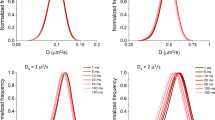
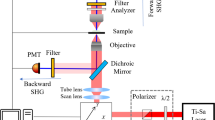
Abstract
In this study, we aimed at improving the temporal resolution of scanning probe microscopy (SPM) for observing living cells by introducing soft cantilevers, low feedback-gain operations, and cantilever deflection imaging. We achieved visualization of the mechanical architecture in leading lamellae of living fibroblasts at a temporal resolution of around 10 s, which is higher than that of conventional contact-mode SPM. Time-lapse SPM could be used to monitor not only cytoskeletal dynamics but also the dynamics of numerous microgranules. Statistical analysis of microgranular motion revealed that the microgranules have superdiffusive behaviors and significant directional order of motion. We also found that the direction of their motion is correlated with the direction of growing actin stress fibers. The combination of SPM with fluorescence microscopy showed that vinculin, a component of cell-substratum adhesion sites, localizes at the microgranules. Our experimental data provides a new insight into the intracellular mechanical architecture and its structural dynamics, suggesting that high-speed live-cell SPM has great potential for investigating the structural origin of cellular dynamics.





Similar content being viewed by others
References
Alcaraz J, Buscemi L, Grabulosa M, Trepat X, Fabry B, Farré R, Navajas D (2003) Microrheology of human lung epithelial cells measured by atomic force microscopy. Biophys J 84:2071–2079
Anderson TW, Vaughan AN, Cramer LP (2008) Retrograde flow and myosin II activity within the leading cell edge deliver F-actin to the lamella to seed the formation of graded polarity actomyosin II filament bundles in migrating fibroblasts. Mol Biol Cell 19:5006–5018
Ando T, Kodera N, Takai E, Maruyama D, Saito K, Toda A (2001) A high-speed atomic force microscope for studying biological macromolecules. Proc Natl Acad Sci USA 98:12468–12472
Baumgartner W, Hinterdorfer P, Ness W, Raab A, Vestweber D, Schindler H, Dreckhahn D (2000) Cadherin interaction probed by atomic force microscopy. Proc Natl Acad Sci USA 97:4005–4010
Benoit M, Gabriel D, Gerisch G, Gaub HE (2000) Discrete interactions in cell adhesion measured by single-molecule force spectroscopy. Nat Cell Biol 2:313–317
Brown CM, Hebert B, Kolin DL, Zareno J, Whitmore L, Horwitz AR, Wiseman PW (2006) Probing the integrin-actin linkage using high-resolution protein velocity mapping. J Cell Sci 119:5204–5214
Bursac P, Lenormand G, Fabry B, Oliver M, Weitz DA, Viasnoff V, Butler JP, Fredberg JJ (2005) Cytoskeletal remodelling and slow dynamics in the living cell. Nat Mater 4:557–561
Cai Y, Biais N, Giannone G, Tanase M, Jiang G, Hofman JM, Wiggins CH, Silberzan P, Buguin A, Ladoux B, Sheetz MP (2006) Nonmuscle myosin IIA-dependent force inhibits cell spreading and drives F-actin flow. Biophys J 91:3907–3920
Goffin JM, Pittet P, Csucs G, Lussi JW, Meister J-J, Hinz B (2006) Focal adhesion size controls tension-dependent recruitment of α-smooth muscle actin to stress fibers. J Cell Biol 172:259–268
Gorelik J, Shevchuk AI, Frolenkov GI, Diakonov IA, Lab MJ, Kros CJ, Richardson GP, Vodyanoy I, Edwards CR, Klenerman D, Korchev YE (2003) Dynamic assembly of surface structures in living cells. Proc Natl Acad Sci USA 100:5819–5822
Greenwood JA, Durand D (1955) The distribution of length and components of the sum of n random unit vectors. Ann Math Stat 26:233–246
Grzywa EL, Lee AC, Lee GU, Suter DM (2006) High-resolution analysis of neuronal growth cone morphology by comparative atomic force and optical microscopy. J Neurobiol 66:1529–1543
Habelitz S, Balooch M, Marshall SJ, Balooch G, Marshall GW Jr (2002) In situ atomic force microscopy of partially demineralized human dentin collagen fibrils. J Struct Biol 138:227–236
Haga H, Sasaki S, Kawabata K, Ito E, Ushiki T, Sambongi T (2000) Elasticity mapping of living fibroblasts by AFM and immunofluorescence observation of the cytoskeleton. Ultramicroscopy 82:253–258
Hansma PK, Schitter G, Fantner GE, Prater C (2006) High-speed atomic force microscopy. Science 314:601–602
Helenius J, Heisenberg C-P, Gaub HE, Müller DJ (2008) Single-cell force spectroscopy. J Cell Sci 121:1785–1791
Henderson E, Haydon PG, Sakaguchi DS (1992) Actin filament dynamics in living glial cells imaged by atomic force microscopy. Science 257:1944–1946
