Abstract
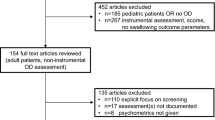
Adults with COPD frequently present with dysphagia, which often leads to clinical complications and hospital admissions. This study investigates the ability of the Eating Assessment Tool (EAT-10) to predict aspiration during objective dysphagia evaluation in adults with stable COPD. Thirty adults (20 male, 10 female; mean age = 69.07 ± 16.82) with stable COPD attended an outpatient dysphagia clinic for a fiberoptic endoscopic evaluation of swallowing (FEES) in an acute teaching hospital (January 2015–November 2016). During evaluations, individuals completed an EAT-10 rating scale followed immediately by a standardised FEES exam. Aspiration status during FEES was rated using the penetration–aspiration scale by clinicians blinded to EAT-10 scores. Data were retrospectively analysed. Significant differences in mean EAT-10 scores were found between aspirators (16.3; SEM = 2.165) and non-aspirators (7.3; SEM = 1.009) (p = 0.000). The EAT-10 predicted aspiration with a high level of accuracy (AUC = 0.88). An EAT-10 cut-off value of >9 presented a sensitivity of 91.67, specificity of 77.78 with positive and negative likelihood ratios of 4.12 and 0.11, respectively. Positive and negative predictive values were 73.30 and 93.30, respectively. Diagnostic odds ratio was 38.50 (p < 0.01, CI 3.75–395.42). EAT-10 is a quick, easy to administer tool, which can accurately predict the presence of aspiration in adults with COPD. The scale can also very accurately exclude the absence of aspiration, helping clinicians to determine the need for onward referral for a comprehensive dysphagia evaluation. This may ultimately reduce clinical complications and hospital admissions resulting from dysphagia in this clinical population.


Similar content being viewed by others
References
Vestbo J. COPD: definition and phenotypes. Clin Chest Med. 2014;35(1):1–6.
Global Strategy for the Diagnosis. Management, and prevention of chronic obstructive pulmonary disease (GOLD). 2008. Accessed 26 Jan 2009.
Mannino DM. COPD: epidemiology, prevalence, morbidity and mortality, and disease heterogeneity. Chest. 2002;121:121S–6S.
Pifarre R, Falguera M, Vicente-de-Vera C, Nogues C. Characteristics of community-acquired pneumonia in patients with chronic obstructive pulmonary disease. Respir Med. 2007. doi:10.1016/j.rmed.2007.05.011.
Cvejic L, Harding R, Churchward T, Turton A, Finlay P, Massey D, Bardin PG, Guy P. Laryngeal penetration and aspiration in individuals with stable COPD. Respirology. 2011;16:269–75.
Mokhlesi B, Logemann JA, Rademaker AW, Stangl CA, Corbridge TC. Oropharyngeal deglutition in stable COPD. Chest. 2002;121(2):361–9.
Shaker R, Li Q, Ren J, Townsend WF, Dodds WJ, Martin BJ, Kern MK, Rynders A. Coordination of deglutition and phases of respiration: effect of aging, tachypnea, bolus volume, and chronic obstructive pulmonary disease. Am J Physiol. 1992;263(5.1):750–5.
Gross DR, Atwood CW, Ross SB, Olszewski JW, Eichhorn KA. The coordination of breathing and swallowing in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2009;179:559–65.
Martin-Harris B. Clinical implications of respiratory—swallowing interactions. Curr Opin Otolaryngol Head Neck Surg. 2008;16(3):194–9.
Good-Fratturelli MD, Curlee RF, Holle JL. Prevalence and nature of dysphagia in VA patients with COPD referred for videofluoroscopic swallow examination. J Commun Disord. 2000;33(2):93–110.
Zheng Z, Wu Z, Liu N, Chen P, Hou P, Wang X, Fu Y, Liang W, Chen R. Silent aspiration in patients with exacerbation of COPD. Eur Respir J. 2016;48(2):570–3.
Stein M, Williams AJ, Grossman F, Weinberg AS, Zuckerbraun L. Cricopharyngeal dysfunction in chronic obstructive pulmonary disease. Chest. 1990;97(2):347–52.
Clayton NA, Carnaby-Mann GD, Peters MJ, Ing AJ. The effect of chronic obstructive pulmonary disease on laryngopharyngeal sensitivity. Ear Nose Throat J. 2012;91(9):370–82.
Coelho CA. Preliminary findings on the nature of dysphagia in patients with chronic obstructive pulmonary disease. Dysphagia. 1987;2(1):28–31.
Takada K, Matsumoto S, Kojima E, Iwata S, Okachi S, Ninomiya K, Morioka H, Tanaka K, Enomoto Y. Prospective evaluation of the relationship between acute exacerbations of COPD and gastroesophageal reflux disease diagnosed by questionnaire. Respir Med. 2011;105(10):1531–6.
Chatila WM, Thomashow BM, Minai OA, Criner GJ, Make BJ. Comorbidities in chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2008;5(4):549–55.
