Abstract
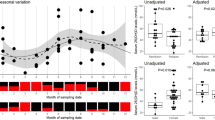
Inflammatory serum parameters are intensely investigated in the search of biomarkers for disease activity and treatment response in multiple sclerosis (MS). A reason for contradictory results might be the timing of blood collection for analyzing serum concentrations of inflammatory parameters which are subject to diurnal changes. We included 34 untreated patients with relapsing-remitting MS and 34 age- and sex-matched healthy controls. 12 MS patients showed acute disease activity in corresponding MRI scans. Blood samples were obtained at 7.00, 11.00 am, 2.30, 6.00 and 9.30 pm within 1 day. We determined serum levels of cortisol and inflammatory markers including soluble tumor necrosis factor-beta (sTNF-β), soluble TNF-Receptor-1 (sTNF-R1) and -2 (sTNF-2), soluble vascular adhesion molecule-1 (sVCAM-1) and soluble intercellular adhesion molecule-1 (sICAM-1) by ELISA. We observed significantly higher serum levels of sTNF-R1 (p < 0.001) and sTNF-R2 (p < 0.001) in the morning and a significant decline of sICAM-1 (p < 0.005) and sVCAM-1 (p < 0.001) in the afternoon in both, MS patients and healthy controls. Comparison of diurnal serum levels between MS patients with active versus with non-active disease revealed significantly higher serum levels of sVCAM-1 (p < 0.05) around noon and in the early afternoon in MS patients with active disease. A significant decline of sICAM-1 (p < 0.05) in the afternoon was seen in MS patients with active and non-active disease. Our data indicate that increased awareness of potential diurnal serum concentration changes of biomarkers can eliminate one major cause of biased data as they occur in most of the investigated immunological parameters.


Similar content being viewed by others
References
Noseworthy JH et al (2000) Multiple sclerosis. N Engl J Med 343:938–952
Engelhardt B, Kappos L (2008) Natalizumab: targeting alpha4-integrins in multiple sclerosis. Neurodegener Dis 5:16–22
Wipfler P et al (2011) Adhesion molecules are promising candidates to establish surrogate markers for natalizumab treatment. Mult Scler 17:16–23
Kraus J et al (1998) Soluble and cell surface ICAM-1 as markers for disease activity in multiple sclerosis. Acta Neurol Scand 98:102–109
Kraus J et al (2002) Cell surface bound and soluble adhesion molecules in CSF and blood in multiple sclerosis: correlation with MRI-measures of subclinical disease severity and activity. J Neuroimmunol 122:175–185
Rieckmann P, Altenhofen B, Riegel A, Kallmann B, Felgenhauer K (1998) Correlation of soluble adhesion molecules in blood and cerebrospinal fluid with magnetic resonance imaging activity in patients with multiple sclerosis. Mult Scler 4:178–182
Hartung HP, Reiners K, Archelos JJ et al (1996) Circulating adhesion molecules and tumor necrosis factor receptor in multiple sclerosis: correlation with magnetic resonance imaging. Ann Neurol 38:186–193
McDonnell GV, McMillan SA, Douglas JP, Droogan AG, Hawkins SA (1998) Raised CSF levels of soluble adhesion molecules across the clinical spectrum of multiple sclerosis. J Neuroimmunol 85:186–192
Niehaus GD et al (2002) Circadian variation in cell-adhesion molecule expression by normal human leukocytes. Can J Physiol Pharmacol 80:935–940
Young MW (2000) The tick–tock of the biological clock. Sci Am 282:64–71
Petrovsky N, McNair P, Harrison LC (1998) Diurnal rhythms of pro-inflammatory cytokines: regulation by plasma cortisol and therapeutic implications. Cytokine 10:307–312
Glass-Marmor L et al (2007) Chronotherapy using corticosteroids for multiple sclerosis relapses. J Neurol Neurosurg Psychiatry 78:886–888
Glass-Marmor L et al (2009) Immunomodulation by chronobiologically based glucocorticoids treatment for multiple sclerosis relapses. J Neuroimmunol 210:124–127
Laske C et al (2001) Induction of sTNF-R1 and sTNF-R2 by interferon beta-1b in correlation with clinical and MRI activity. Acta Neurol Scand 103:105–113
Polman CH, Reingold SC, Edan G et al (2005) Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann Neurol 58:840–846
Pilz G, Wipfler P, Ladurner G et al (2008) Modern multiple sclerosis treatment—what is approved, what is on the horizon. Drug Discov Today 13:1013–1025
Brambilla R, Ashbaugh JJ, Magliozzi R et al (2011) Inhibition of soluble tumour necrosis factor is therapeutic in experimental autoimmune encephalomyelitis and promotes axon preservation and remyelination. Brain 134:2736–2754
Redford EJ, Hall SM, Smith KJ (1995) Vascular changes and demyelination induced by the intraneural injection of tumour necrosis factor. Brain 118:869–878
Kraus J, Kuehne BS, Tofghi J et al (2002) Serum cytokine levels do not correlate with disease activity and severity assessed by brain MRI in multiple sclerosis. Acta Neurol Scand 105:300–308
Archelos JJ, Hartung HP (1997) The role of adhesion molecules in multiple sclerosis: biology, pathogenesis and therapeutic implications. Mol Med Today 3:310–321
Giovannoni G, Lai M, Thorpe J, Kidd D, Chamoun V, Thompson AJ, Miller DH, Feldmann M, Thompson EJ (1997) Longitudinal study of soluble adhesion molecules in multiple sclerosis: correlation with gadolinium enhanced magnetic resonance imaging. Neurology 48:1557–1565
Engelhardt B (2006) Molecular mechanisms involved in T cell migration across the blood-brain barrier. J Neural Transm 113:477–485
Michelson D, Stone L, Galliven E et al (1994) Multiple sclerosis is associated with alterations in hypothalamic-pituitary-adrenal axis function. J Clin Endocrinol Metab 79:848–853
Grasser A, Moller A, Backmund H et al (1996) Heterogeneity of hypothalamic–pituitary–adrenal system response to a combined dexamethasone-CRH test in multiple sclerosis. Exp Clin Endocrinol Diabet 104:31–37
Reuss R et al (2009) Interferon beta-1a induces tumor necrosis factor receptor 1 but decreases tumor necrosis factor receptor 2 leukocyte mRNA levels in relapsing-remitting multiple sclerosis. NeuroImmunoModulation 16:171–176
Conflicts of interest
We certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
Ethical standard
This study has been approved by the appropriate ethics committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wipfler, P., Heikkinen, A., Harrer, A. et al. Circadian rhythmicity of inflammatory serum parameters: a neglected issue in the search of biomarkers in multiple sclerosis. J Neurol 260, 221–227 (2013). https://doi.org/10.1007/s00415-012-6622-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-012-6622-3