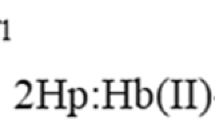Abstract
Haptoglobin (Hp) sequesters hemoglobin (Hb) preventing the Hb-based damage occurring upon its physiological release into plasma. Here, reductive nitrosylation of ferric human hemoglobin [Hb(III)] bound to human haptoglobin (Hp) 1-1 and 2-2 [Hp1-1:Hb(III) and Hp2-2:Hb(III), respectively] has been investigated between pH 7.5 and 9.5, at T=20.0 °C. Over the whole pH range explored, only one process is detected reflecting NO binding to Hp1-1:Hb(III) and Hp2-2:Hb(III). Values of the pseudo-first-order rate constant for Hp1-1:Hb(III) and Hp2-2:Hb(III) nitrosylation (k) do not depend linearly on the ligand concentration but tend to level off. The conversion of Hp1-1:Hb(III)-NO to Hp1-1:Hb(II)-NO and of Hp2-2:Hb(III)-NO to Hp2-2:Hb(II)-NO is limited by the OH−- and H2O-based catalysis. In fact, bimolecular NO binding to Hp1-1:Hb(III), Hp2-2:Hb(III), Hp1-1:Hb(II), and Hp2-2:Hb(II) proceeds very rapidly. The analysis of data allowed to determine the values of the dissociation equilibrium constant for Hp1-1:Hb(III) and Hp2-2:Hb(III) nitrosylation [K = (1.2 ± 0.1) × 10−4 M], which is pH-independent, and of the first-order rate constant for Hp1-1:Hb(III) and Hp2-2:Hb(III) conversion to Hp1-1:Hb(II)-NO and Hp2-2:Hb(II)-NO, respectively (k′). From the dependence of k′ on [OH−], values of hOH– [(4.9 ± 0.6) × 103 M−1 s−1 and (6.79 ± 0.7) × 103 M−1 s−1, respectively] and of \( h_{{{\text{H}}_{ 2} {\text{O}}}} \) [(2.6 ± 0.3) × 10−3 s−1] were determined. Values of kinetic and thermodynamic parameters for Hp1-1:Hb(III) and Hp2-2:Hb(III) reductive nitrosylation match well with those of the Hb R-state, which is typical of the αβ dimers of Hb bound to Hp.




Similar content being viewed by others
Abbreviations
- CCP:
-
Complement control protein
- Hb:
-
Hemoglobin
- Hb(II):
-
Ferrous Hb
- Hb(III):
-
Ferric Hb
- Hb(II)-NO:
-
Nitrosylated Hb
- Hp:
-
Haptoglobin
- Hp1-1:
-
Phenotype 1-1 of Hp
- Hp2-2:
-
Phenotype 2-2 of Hp
- Hp1-1:Hb:
-
Hp1-1:Hb complex
- Hp1:Hb(II):
-
Hp1-1:Hb(II) complex
- Hp1-1:Hb(III):
-
Hp1-1:Hb(III) complex
- Hp1-1:Hb(II)-NO:
-
Hp1-1:Hb(II)-NO complex
- Hp2-2:Hb:
-
Hp2-2:Hb complex
- Hp2-2:Hb(II):
-
Hp2-2Hb(II) complex
- Hp2-2:Hb(III):
-
Hp2-2:Hb(III) complex
- Hp1-1:Hb(II)-NO:
-
Hp1-1:Hb(II)-NO complex
- Mb:
-
Myoglobin
- Mb(III):
-
Ferric Mb
- Ngb:
-
Neuroglobin
- SP-like domain:
-
Serine protease-like domain
References
Bunn HF, Forget BG (1986) Hemoglobin: molecular, genetic and clinical aspects. WB Saunders Company, Philadelphia
Perutz MF (1990) Annu Rev Physiol 52:1–25
Gow AJ, Luchsinger BP, Pawloski JR, Singel DJ, Stamler JS (1999) Proc Natl Acad Sci USA 96:9027–9032
Imai K (1999) Nature 401:437–439
McMahon TJ, Moon RE, Luschinger BP, Carraway MS, Stone AE, Stolp BW, Gow AJ, Pawloski JR, Watke P, Singel DJ, Piantadosi CA, Stamler JS (2002) Nat Med 8:711–717
Ascenzi P, Brunori M (2016) J Porphyrins Phthalocyanines 20:134–149
Ascenzi P, Bocedi A, Visca P, Altruda F, Tolosano E, Beringhelli T, Fasano M (2005) IUBMB Life 57:749–759
Smith A, McCulloh RJ (2015) Front Physiol 6:187
Andersen CBF, Stødkilde K, Sæderup KL, Kuhlee A, Raunser S, Graversen JH, Moestrup SK (2017) Antioxid Redox Signal 26:814–831
Wang Y, Kinzie E, Berger FG, Lim SK, Baumann H (2001) Redox Rep 6:379–385
Polticelli F, Bocedi A, Minervini G, Ascenzi P (2008) FEBS J 275:5648–5656
Andersen CB, Torvund-Jensen M, Nielsen MJ, de Oliveira CL, Hersleth HP, Andersen NH, Pedersen JS, Andersen GR, Moestrup SK (2012) Nature 489:456–459
Stødkilde K, Torvund-Jensen M, Moestrup SK, Andersen CB (2014) Nat Commun 5:5487
