate on the viscoelastic response of aortic valve tissue: a direct-fit approach. Ann. Biomed. Eng. 32:223–232, 2004.
Elsheikh, A., and D. Alhasso. Mechanical anisotropy of porcine cornea and correlation with stromal microstructure. Exp. Eye Res. 88:1084–1091, 2009.
Elsheikh, A., D. Alhasso, and P. Rama. Biomechanical properties of human and porcine corneas. Exp. Eye Res. 86:783–790, 2008.
Elsheikh, A., and K. Anderson. Comparative study of corneal strip extensometry and inflation tests. Journal of The Royal Society Interface 2:177–185, 2005.
Elsheikh, A., M. Brown, D. Alhasso, P. Rama, M. Campanelli, and D. Garway-Heath. Experimental assessment of corneal anisotropy. J. Refract. Surg. 24:178–187, 2008.
Elsheikh, A., W. Kassem, and S. W. Jones. Strain-rate sensitivity of porcine and ovine corneas. Acta Bioeng. Biomech. 13:25–36, 2011.
Elsheikh, A., D. Wang, and D. Pye. Determination of the modulus of elasticity of the human cornea. J. Refract. Surg. 23:808–818, 2007.
Fricke, T. R., B. A. Holden, D. A. Wilson, et al. Global cost of correcting vision impairment from uncorrected refractive error. Bull. World Health Organ. 90:728–738, 2012.
Fung, Y. C. Biomechanics: Mechanical Properties of Living Tissues (2nd ed.). New York: Springer, 1993.
Han, L., D. Dean, L. A. Daher, A. J. Grodzinsky, and C. Ortiz. Cartilage aggrecan can undergo self-adhesion. Biophys. J. 95:4862–4870, 2008.
Hatami-Marbini, H., and E. Etebu. An experimental and theoretical analysis of unconfined compression of corneal stroma. J. Biomech. 46:1752–1758, 2013.
Hatami-Marbini, H., and E. Etebu. Hydration dependent biomechanical properties of the corneal stroma. Exp. Eye Res. 116:47–54, 2013.
Hatami-Marbini, H., and E. Etebu. A New Method to Determine Rate-dependent Material Parameters of Corneal Extracellular Matrix. Ann. Biomed. Eng. 41:2399–2408, 2013.
Hatami-Marbini, H., E. Etebu, and A. Rahimi. Swelling Pressure and Hydration Behavior of Porcine Corneal Stroma. Curr. Eye Res. 38:1124–1132, 2013.
Hatami-Marbini, H., and P. M. Pinsky. On mechanics of connective tissue: assessing the electrostatic contribution to corneal stroma elasticity. In: Material Research Society Proceedings, Boston, 2009, p. 1239.
Hatami-Marbini, H., and A. Rahimi. Effects of bathing solution on tensile properties of the cornea. Exp. Eye Res. 120:103–108, 2014.
Hedbys, B. O., and C. H. Dohlman. A new method for the determination of the swelling pressure of the corneal stroma in vitro. Exp. Eye Res. 2:122–129, 1963.
Hedbys, B. O., and S. Mishima. The thickness-hydration relationship of the cornea. Exp. Eye Res. 5:221–228, 1966.
Hodson, S. Why the cornea Swells. J. Theor. Biol. 33:419–427, 1971.
Hodson, S. A. Corneal stromal swelling. Prog. Retin. Eye Res. 16:99–116, 1997.
Hoeltzel, D. A., P. Altman, K. Buzard, and K.-I. Choe. Strip Extensiometry for Comparison of the Mechanical Response of Bovine, Rabbit, and Human Corneas. J. Biomech. Eng. 114:202–215, 1992.
Howland, H. C., R. H. Rand, and S. R. Lubkin. A thin-shell model of the cornea and its application to corneal surgery. Refract. Corneal Surg. 8:183–186, 1992.
Iatridis, J. C., L. A. Setton, M. Weidenbaum, and V. C. Mow. The viscoelastic behavior of the non-degenerate human lumbar nucleus pulposus in shear. J. Biomech. 30:1005–1013, 1997.
Jue, B., and D. M. Maurice. The mechanical properties of the rabbit and human cornea. J. Biomech. 19:847–853, 1986.
Kampmeier, J., B. Radt, R. Birngruber, and R. Brinkmann. Thermal and Biomechanical Parameters of Porcine Cornea. Cornea 19:355–363, 2000.
Kim, W., A. Argento, F. W. Rozsa, and K. Mallett. Constitutive behavior of ocular tissues over a range of strain rates. J. Biomech. Eng. 134:061002-061002, 2012.
