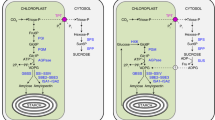
Abstract
Adenosine diphosphate glucose pyrophosphorylase (AGPase; EC 2.7.7.27) synthesizes the starch precursor, ADP-glucose. It is a rate-limiting enzyme in starch biosynthesis and its activation by 3-phosphoglyceric acid (3PGA) and/or inhibition by inorganic phosphate (Pi) are believed to be physiologically important. Leaf, tuber and cereal embryo AGPases are highly sensitive to these effectors, whereas endosperm AGPases are much less responsive. Two hypotheses can explain the 3PGA activation differences. Compared to leaf AGPases, endosperm AGPases (i) lack the marked ability to be activated by 3PGA or (ii) they are less dependent on 3PGA for activity. The absence of purified preparations has heretofore negated answering this question. To resolve this issue, heterotetrameric maize (Zea mays L.) endosperm and potato (Solanum tuberosum L.) tuber AGPases expressed in Escherichia coli were isolated and the relative amounts of enzyme protein were measured by reaction to antibodies against a motif resident in both small subunits. Resulting reaction rates of both AGPases are comparable in the presence but not in the absence of 3PGA when expressed on an active-protein basis. We also placed the potato tuber UpReg1 mutation into the maize AGPase. This mutation greatly enhances 3PGA sensitivity of the potato AGPase but it has little effect on the maize AGPase. Thirdly, lysines known to bind 3PGA in potato tuber AGPase, but missing from the maize endosperm AGPase, were introduced into the maize enzyme. These had minimal effect on maize endosperm activity. In conclusion, the maize endosperm AGPase is not nearly as dependent on 3PGA for activity as is the potato tuber AGPase.




Similar content being viewed by others
Abbreviations
- AGPase:
-
ADP-glucose pyrophosphorylase
- Bt2 :
-
brittle2
- Glc-1-P:
-
glucose-1-phosphate
- K a :
-
3PGA concentration giving one-half maximal activation
- 3PGA:
-
3-phosphoglyceric acid
- Sh2 :
-
shrunken2
References
Ball K, Preiss J (1994) Allosteric sites of the large subunit of the spinach leaf ADP-glucose pyrophosphorylase. J Biol Chem 269:24706–24711
Ballicora MA, Laughlin MJ, Fu Y, Okita TW, Barry GF, Preiss J (1995) Adenosine 5′-diphosphate-glucose pyrophosphorylase from potato tuber. Significance of the N terminus of the small subunit for catalytic properties and heat stability. Plant Physiol 109:245–251
Ballicora MA, Fu Y, Nesbitt NM, Preiss J (1998) ADP-glucose pyrophosphorylase from potato tubers. Site-directed mutagenesis studies of the regulatory sites. Plant Physiol 118:265–274
Beckles DM, Smith AM, ap Rees T (2001) A cytosolic ADP-glucose pyrophosphorylase is a feature of graminaceous endosperms, but not of other starch-storing organs. Plant Physiol 125:818–827
Brangeon J, Reyss A, Prioul JL (1997) In situ detection of ADPglucose pyrophosphorylase expression during maize endosperm development. Plant Physiol Biochem 35:847–858
Burton RA, Johnson E, Beckles DM, Fincher GB, Jenner HL, Naldrett MJ, Denyer K (2002) Characterization of the genes encoding the cytosolic and plastidial forms of ADP-glucose pyrophosphorylase in wheat endosperm. Plant Physiol 130:1464–1475
Cao H, Sullivan TD, Boyer CD, Shannon JC (1995) Bt1, a structural gene for the major 39–44 kD amyloplast membrane polypeptides. Physiol Plant 95:176–186
Denyer K, Dunlap F, Thorbjornsen T, Keeling P, Smith AM (1996) The major form of ADPglucose pyrophosphorylase in maize endosperm is extra-plastidial. Plant Physiol 112:779–785
Dickinson DB, Preiss J (1969) ADP glucose pyrophosphorylase from maize endosperm. Arch Biochem Biophys 130:119–128
Doan DN, Rudi H, Olsen OA (1999) The allosterically unregulated isoform of ADP-glucose pyrophosphorylase from barley endosperm is the most likely source of ADP-glucose incorporated into endosperm starch. Plant Physiol 121:965–975
Giroux MJ, Hannah LC (1994) ADP-glucose pyrophosphorylase in shrunken-2 and brittle-2 mutants of maize. Mol Gen Genet 243:400–408
Giroux MJ, Shaw JR, Barry G, Cobb BG, Greene TW, Okita T, Hannah LC (1996) A single gene mutation that increases maize seed weight. Proc Natl Acad Sci USA 93:5824–5829
Greene TW, Hannah LC (1998a) Maize endosperm ADP-glucose pyrophosphorylase SHRUNKEN2 and BRITTLE2 subunit interactions. Plant Cell 10:1295–1306
Greene TW, Hannah LC (1998b) Enhanced stability of maize endosperm ADP-glucose pyrophosphorylase is gained through mutants that alter subunit interactions. Proc Natl Acad Sci USA 95:13342–13357
Greene TW, Chantler SE, Kahn ML, Barry GF, Preiss J, Okita TW (1996) Mutagenesis of the potato ADPglucose pyrophosphorylase and characterization of an allosteric mutant defective in 3-phosphoglycerate activation. Proc Natl Acad Sci 93:1509–1513
Greene TW, Kavakli IH, Kahn ML, Okita TW (1998) Generation of up-regulated allosteric variants of potato ADP-glucose pyrophosphorylase by reversion genetics. Proc Natl Acad Sci 95:10322–10327
