Abstract
Purpose
One-lung ventilation (OLV) is commonly used during thoracic surgery. Clinical studies using bronchoalveolar lavage fluid analysis have demonstrated that OLV induces pulmonary inflammatory reactions in the ventilated dependent lung. However, few clinical studies have investigated such inflammatory reactions in the dependent lung compared with the collapsed nondependent lung. Here we used a bronchoscopic microsampling method to obtain epithelial lining fluid (ELF) from each lung, and then compared the inflammatory reactions in the dependent lung and the nondependent lung during thoracic surgery.
Methods
Twenty adult patients were studied. All patients underwent thoracic surgery using OLV. Propofol and remifentanil were used for total intravenous anesthesia. A double-lumen endotracheal tube was used to perform OLV. ELF was obtained from each lung using the bronchoscopic microsampling method. ELF levels of inflammatory mediators, tumor necrosis factor α, interleukin (IL)-1β, IL-6, IL-8, IL-10, and IL-12p70 were measured using ELISA before and after OLV.
Results
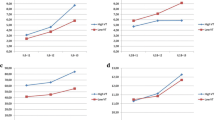
ELF levels of IL-1β, IL-6, and IL-8 were significantly increased in the dependent lung and the nondependent lung at the end of surgery compared with their baseline levels (p < 0.05). ELF level of IL-6 was significantly higher in the dependent lung than in the nondependent lung at the end of surgery (p = 0.019).
Conclusions
One-lung ventilation induced inflammatory responses of the bronchial epithelia in the dependent lung and the nondependent lung during thoracic surgery. In addition, these inflammatory responses were more augmented in the dependent lung than in the nondependent lung.



Similar content being viewed by others
References
Schilling T, Kozian A, Huth C, Bühling F, Kretzschmar M, Welte T, Hachenberg T. The pulmonary immune effects of mechanical ventilation in patients undergoing thoracic surgery. Anesth Analg. 2005;101:957–65.
De Conno E, Steurer M, Wittlinger M, Zalunardo M, Weder W, Schneiter D, Schimmer R, Klaghofer R, Neff T, Schmid E, Spahn D, Z’graggen B, Urner M, Beck-Schimmer B. Anesthetic-induced improvement of the inflammatory response to one-lung ventilation. Anesthesiology. 2009;110:1316–26.
Baudouin S. Lung injury after thoracotomy. Br J Anaesth. 2003;91:132–42.
Fernández-Pérez E, Keegan M, Brown D, Hubmayr R, Gajic O. Intraoperative tidal volume as a risk factor for respiratory failure after pneumonectomy. Anesthesiology. 2006;105:14–8.
von Bethmann A, Brasch F, Nüsing R, Vogt K, Volk H, Müller K, Wendel A, Uhlig S. Hyperventilation induces release of cytokines from perfused mouse lung. Am J Respir Crit Care Med. 1998;157:263–72.
Madjdpour C, Oertli B, Ziegler U, Bonvini J, Pasch T, Beck-Schimmer B. Lipopolysaccharide induces functional ICAM-1 expression in rat alveolar epithelial cells in vitro. Am J Physiol Lung Cell Mol Physiol. 2000;278:L572–9.
Voigtsberger S, Lachmann R, Leutert A, Schläpfer M, Booy C, Reyes L, Urner M, Schild J, Schimmer R, Beck-Schimmer B. Sevoflurane ameliorates gas exchange and attenuates lung damage in experimental lipopolysaccharide-induced lung injury. Anesthesiology. 2009;111:1238–48.
Bless NM, Huber-Lang M, Guo RF, Warner RL, Schmal H, Czermak BJ, Shanley TP, Crouch LD, Lentsch AB, Sarma V, Mulligan MS, Friedl HP, Ward PA. Role of CC chemokines (macrophage inflammatory protein-1 beta, monocyte chemoattractant protein-1, RANTES) in acute lung injury in rats. J Immunol. 2000;164:2650–9.
Vozzelli MA, Mason SN, Whorton MH, Auten RL. Antimacrophage chemokine treatment prevents neutrophil and macrophage influx in hyperoxia-exposed newborn rat lung. Am J Physiol Lung Cell Mol Physiol. 2004;286:L488–93.
Beck-Schimmer B, Schwendener R, Pasch T, Reyes L, Booy C, Schimmer RC. Alveolar macrophages regulate neutrophil recruitment in endotoxin-induced lung injury. Respir Res. 2005;6:61.
Michelet P, D’Journo X, Roch A, Doddoli C, Marin V, Papazian L, Decamps I, Bregeon F, Thomas P, Auffray J. Protective ventilation influences systemic inflammation after esophagectomy: a randomized controlled study. Anesthesiology. 2006;105:911–9.
D’Journo X, Michelet P, Marin V, Diesnis I, Blayac D, Doddoli C, Bongrand P, Thomas P. An early inflammatory response to oesophagectomy predicts the occurrence of pulmonary complications. Eur J Cardiothorac Surg. 2010;37:1144–51.
Schilling T, Kozian A, Kretzschmar M, Huth C, Welte T, Bühling F, Hedenstierna G, Hachenberg T. Effects of propofol and desflurane anaesthesia on the alveolar inflammatory response to one-lung ventilation. Br J Anaesth. 2007;99:368–75.
Ishizaka A, Watanabe M, Yamashita T, Ogawa Y, Koh H, Hasegawa N, Nakamura H, Asano K, Yamaguchi K, Kotani M, Kotani T, Morisaki H, Takeda J, Kobayashi K, Ogawa S. New bronchoscopic microsample probe to measure the biochemical constituents in epithelial lining fluid of patients with acute respiratory distress syndrome. Crit Care Med. 2001;29:896–8.