Lari, D. R., D. S. Schultz, A. S. Wang, O. T. Lee, and J. M. Stewart. Scleral mechanics: comparing whole globe inflation and uniaxial testing. Exp. Eye Res. 94:128–135, 2012.
Lee, R. E., and P. F. Davison. The collagens of the developing bovine cornea. Exp. Eye Res. 39:639–652, 1984.
Lewis, P. N., C. Pinali, R. D. Young, K. M. Meek, A. J. Quantock, and C. Knupp. Structural Interactions between Collagen and Proteoglycans Are Elucidated by Three-Dimensional Electron Tomography of Bovine Cornea. Structure 18:239–245, 2010.
Maurice, D. M. The cornea and sclera. In: The Eye, edited by H. Davason. London: Academic Press, pp. 1–184, 1984.
Meek, K. M. The cornea and sclera. In: Collagen: Structure and Mechanics, edited by P. Fratzl. New York: Springer, pp. 359–396, 2008.
Meek, K. M., N. J. Fullwood, P. H. Cooke, et al. Synchrotron x-ray diffraction studies of the cornea, with implications for stromal hydration. Biophys. J. 60:467–474, 1991.
Nigul, I., and U. Nigul. On algorithms of evaluation of Fung’s relaxation function parameters. J Biomech 20:343–352, 1987.
Nyquist, G. W. Rheology of the cornea: experimental techniques and results. Exp. Eye Res. 7:183–188, 1968.
Olsen, T., and S. Sperling. The swelling pressure of the human corneal stroma as determined by a new method. Exp. Eye Res. 44:481–490, 1987.
Pandolfi, A. Computational biomechanics of the human cornea. In: Computational Modeling in Biomechanics, edited by S. De, F. GuilaK, and M. Mofrad. New York: Springer, pp. 435–466, 2010.
Pandolfi, A., and G. A. Holzapfel. Three-dimensional modeling and computational analysis of the human cornea considering distributed collagen fibril orientations. J. Biomech. Eng. 130:061006-061001-061006-061012, 2008.
Provenzano, P. P., R. S. Lakes, D. T. Corr, and R. Vanderby, Jr. Application of nonlinear viscoelastic models to describe ligament behavior. Biomech Model Mechanobiol 1:45–57, 2002.
Scott, J. E. Proteoglycan: collagen interactions and corneal ultrastructure. Biochem. Soc. Trans 19:877–881, 1991.
Scott, J. E. Morphometry of cupromeronic blue-stained proteoglycan molecules in animal corneas, versus that of purified proteoglycans stained in vitro, implies that tertiary structures contribute to corneal ultrastructure. J. Anat. 180:155–164, 1992.
Scott, J. E., and T. R. Bosworth. A comparative biochemical and ultrastructural study of proteoglycan-collagen interactions in corneal stroma - functional and metabolic implications. Biochem. J. 270:491–497, 1990.
Smith, T. S. T., K. D. Frick, B. A. Holden, T. R. Fricke, and K. S. Naidoo. Potential lost productivity resulting from the global burden of uncorrected refractive errors. Bull. World Health Organ. 87:431–437, 2009.
Sverdlik, A., and Y. Lanir. Time-dependent mechanical behavior of sheep digital tendons, including the effects of preconditioning. Journal of Biomechanical Engineering-Transactions of the Asme 124:78–84, 2002.
Wollensak, G., E. Spoerl, and T. Seiler. Stress-strain measurements of human and porcine corneas after riboflavin–ultraviolet-A-induced cross-linking. J. Cataract Refract. Surg. 29:1780–1785, 2003.
Woo, S. L., M. A. Gomez, and W. H. Akeson. The time and history-dependent viscoelastic properties of the canine medical collateral ligament. J. Biomech. Eng. 103:293–298, 1981.
Woo, S. L., A. S. Kobayashi, W. A. Schlegel, and C. Lawrence. Nonlinear material properties of intact cornea and sclera. Exp. Eye Res. 14:29–39, 1972.
Zeng, Y., J. Yang, K. Huang, Z. Lee, and X. Lee. A comparison of biomechanical properties between human and porcine cornea. J. Biomech. 34:533–537, 2001.
Acknowledgments
This project has been funded in whole or in part with the start-up fund from Oklahoma State University. The author would like to thank the members of computational biomechanics laboratory.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Dan Elson oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Hatami-Marbini, H. Hydration Dependent Viscoelastic Tensile Behavior of Cornea. Ann Biomed Eng 42, 1740–1748 (2014). https://doi.org/10.1007/s10439-014-0996-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-014-0996-6