Abstract
Goal of work
The aim of this study was to investigate the mechanisms whereby low-intensity laser therapy may affect the severity of oral mucositis.
Materials and methods
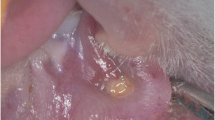
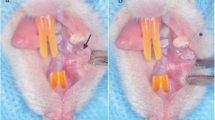
A hamster cheek pouch model of oral mucositis was used with all animals receiving intraperitoneal 5-fluorouracil followed by surface irritation. Animals were randomly allocated into three groups and treated with a 35 mW laser, 100 mW laser, or no laser. Clinical severity of mucositis was assessed at four time-points by a blinded examiner. Buccal pouch tissue was harvested from a subgroup of animals in each group at four time-points. This tissue was used for immunohistochemistry for cyclooxygenase-2 (COX-2), vascular endothelial growth factor (VEGF), and factor VIII (marker of microvessel density) and the resulting staining was quantified.
Main results
Peak severity of mucositis was reduced in the 35 mW laser group as compared to the 100 mW laser and control groups. This reduced peak clinical severity of mucositis in the 35 mW laser group was accompanied by a significantly lower level of COX-2 staining. The 100 mW laser did not have an effect on the severity of clinical mucositis, but was associated with a decrease in VEGF levels at the later time-points, as compared to the other groups. There was no clear relationship of VEGF levels or microvessel density to clinical mucositis severity.
Conclusion
The tissue response to laser therapy appears to vary by dose. Low-intensity laser therapy appears to reduce the severity of mucositis, at least in part, by reducing COX-2 levels and associated inhibition of the inflammatory response.



Similar content being viewed by others
References
Wilkes JD (1998) Prevention and treatment of oral mucositis following cancer chemotherapy. Semin Oncol 25(5):538–551
Lockhart PB, Sonis ST (1981) Alterations in the oral mucosa caused by chemotherapeutic agents. J Dermatol Surg Oncol 7(12):1019–1025
Elting LS, Keefe DM, Sonis ST, Garden AS, Spijkervet FK, Barasch A, Tishler RB, Canty TP, Kudrimoti MK, Vera-Llonch M (2008) Burden of Illness Head and Neck Writing Committee. Patient-reported measurements of oral mucositis in head and neck cancer patients treated with radiotherapy with or without chemotherapy: demonstration of increased frequency, severity, resistance to palliation, and impact on quality of life. Cancer 113(10):2704–2713
Jones JA, Avritscher EB, Cooksley CD, Michelet M, Bekele BN, Elting LS (2006) Epidemiology of treatment-associated mucosal injury after treatment with newer regimens for lymphoma, breast, lung, or colorectal cancer. Support Care Cancer 14(6):505–515
Trotti A, Bellm LA, Epstein JB, Frame D, Fuchs HJ, Gwede CK, Komaroff E, Nalysnyk L, Zilberberg MD (2003) Mucositis incidence, severity and associated outcomes in patients with head and neck cancer receiving radiotherapy with or without chemotherapy: a systematic literature review. Radiother Oncol 66:253–262
McGuire DB, Altomonte V, Peterson DE, Wingard JR, Jones RJ, Grochow LB (1993) Patterns of mucositis and pain in patients receiving preparative chemotherapy and bone marrow transplantation. Oncol Nurs Forum 20(10):1493–502
Sonis ST (2004) The Pathobiology of mucositis. Nat Rev Cancer 4(4):277–284
Lalla RV, Sonis ST, Peterson DE (2008) Management of oral mucositis in patients who have cancer. Dent Clin North Am 52(1):61–77
Lalla RV, Peterson DE (2006) Treatment of mucositis, including new medications. Cancer J 12(5):348–354
Karu T (1989) Photobiology of low-power laser effects. Health Phys 56(5):691–704
Barasch A, Peterson DE, Tanzer JM, D’Ambrosio JA, Nuki K, Schubert MM, Franquin JC, Clive J, Tutschka P (1995) Helium-neon laser effects on conditioning-induced oral mucositis in bone marrow transplantion patients. Cancer 76(12):2550–2556
Bensadoun RJ, Franquin JC, CIais G (1999) Low-energy He/Ne laser in the prevention of radiation mucositis. A multicenter phase III randomized study in patients with head and neck cancer. Support Care Cancer 7:244–252
Cowen D, Tardieu C, Schubert M, Peterson D, Resbeut M, Faucher C, Franquin JC (1997) Low energy helium-neon laser in the prevention of oral mucositis in patients undergoing bone marrow transplant: results of a double blind randomized trial. Int J Radiat Oncol Biol Phys 38:697–703
Bensadoun RJ, Magné N, Marcy PY, Demard F (2001) Chemotherapy and radiotherapy induced mucositis in head and neck cancer patients: new trends in pathophysiology, prevention and treatment. Eur Arch Otorhinol Laryngol 258:481–487
Arun MG, Sagar MS, Fernandes D (2006) Effect of low level helium-neon (He-Ne) laser therapy in the prevention and treatment of radiation induced mucositis in head and neck cancer patients. Indian J Med Res 124:399–402
