Abstract
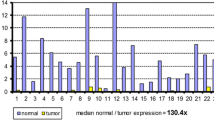
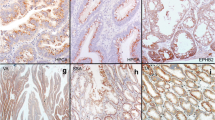
Programmed cell death 4 (PDCD4) has recently been demonstrated to be a new tumor suppressor gene involved in colon carcinogenesis. PDCD4 immunohistochemical expression was assessed in 300 polypoid lesions of the colon mucosa (50 hyperplastic polyps [HP], 50 serrated adenomas [SA], 50 tubular adenomas with low-grade-intraepithelial neoplasia [LG-IEN], 50 tubular adenomas with high-grade-IEN [HG-IEN]), and in 50 colon adenocarcinomas (CRC). As normal controls, we considered 50 biopsy samples obtained from patients with irritable bowel syndrome (N). We further investigated PDCD4 messenger RNA (mRNA) levels by quantitative real-time polymerase chain reaction (PCR) in a different series of N, LG-IEN, HG-IEN, and CRC biopsy samples. miR-21 expression (an important PDCD4-expression regulator) was also determined by quantitative real-time PCR and in situ hybridization. Normal colocytes and HP featured strong PDCD4 nuclear immunostaining whereas a significantly lower PDCD4 nuclear expression was observed in dysplasia (low- and high-grade adenomas and SA) and invasive CRC. PDCD4 immunostaining and mRNA levels decreased significantly as the phenotypic changes occurring during colon carcinogenesis progressively increased (p < 0.001). As expected, miR-21 expression was significantly upregulated in preneoplastic/neoplastic samples, consistent with PDCD4 downregulation. These results consistently support the use of nuclear PDCD4 immunohistochemical downregulation as a novel biomarker for the diagnosis of dysplastic/neoplastic lesions in colon biopsy samples.



Similar content being viewed by others
References
Jemal A, Siegel R, Ward E et al (2009) Cancer statistics, 2009. CA Cancer J Clin 59:225–249
Cunningham D, Atkin W, Lenz HJ et al (2010) Colorectal cancer. Lancet 375:1030–1047
Lieberman D (2010) Progress and challenges in colorectal cancer screening and surveillance. Gastroenterology 138:2115–2126
Vogelstein B, Fearon ER, Hamilton SR et al (1988) Genetic alterations during colorectal tumor development. N Engl J Med 319:525–532
Renehan AG, O'Dwyer ST, Haboubi NJ et al (2002) Early cellular events in colorectal carcinogenesis. Colorectal Dis 4:76–89
Cappell MS (2008) Reducing the incidence and mortality of colon cancer: mass screening and colonoscopic polypectomy. Gastroenterol Clin North Am 37:129–160
Hewett DG, Kahi CJ, Rex DK (2010) Does colonoscopy work? J Natl Compr Canc Netw 8:67–76, quiz 77
Bohm M, Sawicka K, Siebrasse JP et al (2003) The transformation suppressor protein Pdcd4 shuttles between nucleus and cytoplasm and binds RNA. Oncogene 22:4905–4910
Yang HS, Cho MH, Zakowicz H et al (2004) A novel function of the MA-3 domains in transformation and translation suppressor Pdcd4 is essential for its binding to eukaryotic translation initiation factor 4A. Mol Cell Biol 24:3894–3906
Afonja O, Juste D, Das S et al (2004) Induction of PDCD4 tumor suppressor gene expression by RAR agonists, antiestrogen and HER-2/neu antagonist in breast cancer cells. Evidence for a role in apoptosis. Oncogene 23:8135–8145
Bitomsky N, Bohm M, Klempnauer KH (2004) Transformation suppressor protein Pdcd4 interferes with JNK-mediated phosphorylation of c-Jun and recruitment of the coactivator p300 by c-Jun. Oncogene 23:7484–7493
Zakowicz H, Yang HS, Stark C et al (2005) Mutational analysis of the DEAD-box RNA helicase eIF4AII characterizes its interaction with transformation suppressor Pdcd4 and eIF4GI. RNA 11:261–274
Jansen AP, Camalier CE, Colburn NH (2005) Epidermal expression of the translation inhibitor programmed cell death 4 suppresses tumorigenesis. Cancer Res 65:6034–6041
Dorrello NV, Peschiaroli A, Guardavaccaro D et al (2006) S6K1- and betaTRCP-mediated degradation of PDCD4 promotes protein translation and cell growth. Science 314:467–471
Bitomsky N, Wethkamp N, Marikkannu R et al (2008) siRNA-mediated knockdown of Pdcd4 expression causes upregulation of p21(Waf1/Cip1) expression. Oncogene 27:4820–4829
Carayol N, Katsoulidis E, Sassano A et al (2008) Suppression of programmed cell death 4 (PDCD4) protein expression by BCR-ABL-regulated engagement of the mTOR/p70 S6 kinase pathway. J Biol Chem 283:8601–8610
Mudduluru G, Medved F, Grobholz R et al (2007) Loss of programmed cell death 4 expression marks adenoma–carcinoma transition, correlates inversely with phosphorylated protein kinase B, and is an independent prognostic factor in resected colorectal cancer. Cancer 110:1697–1707