Kobayashi S, Kubo H, Yanai M. Impairment of the swallowing reflex in exacerbations of COPD. Thorax. 2007;62(11):1017.
Schermer TRJ, Saris CGJ, Van Den Bosch WJHM, Chavannes NH, Van Schayck CP, Dekhuijzen PNR, Van Weel C. Exacerbations and associated healthcare cost in patients with COPD in general practice. Monaldi Arch Chest Dis. 2016. doi:10.4081/monaldi.2006.558.
Belafsky PC, Mouadeb DA, Rees CJ, Pryor JC, Postma GN, Allen J, Leonard RJ. Validity and reliability of the Eating Assessment Tool (EAT-10). Ann Otol Rhinol Laryngol. 2008;117(12):919–24.
Cheney DM, Siddiqui MT, Litts JK, Kuhn MA, Belafsky PC. The ability of the 10-item eating assessment tool (EAT-10) to predict aspiration risk in persons with dysphagia. Ann Otol Rhinol Laryngol. 2015;124(5):351–4.
Plowman EK, Tabor LC, Robison R, Gaziano J, Dion C, Watts SA, Vu T, Gooch C. Discriminant ability of the Eating Assessment Tool-10 to detect aspiration in individuals with amyotrophic lateral sclerosis. Neurogastroenterol Motil. 2016;28(1):85–90.
Arrese LC, Carrau R, Plowman EK. Relationship between the Eating Assessment Tool-10 and objective clinical ratings of swallowing function in individuals with head and neck cancer. Dysphagia. 2016;32:83–9.
Rofes L, Arreola V, Mukherjee R, Clave P. Sensitivity and specificity of the Eating Assessment Tool and the volume-viscosity swallow test for clinical evaluation of oropharyngeal dysphagia. Neurogastroenterol Motil. 2014;26(9):1256–65.
Wakabayashi H, Matsushima M. Dysphagia assessed by the 10-item Eating Assessment Tool is associated with nutritional status and activities of daily living in elderly individuals requiring long-term care. J Nutr Health Aging. 2016;20(1):22–7.
Rosenbek JC, Robbins JA, Roecker EB, Coyle JL, Wood JL. A penetration–aspiration scale. Dysphagia. 1996;11(2):93–8.
Colodny N. Interjudge and intrajudge reliabilities in fiberoptic endoscopic evaluation of swallowing (Fees®) using the penetration–aspiration scale: a replication study. Dysphagia. 2002;17(4):308–15.
Kelly AM, Drinnan MJ, Leslie P. Assessing penetration and aspiration: how do videofluoroscopy and fiberoptic endoscopic evaluation of swallowing compare? Laryngoscope. 2007;117(10):1723–7.
Pisegna JM, Langmore SE. Parameters of instrumental swallowing evaluations: describing a diagnostic dilemma. Dysphagia. 2016;31(3):462–72.
MedCalc, Version 16.4.3 (MedCalc Software, Ostend, Belgium). 2016. https://www.medcalc.org.
Dollaghan CA. The handbook for evidence-based practice in communication disorders. Baltimore: Paul H Brookes Publishing Company; 2007.
Almirall J, Rofes L, Serra-Prat M, Icart R, Palomera E, Arreola V, Clave P. Oropharyngeal dysphagia is a risk factor for community-acquired pneumonia in the elderly. Eur Respir J. 2013;41:923–8.
Ickenstein GW, Riecker A, Höhlig C, Müller R, Becker U, Reichmann H, Prosiegel M. Pneumonia and in-hospital mortality in the context of neurogenic oropharyngeal dysphagia (NOD) in stroke and a new NOD step-wise concept. J Neurol. 2010;257:1492–9.
Hinchey JA, Shephard T, Furie K, Smith D, Wang D, Tonn S. Formal dysphagia screening protocols prevent pneumonia. Stroke. 2005;36:1972–6.
Rofes L, Arreola V, Romea M, Palomera E, Almirall J, Cabré M, Serra-Prat M, Clavé P. Pathophysiology of oropharyngeal dysphagia in frail elderly. Neurogastroenterol Motil. 2010;22:851.
Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, Leeflang MM, Sterne JA, Bossuyt PM. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011;155(8):529–36.
Speyer R, Cordier R, Kertscher B, Heijnen BJ. Psychometric properties of questionnaires on functional health status in oropharyngeal dysphagia: a systematic literature review. BioMed Res Int. 2014. doi:10.1155/2014/458678.
Cordier R, Joosten A, Clavé P, Schindler A, Bülow M, Demir N, Serel Arslan S, Speyer R. Evaluating the psychometric properties of the Eating Assessment Tool (EAT-10) using rasch analysis. Dysphagia. 2016;32:250–60.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Regan, J., Lawson, S. & De Aguiar, V. The Eating Assessment Tool-10 Predicts Aspiration in Adults with Stable Chronic Obstructive Pulmonary Disease. Dysphagia 32, 714–720 (2017). https://doi.org/10.1007/s00455-017-9822-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00455-017-9822-2