Kristiansen M, Graversen JH, Jacobsen C, Sonne O, Hoffman HJ, Law SK, Moestrup SK (2001) Nature 409:198–201
Nagel RL, Gibson QH (1966) J Mol Biol 22:249–255
Chiancone E, Antonini E, Brunori M, Alfsen A, Lavialle F (1973) Biochem J 133:205–207
Azarov I, He X, Jeffers A, Basu S, Ucer B, Hantgan RR, Levy A, Kim-Shapiro DB (2008) Nitric Oxide 18:296–302
Antonini E, Brunori M (1971) Hemoglobin and myoglobin in their reactions with ligands. North-Holland Publishing Co, Amsterdam
Brunori M, Alfsen A, Saggese U, Antonini E, Wyman J (1968) J Biol Chem 243:2950–2954
Nagel RL, Whittenberg JB, Ranney HM (1965) Biochim Biophys Acta 100:286–289
Chiancone E, Wittenberg JB, Wittenberg BA, Antonini E, Wyman J (1966) Biochim Biophys Acta 117:379–386
Ascenzi P, Brunori M, Pennesi G, Ercolani C, Monacelli F (1987) J Chem Soc Dalton Trans 369–371
Hoshino M, Ozawa K, Seki H, Ford PC (1993) J Am Chem Soc 115:9568–9575
Hoshino M, Maeda M, Konishi R, Seki H, Ford PC (1996) J Am Chem Soc 118:5702–5707
Boffi A, Sarti P, Amiconi G, Chiancone E (2002) Biophys Chem 98:209–216
Herold S, Fago A, Weber RE, Dewilde S, Moens L (2004) J Biol Chem 279:22841–22847
Herold S, Puppo A (2005) J Biol Inorg Chem 10:946–957
Ascenzi P, Bocedi A, Antonini G, Bolognesi M, Fasano M (2007) FEBS J 274:551–562
Ascenzi P, di Masi A, Gullotta F, Mattu M, Ciaccio C, Coletta M (2010) Biochem Biophys Res Commun 393:196–200
Ascenzi P, Yu C, di Masi A, Gullotta F, De Sanctis G, Fanali G, Fasano M, Coletta M (2010) FEBS J 277:2474–2485
Ascenzi P, Pesce A, Nardini M, Bolognesi M, Ciaccio C, Coletta M, Dewilde S (2013) Biochem Biophys Res Commun 430:1301–1305
Ascenzi P, Marino M, Ciaccio C, Santucci R, Coletta M (2014) IUBMB Life 66:438–447
Ascenzi P, di Masi A, Tundo GR, Pesce A, Visca P, Coletta M (2014) PLoS One 9:e102811
Ascenzi P, Bocedi A, Gioia M, Fanali G, Fasano M, Coletta M (2017) J Inorg Biochem 177:63–75
Ascenzi P, Ciaccio C, De Simone G, Santucci R, Coletta M (2017) J Porphyrins Phthalocyanines 21:1–9
Antonini E, Ascenzi P, Menegatti E, Guarneri M (1983) Biopolymers 22:363–375
Bolli A, Ciaccio C, Coletta M, Nardini M, Bolognesi M, Pesce A, Guertin M, Visca P, Ascenzi P (2008) FEBS J 275:633–645
Moore EG, Gibson QH (1976) J Biol Chem 251:2788–2794
Perutz MF (1979) Annu Rev Biochem 48:327–386
Perutz MF (1989) Trends Biochem Sci 14:42–44
Bolognesi M, Bordo D, Rizzi M, Tarricone C, Ascenzi P (1997) Prog Biophys Mol Biol 68:29–68
Pesce A, Dewilde S, Nardini M, Moens L, Ascenzi P, Hankeln T, Burmester T, Bolognesi M (2003) Structure 11:1087–1095
Hamdane D, Kiger L, Dewilde S, Green BN, Pesce A, Uzan J, Burmester T, Hankeln T, Bolognesi M, Moens L, Marden MC (2003) J Biol Chem 278:51713–51721
Brucker EA, Olson JS, Ikeda-Saito M, Phillips GN Jr (1998) Proteins 30:352–356
Beetlestone JG, Adeosun OS, Goddard JE, Kushimo JB, Ogunlesi MM, Ogunmola GB, Okonjo KO, Seamonds B (1976) J Chem Soc Dalton Trans 1251–1278
Miele AE, Santanché S, Travaglini-Allocatelli C, Vallone B, Brunori M, Bellelli A (1999) J Mol Biol 290:515–524
Giacometti GM, Ascenzi P, Bolognesi M, Brunori M (1981) J Mol Biol 146:363–374
Antonini E, Ascenzi P, Bolognesi M, Menegatti E, Guarneri M (1983) J Biol Chem 258:4676–4678
Ascenzi P, Gianni S (2013) IUBMB Life 65:836–844
Acknowledgements
The grant of Excellence Departments, MIUR (Legge 232/2016, Articolo 1, Comma 314-337), is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ascenzi, P., De Simone, G., Polticelli, F. et al. Reductive nitrosylation of ferric human hemoglobin bound to human haptoglobin 1-1 and 2-2. J Biol Inorg Chem 23, 437–445 (2018). https://doi.org/10.1007/s00775-018-1551-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-018-1551-y