Koh H, Tasaka S, Hasegawa N, Asano K, Kotani T, Morisaki H, Takeda J, Fujishima S, Matsuda T, Hashimoto S, Ishizaka A. Vascular endothelial growth factor in epithelial lining fluid of patients with acute respiratory distress syndrome. Respirology. 2008;13:281–4.
Wilson WC, Benumof JL. Respiratory physiology and respiratory function during anesthesia. In: Miller RD, editor. Miller’s anesthesia. 6th ed. Philadelphia: Elsevier/Churchill Livingstone; 2005. p. 679–722.
Misthos P, Katsaragakis S, Theodorou D, Milingos N, Skottis I. The degree of oxidative stress is associated with major adverse effects after lung resection: a prospective study. Eur J Cardiothorac Surg. 2006;29:591–5.
Tekinbas C, Ulusoy H, Yulug E, Erol M, Alver A, Yenilmez E, Geze S, Topbas M. One-lung ventilation: for how long? J Thorac Cardiovasc Surg. 2007;134:405–10.
Galvin I, Drummond G, Nirmalan M. Distribution of blood flow and ventilation in the lung: gravity is not the only factor. Br J Anaesth. 2007;98:420–8.
Chang H, Lai-Fook S, Domino K, Schimmel C, Hildebrandt J, Robertson H, Glenny R, Hlastala M. Spatial distribution of ventilation and perfusion in anesthetized dogs in lateral postures. J Appl Physiol. 2002;92:745–62.
Halliwell B. Reactive oxygen species in living systems: source, biochemistry, and role in human disease. Am J Med. 1991;91:14S–22S.
Winter P, Smith G. The toxicity of oxygen. Anesthesiology. 1972;37:210–41.
Clark J, Lambertsen C. Pulmonary oxygen toxicity: a review. Pharmacol Rev. 1971;23:37–133.
Clark J. Pulmonary limits of oxygen tolerance in man. Exp Lung Res. 1988;14(Suppl):897–910.
Lohser J. Evidence-based management of one-lung ventilation. Anesthesiol Clin. 2008;26:241–72.
Kolobow T, Moretti M, Fumagalli R, Mascheroni D, Prato P, Chen V, Joris M. Severe impairment in lung function induced by high peak airway pressure during mechanical ventilation. An experimental study. Am Rev Respir Dis. 1987;135:312–5.
Whitehead T, Slutsky A. The pulmonary physician in critical care. 7: Ventilator induced lung injury. Thorax. 2002;57:635–42.
Kozian A, Schilling T, Fredén F, Maripuu E, Röcken C, Strang C, Hachenberg T, Hedenstierna G. One-lung ventilation induces hyperperfusion and alveolar damage in the ventilated lung: an experimental study. Br J Anaesth. 2008;100:549–59.
Kozian A, Schilling T, Schütze H, Heres F, Hachenberg T, Hedenstierna G. Lung computed tomography density distribution in a porcine model of one-lung ventilation. Br J Anaesth. 2009;102:551–60.
Wolthuis E, Choi G, Dessing M, Bresser P, Lutter R, Dzoljic M, van der Poll T, Vroom M, Hollmann M, Schultz M. Mechanical ventilation with lower tidal volumes and positive end-expiratory pressure prevents pulmonary inflammation in patients without preexisting lung injury. Anesthesiology. 2008;108:46–54.
Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The Acute Respiratory Distress Syndrome Network. N Engl J Med. 2000;342:1301–8.
Slutsky A. Mechanical ventilation. American College of Chest Physicians’ Consensus Conference. Chest. 1993;104:1833–59.
Funakoshi T, Ishibe Y, Okazaki N, Miura K, Liu R, Nagai S, Minami Y. Effect of re-expansion after short-period lung collapse on pulmonary capillary permeability and pro-inflammatory cytokine gene expression in isolated rabbit lungs. Br J Anaesth. 2004;92:558–63.
Hamvas A, Palazzo R, Kaiser L, Cooper J, Shuman T, Velazquez M, Freeman B, Schuster D. Inflammation and oxygen free radical formation during pulmonary ischemia–reperfusion injury. J Appl Physiol. 1992;72:621–8.
De Leyn P, Lerut T, Schreinemakers H, Van Raemdonck D, Mubagwa K, Flameng W. Effect of inflation on adenosine triphosphate catabolism and lactate production during normothermic lung ischemia. Ann Thorac Surg. 1993;55:1073–8.
Hamvas A, Park C, Palazzo R, Liptay M, Cooper J, Schuster D. Modifying pulmonary ischemia–reperfusion injury by altering ventilatory strategies during ischemia. J Appl Physiol. 1992;73:2112–9.
Madjdpour C, Jewell U, Kneller S, Ziegler U, Schwendener R, Booy C, Kläusli L, Pasch T, Schimmer R, Beck-Schimmer B. Decreased alveolar oxygen induces lung inflammation. Am J Physiol Lung Cell Mol Physiol. 2003;284:L360–7.
Padley S, Jordan S, Goldstraw P, Wells A, Hansell D. Asymmetric ARDS following pulmonary resection: CT findings initial observations. Radiology. 2002;223:468–73.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Sugasawa, Y., Yamaguchi, K., Kumakura, S. et al. The effect of one-lung ventilation upon pulmonary inflammatory responses during lung resection. J Anesth 25, 170–177 (2011). https://doi.org/10.1007/s00540-011-1100-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-011-1100-0