Allgayer H (2010) Pdcd4, a colon cancer prognostic that is regulated by a microRNA. Crit Rev Oncol Hematol 73:185–191
Fassan M, Pizzi M, Battaglia G et al (2010) Programmed cell death 4 (PDCD4) expression during multistep Barrett’s carcinogenesis. J Clin Pathol 63:692–696
Chen Y, Knosel T, Kristiansen G et al (2003) Loss of PDCD4 expression in human lung cancer correlates with tumour progression and prognosis. J Pathol 200:640–646
Zhang H, Ozaki I, Mizuta T et al (2006) Involvement of programmed cell death 4 in transforming growth factor-beta1-induced apoptosis in human hepatocellular carcinoma. Oncogene 25:6101–6112
Wang Q, Sun Z, Yang HS (2008) Downregulation of tumor suppressor Pdcd4 promotes invasion and activates both beta-catenin/Tcf and AP-1-dependent transcription in colon carcinoma cells. Oncogene 27:1527–1535
Baffa R, Fassan M, Volinia S et al (2009) MicroRNA expression profiling of human metastatic cancers identifies cancer gene targets. J Pathol 219:214–221
Yamamichi N, Shimomura R, Inada K et al (2009) Locked nucleic acid in situ hybridization analysis of miR-21 expression during colorectal cancer development. Clin Cancer Res 15:4009–4016
Fassan M, Cagol M, Pennelli G et al (2010) Programmed cell death 4 (PDCD4) protein in esophageal cancer. Oncol Rep 24:135–139
Goke R, Barth P, Schmidt A et al (2004) Programmed cell death protein 4 suppresses CDK1/cdc2 via induction of p21(Waf1/Cip1). Am J Physiol Cell Physiol 287:C1541–C1546
Rhodes DR, Yu J, Shanker K et al (2004) ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 6:1–6
Rhodes DR, Yu J, Shanker K et al (2004) Large-scale metaanalysis of cancer microarray data identifies common transcriptional profiles of neoplastic transformation and progression. Proc Natl Acad Sci USA 101:9309–9314
Xin W, Rhodes DR, Ingold C et al (2003) Dysregulation of the annexin family protein family is associated with prostate cancer progression. Am J Pathol 162:255–261
Rugge M, Fassan M, Clemente R et al (2008) Bronchopulmonary carcinoid: phenotype and long-term outcome in a single-institution series of Italian patients. Clin Cancer Res 14:149–154
Reis PP, Tomenson M, Cervigne NK et al (2010) Programmed cell death 4 loss increases tumor cell invasion and is regulated by miR-21 in oral squamous cell carcinoma. Mol Cancer 9:238
Fassan M, Volinia S, Palatini J et al (2011) MicroRNA expression profiling in human Barrett’s carcinogenesis. Int J Cancer (in press)
Sabates-Bellver J, Van der Flier LG, de Palo M et al (2007) Transcriptome profile of human colorectal adenomas. Mol Cancer Res 5:1263–1275
Gaspar C, Cardoso J, Franken P et al (2008) Cross-species comparison of human and mouse intestinal polyps reveals conserved mechanisms in adenomatous polyposis coli (APC)-driven tumorigenesis. Am J Pathol 172:1363–1380
Zou TT, Selaru FM, Xu Y et al (2002) Application of cDNA microarrays to generate a molecular taxonomy capable of distinguishing between colon cancer and normal colon. Oncogene 21:4855–4862
Kaiser S, Park YK, Franklin JL et al (2007) Transcriptional recapitulation and subversion of embryonic colon development by mouse colon tumor models and human colon cancer. Genome Biol 8:R131
Yoshinaga H, Matsuhashi S, Fujiyama C et al (1999) Novel human PDCD4 (H731) gene expressed in proliferative cells is expressed in the small duct epithelial cells of the breast as revealed by an anti-H731 antibody. Pathol Int 49:1067–1077
Leggett B, Whitehall V (2010) Role of the serrated pathway in colorectal cancer pathogenesis. Gastroenterology 138:2088–2100
Smith RA, Cokkinides V, Brooks D et al (2010) Cancer screening in the United States, 2010: a review of current American Cancer Society guidelines and issues in cancer screening. CA Cancer J Clin 60:99–119
Jankowski JA, Odze RD (2009) Biomarkers in gastroenterology: between hope and hype comes histopathology. Am J Gastroenterol 104:1093–1096
Acknowledgements
This work has been partly supported by an AIRC Regional grant 2009 (assigned to M.R.). The authors are grateful to Vincenza Guzzardo, Mariangela Balistreri, Vanni Lazzarin, and Cristiano Lanza for their technical assistance.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fassan, M., Pizzi, M., Giacomelli, L. et al. PDCD4 nuclear loss inversely correlates with miR-21 levels in colon carcinogenesis. Virchows Arch 458, 413–419 (2011). https://doi.org/10.1007/s00428-011-1046-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-011-1046-5