Abstract
Purpose
Interleukin (IL)-25 and IL-33 induce IL-5 production by various types of cells, such as type 2 helper T (Th2) cells and type 2 innate lymphoid cells. The number of Th2 cells and concentration of IL-5 in the bronchoalveolar lavage fluid (BALF) are increased in patients with eosinophilic pneumonia (EP). To examine the contribution of IL-25 and IL-33 to eosinophilic inflammation of the lung in humans, we evaluated IL-5, IL-25 and IL-33 levels in the BALF of patients with EP.
Methods
IL-5, IL-25, and IL-33 concentrations in the BALF were measured by enzyme-linked immunosorbent assay in patients with acute eosinophilic pneumonia (AEP), chronic eosinophilic pneumonia (CEP), idiopathic pulmonary fibrosis (IPF), and sarcoidosis.
Results
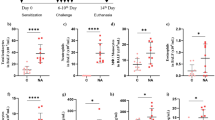
The absolute number of eosinophils, and IL-5 levels, but not IL-33 levels, in the BALF were significantly higher in patients with EP than in patients with IPF and sarcoidosis. IL-25 levels in the BALF were significantly higher in patients with CEP, but not in patients with AEP, than in patients with IPF and sarcoidosis. The absolute number of eosinophils was significantly correlated with the IL-5 concentration in the BALF of patients with EP. IL-5 concentrations were significantly correlated with IL-25 concentrations in the BALF of patients with CEP, but not in patients with AEP. IL-5 levels were not correlated with IL-33 levels in the BALF of patients with EP.
Conclusions
Our findings suggest that IL-25 plays an important role via IL-5 in eosinophilic lung inflammation in patients with CEP.




Similar content being viewed by others
References
Liebow AA, Carrington CB (1969) The eosinophilic pneumonia. Medicine 48:251–255
Allen JN (2006) Acute eosinophilic pneumonia. Semin Respir Crit Care Med 27:142–147
Marchand E, Cordier JF (2006) Idiopathic chronic eosinophilic pneumonia. Semin Respir Crit Care Med 27:134–141
Katoh S, Taniguchi H, Matsubara Y et al (1999) Overexpression of CD44 on alveolar eosinophils with high concentrations of soluble CD44 in bronchoalveolar lavage fluid in patients with eosinophilic pneumonia. Allergy 54:1286–1292
Katoh S, Matsumoto N, Matsumoto K et al (2004) Elevated interleukin-18 levels in bronchoalveolar lavage fluid of patients with eosinophilic pneumonia. Allergy 59:850–856
Katoh S, Fukushima K, Matsumoto N et al (2003) Accumulation of CCR4-expressing CD4 + T cells and high concentration of its ligands (TARC and MDC) in bronchoalveolar lavage fluid of patients with eosinophilic pneumonia. Allergy 58:518–523
Iwakura Y, Ishigame H, Saijo S et al (2011) Functional specialization of interleukin-17 family members. Immunity 34:149–162
Ohno T, Morita H, Arae K et al (2012) Interleukin-33 in allergy. Allergy 67:1203–1214
Ziegier SF (2012) Thymic stromal lymphopoietin and allergic disease. J Allergy Clin Immunol 130:845–852
Ikutani M, Yanagibashi T, Ogasawara M et al (2012) Identification of innate IL-5-producing cells and their role in lung eosinophils regulating and antitumor immunity. J Immunol 188:703–713
Halim TYF, Krauss RH, Sun AC et al (2012) Lung natural helper cells are a critical source of Th2 cell-type cytokines in protease allergen-induced airway inflammation. Immunity 36:451–463
Yasuda K, Muto T, Kawagoe T et al (2012) Contribution of IL-33-activated type II innate lymphoid cells to pulmonary eosinophilia in intestinal nematode-infected mice. Proc Natl Acad Sci 109:3451–3456
Brusselle GG, Maes T, Bracke KR (2013) Eosinophilic airway inflammation in nonallergic asthma. Nat Med 19:977–979
Yu S, Kim HY, Chang Y-J et al (2014) Innate lymphoid cells and asthma. J Allergy Clin Immunol 133:943–950
Travis WD, Costabel U, Hansell DM et al (2013) ATS/ERS Committee on Idiopathic Interstitial Pneumonias. An official American thoracic society/European respiratory society statement: update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am J Respir Crit Care Med 188:733–748
Katoh S, Matsumoto N, Fukushima K et al (2000) Elevated chemokines levels in bronchoalveolar lavage fluid of patients with eosinophilic pneumonia. J Allergy Clin Immunol 106:730–736
Valizadeh A, Khosravi A, Zadeh LJ et al (2015) Role of IL-25 in immunity. J Clin Diag Res 9(4):OE01–OE04
Tang W, Smith SG, Beaudin S et al (2014) IL-25 and IL-25 receptor expression on eosinophils from subjects with allergic asthma. Int Arch Allergy Immunol 163:5–10
Terrier B, Bieche I, Maisonobe T et al (2010) Interleukin-25: a cytokine linking eosinophils and adaptive immunity in Churg–Strauss syndrome. Blood 116:4523–4531
Endo Y, Nakayama T (2015) Pathogenic Th2 (Tpath2) cells in airway inflammation. Oncotarget 6:32303–32304
Liu B, Lee J-B, Chen C-Y et al (2015) Collaborative interactions between type 2 innate lymphoid cells and antigen-specific CD4 + Th2 cells exacerbate murine allergic airway diseases with prominent eosinophilia. J Immunol 194:3583–3593
Kamijo S, Takeda H, Tokura T et al (2013) IL-33-mediated innate response and adaptive immune cells contribute to maximum responses of protease allergen-induced allergic airway inflammation. J Immunol 190:4489–4499
Barlow JL, Peel S, Fox J et al (2013) IL-33 is more potent than IL-25 in provoking IL-13-producing nuocytes (type 2 innate lymphoid cells) and airway contraction. J Allergy Clin Immunol 132:933–941
Kim H-R, Jun C-D, Lee Y-J et al (2010) Levels of circulating IL-33 and eosinophil cationic protein in patients with hypereosinophilia or pulmonary eosinophilia. J Allergy Clin Immunol 126:880–882
Mato N, Bando M, Kusano A et al (2013) Clinical significance of interleukin 33 (IL-33) in patients with eosinophilic pneumonia. Allergol Int 62:45–52
Jeong YJ, Kim K-I, Seo IJ et al (2007) Eosinophilic lung diseases: a clinical, radiologic, and pathologic overview. RadioGraphics 27:617–639
Acknowledgements
The authors thank Ms. Tae Uesaka and Ms. Mariko Takaya for their skillful assistance. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Katoh, S., Ikeda, M., Matsumoto, N. et al. Possible Role of IL-25 in Eosinophilic Lung Inflammation in Patients with Chronic Eosinophilic Pneumonia. Lung 195, 707–712 (2017). https://doi.org/10.1007/s00408-017-0048-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-017-0048-8