Antunes HS, Azevedo AM, Bouzas LFS, Adão CAE, Pinheiro CT, Mayte R, Pinheiro LH, Azevedo R, Matos VD, Rodrigues PC, Small IA, Zangaro RA, Ferreira CG (2007) Low-power laser in the prevention of induced oral mucositis in bone marrow transplantation patients: a randomized trial. Blood 109(5):2250–2255
Cruz LB, Ribeiro AS, Rech A, Rosa JGN, Castro Junior CG, Brunetto A (2007) Influence of low-energy laser in the prevention of oral mucositis in children with cancer receiving chemotherapy. Pediatr Blood Cancer 48:435–440
Schubert MM, Eduardo FP, Guthrie KA, Franquin JC, Bensadoun RJJ, Migliorati CA, Lloid CME, Eduardo CP, Walter NF, Marque MM, Hamdi M (2007) A phase III randomized double-blind placebo-controlled clinical trial to determine the efficacy of low level laser therapy for the prevention of oral mucositis in patients undergoing hematopoietic cell transplantation. Support Care Cancer 15:1145–1154
Jaguar GC, Prado JD, Nishimoto IN, Pinheiro MC, Castro DO Jr, Cruz Perez DE, Alves FA (2007) Low-energy laser therapy for prevention of oral mucositis in hematopoietic stem cell transplantion. Oral Dis 3(6):538–543
Abramoff MMF, Lopes NNF, Almeida-Lopes L, Dib LL, Lee ML, Delboni A, Caran EM, Petrill AS (2008) Low level laser therapy (685 nm) in the prevention and treatment of chemotherapy-induced oral mucositis in young patients. Photomed Laser Surg 26(4):393–400
Keefe DMK, Schubert MM, Elting LS, Sonis ST, Epstein JB, Raber-Durlacher JE, Migliorati CA, McGuire DB, Hutchins RD, Peterson DE (2007) Updated clinical practice guidelines for the prevention and treatment of mucositis. Cancer 109(5):820–831
Sonis ST, Tracey C, Shlar G, Jenson J, Florine D, Almeida C (1990) An animal model for mucositis induced by cancer chemotherapy. Oral Surg Oral Med Oral Pathol 69:437–443
Sonis ST, Peterson RL, Edwards LJ, Lucey CA, Wang L, Mason L, Login G, Ymamkawa M, Moses G Bouchard P, Hayes LL, Bedrosian C, Dorner AJ (2000) Defining mechanisms of action of interleukin-11 on the progression of radiation-induced oral mucositis in hamsters. Oral Oncol 36:373–381
Corazza AV, Jorge J, Kurachi C, Bagnato VS (2007) Photobiomodulation on the angiogenesis of skin wounds in rats using different light sources. Photomed Laser Surg 25(2):102–105
França CM, França CM, Núñz SC, Prates RA, Noborikawa E, Faria RM, Ribeiro MS (2009) Low-intensity red laser on the prevention and treatment of induced-oral mucositis in hamsters. J Photochem Photobiol B 94:25–31, doi:10.1016/j.photobiol.2008.09.006
Herschman HR (1996) Prostaglandin synthase 2. Biochim Biophys Acta 1299:125–140
Sonis ST (2002) The biologic role for nuclear factor-kappa B in disease and its potential involvement in mucosal injury associated with anti-neoplastic therapy. Crit Rev Oral Biol Med 13(5):380–389
Sonis ST, O’Donnell KE, Popat R, Bragdon C, Phelan S, Cocks D, Epstein JB (2004) The relationship between mucosal cyclooxygenase-2 (COX-2) expression and experimental radiation-induced mucositis. Oral Oncol 40:170–176
Logan RM, Gibson RJ, Sonis ST, Keefe DMK (2007) Nuclear factor-kappaB (NF-kappaB) and cyclooxygenase-2 (COX-2) expression in the oral mucosa following cancer chemotherapy. Oral Oncol 43(4):395–401
Albertini R, Aimbire F, Villaverde AB, Silva Jr JA, Costa MS (2007) COX-2 mRNA expression decreases in the subplantar muscle of rat paw subjected to carrageenan-induced inflammation after low level laser therapy. Inflamm Res 56:228–229
Vane JR, Bakhle YS, Botting RM (1998) Cyclooxygenases 1 and 2. Annu Rev Pharmacol Toxicol 38:97–120
Wallace JL (2006) COX-2: a pivotal enzyme in mucosal protection and resolution of inflammation. Scientific World Journal 6:577–588
Gilroy DW, Colville-Nash PR, Willis D, Paul-Clark MJ, Willoughby DA (1999) Inducible cyclooxygenase may have anti-inflammatory properties. Nat Med 5:698–701
Dvorak HF, Brown LF, Detmar M, Dvorak AM (1995) Vascular permeability factor/vascular endothelial growth factor, microvascular hyperpermeability, and angiogenesis. Am J Pathol 146(5):1029–1039
Zhang WG, Wu CY, Pan WX, Tian L, Xia JL (2004) Low-power helium-neon laser irradiation enhances the expression of VEGF in murine myocardium. Chin Med J 117(10):1476–1480
Kipshidze N, Nikolaychik V, Keelan MH, Shankar LR, Khanna A, Kornowski R, Leon M, Moses J (2005) Low-power helium: neon laser irradiation enhances production of vascular endothelial growth factor and promotes growth of endothelial cells in vitro. Lasers Surg Med 28:355–364
Acknowledgements
This study was supported by the Fundação de Amparo à Pesquisa de São Paulo (FAPESP).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lopes, N.N.F., Plapler, H., Chavantes, M.C. et al. Cyclooxygenase-2 and vascular endothelial growth factor expression in 5-fluorouracil-induced oral mucositis in hamsters: evaluation of two low-intensity laser protocols. Support Care Cancer 17, 1409–1415 (2009). https://doi.org/10.1007/s00520-009-0603-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-009-0603-9