Hertz H (1882) Über die berührung fester elastischer körper. J Reine Angew Math 92:156–171
Hirata H, Tatsumi H, Sokabe M (2007) Dynamics of actin filaments during tension-dependent formation of actin bundles. Biochim Biophys Acta 1770:1115–1127
Hu K, Ji L, Applegate KT, Danuser G, Waterman-Storer CM (2007) Differential transmission of actin motion within focal adhesions. Science 315:111–115
Le Grimellec C, Lesniewska E, Giocondi MC, Finot E, Vié V, Goudonnet JP (1998) Imaging of the surface of living cells by low-force contact-mode atomic force microscopy. Biophys J 75:695–703
Levenberg K (1944) A method for the solution of certain nonlinear problems in least squares. Q Appl Math 2:164–168
Marquardt D (1963) An algorithm for least-squares estimation of nonlinear parameters. J Appl Math 11:431–441
McGregor A, Blanchard AD, Rowe AJ, Critchley DR (1994) Identification of the vinculin-binding site in the cytoskeletal protein alpha-actinin. Biochem J 301:225–233
Menkel AR, Kroemker M, Bubeck P, Ronsiek M, Nikolai G, Jockusch BM (1994) Characterization of an F-actin-binding domain in the cytoskeletal protein vinculin. J Cell Biol 126:1231–1240
Pesen D, Hoh JH (2005) Micromechanical architecture of the endothelial cell cortex. Biophys J 88:670–679
Picco LM, Bozec L, Ulcinas A, Engledew DJ, Antognozzi M, Horton MA, Miles MJ (2007) Breaking the speed limit with atomic force microscopy. Nanotechnology 18:044030
Picco LM, Dunton PG, Ulcinas A, Engledew DJ, Hoshi O, Ushiki T, Miles MJ (2008) High-speed AFM of human chromosomes in liquid. Nanotechnology 19:384018
Ponti A, Machacek M, Gupton SL, Waterman-Storer CM, Danuser G (2004) Two distinct actin networks drive the protrusion of migrating cells. Science 305:1782–1786
Radmacher M, Fritz M, Kacher CM, Cleveland JP, Hansma PK (1996) Measuring the viscoelastic properties of human platelets with the atomic force microscope. Biophys J 70:556–567
Raspanti M, Congiu T, Guizzardi S (2001) Tapping-mode atomic force microscopy in fluid of hydrated extracellular matrix. Matrix Biol 20:601–604
Rottner K, Hall A, Small JV (1999) Interplay between Rac and Rho in the control of substrate contact dynamics. Curr Biol 9:640–648
Schoenenberger CA, Hoh JH (1994) Slow cellular dynamics in MDCK and R5 cells monitored by time-lapse atomic force microscopy. Biophys J 67:929–936
Tamura K, Mizutani T, Haga H, Kawabata K (2007) Visualization of stretch-induced intracellular tensional response of single fibroblasts by mechanical scanning probe microscopy. Jpn J Appl Phys 46:5631–5635
Trepat X, Deng L, An SS, Navajas D, Tschumperlin DJ, Gerthoffer WT, Butler JP, Fredberg JJ (2007) Universal physical responses to stretch in the living cell. Nature 447:592–595
Vallotton P, Gupton SL, Waterman-Storer CM, Danuser G (2004) Simultaneous mapping of filamentous actin flow and turnover in migrating cells by quantitative fluorescent speckle microscopy. Proc Natl Acad Sci USA 101:9660–9665
Waterman-Storer CM, Salmon ED (1997) Actomyosin-based retrograde flow of microtubules in the lamella of migrating epithelial cells influences microtubule dynamic instability and turnover and is associated with microtubule breakage and treadmilling. J Cell Biol 139:417–434
Yamane Y, Shiga H, Asou H, Haga H, Kawabata K, Abe K, Ito E (1999) Dynamics of astrocyte adhesion as analyzed by a combination of atomic force microscopy and immuno-cytochemistry: the involvement of actin filaments and connexin 43 in the early stage of adhesion. Arch Histol Cytol 62:355–361
Zar JH (1999) Biostatistical analysis. Prentice-Hall, Upper Saddle River
Acknowledgments
We are grateful to Dr. Masayuki Takahashi (Division of Chemistry, Graduate School of Science, Hokkaido University, Sapporo, Japan) for the gift of anti-myosin-IIA antibody. We thank Dr. Koji Nemoto (Division of Physics, Graduate School of Science, Hokkaido University, Sapporo, Japan) for the valuable discussions. This study was supported by Grants-in-Aid for Exploratory Research (21654058) to K. K., Scientific Research (C) (21570158) to H. H., and JSPS Fellows (20.4710) to K. T. from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tamura, K., Mizutani, T., Haga, H. et al. Dynamics of leading lamellae of living fibroblasts visualized by high-speed scanning probe microscopy. Histochem Cell Biol 133, 59–67 (2010). https://doi.org/10.1007/s00418-009-0644-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-009-0644-7