Hannah LC (1997) Starch synthesis in the maize endosperm. In: Larkins BA, Vasil IK (eds) Advances in cellular and molecular biology of plants: Cellular and molecular biology of plant seed development, vol 4. Kluwer, Dordrecht, pp 375–405
Hannah LC, Nelson OE (1975) Characterization of adenosine diphosphate glucose pyrophosphorylases from developing maize seeds. Plant Physiol 55:297–302
Hannah LC, Nelson OE Jr (1976) Characterization of ADP-glucose pyrophosphorylase from shrunken-2 and brittle-2 mutants of maize. Biochem Genet 14:547–560
Hannah LC, Baier J, Caren J, Giroux M (1995) 3-Phosphoglyceric acid activation of maize endosperm ADP-glucose pyrophosphorylase following proteolytic cleavage of the SH2 or BT2 subunits. In: Pontis HD, Salerno GL, Echeverria E (eds) Sucrose metabolism, biochemistry and molecular biology, vol 14. American Society of Plant Physiologists, Rockville, pp 72–79
Hylton C, Smith AM (1992) The rb mutation of peas causes structural and regulatory changes in ADP-glucose pyrophosphorylase from developing embryos. Plant Physiol 99:1626–1634
Iglesias AA, Barry GF, Meyer C, Bloksberg L, Nakata PA, Greene T, Laughlin MJ, Okita TW, Kishore GM, Preiss J (1993) Expression of the potato tuber ADP-glucose pyrophosphorylase in Escherichia coli. J Biol Chem 268:1081–1086
Kim WT, Franceschi VR, Okita TW, Robinson NL, Morell M, Preiss J (1989) Immunocytochemical localization of ADPglucose pyrophosphorylase in developing potato tuber cells. Plant Physiol 91:217–220
Kleczkowski LA, Villand P, Luthi E, Olsen O-A, Preiss J (1993) Insensitivity of barley endosperm ADP-glucose pyrophosphorylase to 3-phosphoglycerate and orthophosphate regulation. Plant Physiol 101:179–186
Martin C, Smith AM (1995) Starch biosynthesis. Plant Cell 7:971–985
Nelson OE, Pan D (1995) Starch synthesis in maize endosperms. Annu Rev Plant Physiol Plant Mol Biol 46:475–496
Okita TW, Greenberg E, Kuhn DN, Preiss J (1979) Subcellular localization of the starch degradative and biosynthetic enzymes of spinach leaves. Plant Physiol 64:187–192
Olive MR, Ellis RJ, Schuch WW (1989) Isolation and nucleotide sequences of cDNA clones encoding ADP-glucose pyrophosphorylase peptides from wheat leaf and endosperm. Plant Mol Biol 12:525–538
Plaxton WC, Preiss J (1987) Purification and properties of non-proteolytically degraded ADP-glucose pyrophosphorylase from maize endosperm. Plant Physiol 83:105–112
Preiss J (1982) Regulation of biosynthesis and degradation of starch. Annu Rev Plant Physiol 33:431–54
Shannon JC, Fang-Mei P, Kang-Chein L (1996) Nucleotides and nucleotide sugars in developing maize endosperms. Plant Physiol 110:835–843
Sikka, VK, Choi S-B, Kavakli IH, Sakulsingharoj C, Gupta S, Ito H, Okita TW (2001) Subcellular compartmentation and allosteric regulation of the rice endosperm ADPglucose pyrophosphorylase. Plant Sci 161:461–468
Smidansky ED, Clancy M, Meyer FD, Lanning SP, Blake NK, Talbert LE, Giroux MJ (2002) Enhanced ADP-glucose pyrophosphorylase activity in wheat endosperm increases seed yield. Proc Natl Acad Sci USA 99:1724–1729
Stark DM, Timmerman KP, Barry G, Preiss J, Kishore GM (1992) Regulation of the amount of starch in plant tissues by ADP-glucose pyrophosphorylase. Science 258:287–292
Thorbjornsen T, Villand P, Denyer K, Olsen O, Smith AM (1996) Distinct isoforms of ADPglucose pyrophosphorylase occur inside and outside the amyloplast in barley endosperm. Plant J 10:243–250
Villand P, Kleczkowski LA (1994) Is there an alternative pathway for starch biosynthesis in cereal seeds? Z Naturforsch Teil C 49: 215–219
Weber H, Heim U, Borisjuk L, Wobus U (1995) Cell-type specific, coordinate expression of two ADP-glucose pyrophosphorylase genes in relation to starch biosynthesis during seed development of Vicia faba L. Planta 195:352–361
Acknowledgments
We gratefully acknowledge support from the National Science Foundation (L.C.H.: IBN-9316887, IBN-960416, IBN-9982626 and MCB-9420422), the USDA Competitive Grants Program (L.C.H.: 94-37300-453, 9500836, 95-37301-2080, 9701964, 97-36306-4461, 98-01006 and 2000-01488), the Florida Agricultural Experiment Station (Journal Series No. R-08238) and Department of Energy (T.W.O.: DEFG03–96ER20216). We thank Drs. Rob Ferl, Don McCarty and Karen Koch of the University of Florida for many helpful discussions. We thank Dr. Ken Cline (University of Florida) for assistance with image analysis, Dr. Nancy Denslow (University of Florida) for help with antibody production and the Interdisciplinary Center for Biotechnology Research at the University of Florida for DNA sequencing and protein synthesis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Burger, B.T., Cross, J.M., Shaw, J.R. et al. Relative turnover numbers of maize endosperm and potato tuber ADP-glucose pyrophosphorylases in the absence and presence of 3-phosphoglyceric acid. Planta 217, 449–456 (2003). https://doi.org/10.1007/s00425-003-1000-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-003-1000-0