Abstract.
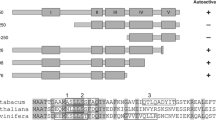
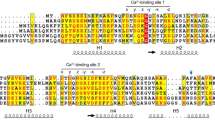
A member of a eukaryotic gene superfamily, encoding a peptidylproline cis-trans-isomerase (rotamase) has been isolated from a maize (Zea mays L. A69Y+) endosperm cDNA library. The maize sequence (mzFKBP-66) encodes a 66-kDa polypeptide most closely related to the subclass of rotamases which bind an immunosuppressive drug, FK506, (termed FK506-binding proteins, FKBPs), and possesses four tandem copies of the FKBP-like binding domain. The sequence mzFKBP-66 is expressed ubiquitously in the maize plant, and the protein encoded is present in both cytosolic and nuclear compartments within the cell. Both the native mzFKBP-66 and a recombinant protein overexpressed in Escherichia coli showed peptidylproline␣cis-trans-isomerase (PPIase) activity at rates comparable to those reported for mammalian immunophilins. This activity was also sensitive to inhibition by FK506. Immunoaffinity chromatography using anti-mzFKBP66 demonstrated an association of the protein with an unknown 36-kDa polypeptide, and affinity chromatography of mzFKBP-66 on calmodulin-agarose beads indicated the presence of a calmodulin-binding site. The existence of mzFKBP-66-associated proteins suggests that plant immunophilins may act as part of multicomponent complexes, as has been shown for other representatives of this class of enzyme.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 9 June 1997 / Accepted: 19 August 1997
Rights and permissions
About this article
Cite this article
Hueros, G., Rahfeld, J., Salamini, F. et al. A maize FK506-sensitive immunophilin, mzFKBP-66, is a peptidylproline cis-trans-isomerase that interacts with calmodulin and a 36-kDa cytoplasmic protein. Planta 205, 121–131 (1998). https://doi.org/10.1007/s004250050303
Issue Date:
DOI: https://doi.org/10.1007/s004250050303