Abstract
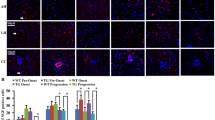
Connective tissue growth factor (CTGF) is a secreted protein involved in a variety of cellular events such as survival, proliferation, and extracellular matrix production. Recent studies suggest a role for this protein also in the repair processes of the central nervous system. The distribution and significance of CTGF in human brain is, however, poorly understood, particularly under pathological conditions. In the present study the expression of CTGF protein was investigated in the spinal cord of control and both sporadic and familial amyotrophic lateral sclerosis (sALS and fALS) patients. Western blot analysis showed a consistent increase in CTGF expression in six sALS patients compared with controls. Immunoreactivity signal for CTGF was equally present in blood vessels of control and ALS spinal cord, but was dramatically increased in reactive astrocytes of the ventral horn and white matter in both sALS and fALS. Increased expression was also observed in the cytoplasm of motor neurons of sALS and fALS patients with long duration of the disease. Our data indicate a role for CTGF in the complex reactive process that is associated with the progression of ALS spinal cord damage. The up-regulation in reactive astrocytes supports a role for CTGF in the molecular mechanisms underlying astrogliosis. However, the altered CTGF expression observed in neurons might represent an additional mechanism involved in motor neuron dysfunction and changes in glial-neuronal communication in the course of the neurodegenerative process.




Similar content being viewed by others
References
Abreu JG, Ketpura NI, Reversade B, De Robertis EM (2002) Connective-tissue growth factor (CTGF) modulates cell signalling by BMP and TGF-beta. Nat Cell Biol 4:599–604
Aronica E, Vliet EA van, Mayboroda OA, Troost D, Silva FH da, Gorter JA (2000) Upregulation of metabotropic glutamate receptor subtype mGluR3 and mGluR5 in reactive astrocytes in a rat model of mesial temporal lobe epilepsy. Eur J Neurosci 12:2333–2344
Aronica E, Catania MV, Geurts J, Yankaya B, Troost B (2001) Immunohistochemical localization of group I and II metabotropic glutamate receptors in control and ALS human spinal cord: upregulation in reactive astrocytes. Neuroscience 105:509–520
Aronica E, Leenstra S, Jansen GH, Veelen CW van, Yankaya B, Troost D (2001) Expression of brain-derived neurotrophic factor and tyrosine kinase B receptor proteins in glioneuronal tumors from patients with intractable epilepsy: colocalization with N-methyl-d-aspartic acid receptor. Acta Neuropathol 101:383–392
Aronica E, Troost D, Yankaya B, Jansen GH, Isom L, Gorter JA (2003) Expression and regulation of voltage gated sodium channel (NaCh) β1 subunit protein in human glioses-associated pathologies. Acta Neuropathol 105:515–523
Babic AM, Chen CC, Lau LF (1999) Fisp12/mouse connective tissue growth factor mediates endothelial cell adhesion and migration through integrin alphavbeta3, promotes endothelial cell survival, and induces angiogenesis in vivo. Mol Cell Biol 19:2958–2966
Blom IE, Dijk AJ van, Wieten L, Duran K, Ito Y, Klei jL, deNichilo M, Rabelink TJ, Weening JJ, Aten J, Goldschmeding R (2001) In vitro evidence for differential involvement of CTGF, TGFbeta, and PDGF-BB in mesangial response to injury. Nephrol Dial Transplant 16:1139–1148
Brown RH Jr (1995) Amyotrophic lateral sclerosis: recent insights from genetics and transgenic mice. Cell 80:687–692
Bruccoleri A, Brown H, Harry GJ (1998) Cellular localization and temporal elevation of tumor necrosis factor-alpha, interleukin-1 alpha, and transforming growth factor-beta 1 mRNA in hippocampal injury response induced by trimethyltin. J Neurochem 71:1577–1587
Bruno V, Battaglia G, Casabona G, Copani A, Caciagli F, Nicoletti F (1998) Neuroprotection by glial metabotropic glutamate receptors is mediated by transforming growth factor-beta. J Neurosci 18:9594–9600
Chou SM (1995) Pathology of the motor system. In: Leigh PN, Swash M (eds) Motor neuron disease. Springer, London, pp 53–92
Duncan MR, Frazier KS, Abramson S, Williams S, Klapper H, Huang X, Grotendorst GR (1999) Connective tissue growth factor mediates transforming growth factor beta-induced collagen synthesis: down-regulation by cAMP. FASEB J 13:1774–1786
Elliott JL (2001) Cytokine upregulation in a murine model of familial amyotrophyc lateral sclerosis. Mol Brain Res 95:172–178
Godbout R, Bisgrove DA, Shkolny D, Day RS 3rd (1998) Correlation of B-FABP and GFAP expression in malignant glioma. Oncogene 16:1955–1962
Hall ED, Oostveen JA, Gurney ME (1998) Relationship of microglial and astrocytic activation to disease onset and progression in a transgenic model of familial ALS. Glia 23:249–256
Hertel M, Tretter Y, Alzheimer C, Werner S (2000) Connective tissue growth factor: a novel player in tissue reorganization after brain injury? Eur J Neurosci 12:376–380
Ilzecka J, Stelmasiak Z, Dobosz B (2002) Transforming growth factor-Beta 1 (tgf-Beta 1) in patients with amyotrophic lateral sclerosis. Cytokine 20:239–243
Ince PG, Lowe J, Shaw PJ (1998) Amyotrophic lateral sclerosis: current issues in classification, pathogenesis and molecular pathology. Neuropathol Appl Neurobiol 24:104–117
Inoki I, Shiomi T, Hashimoto G, Enomoto H, Nakamura H, Makino K, Ikeda E, Takata S, Kobayashi K, Okada Y (2002) Connective tissue growth factor binds vascular endothelial growth factor (VEGF) and inhibits VEGF-induced angiogenesis. FASEB J 16:219–221
Ito Y, Aten J, Bende RJ, Oemar BS, Rabelink TJ, Weening JJ, Goldschmeding R (1998) Expression of connective tissue growth factor in human renal fibrosis. Kidney Int 53:853–861
Ito Y, Goldschmeding R, Kasuga H, Matsuo S, Weening JJ, Aten J (2001) Expression of CCN2 (CTGF) and TGFbeta isoforms during glomerulogenesis and in adult glomerulopathies. J Am Soc Nephrol 12:679A
Jackson CE, Bryan WW (1998) Amyotrophic lateral sclerosis. Semin Neurol 18:27–39
Kaluza J, Krupinski J, Kumar P, Kumar S, Wang, JM (1994) VCAM-1 expression on reactive and tumour astrocytes. Folia Histochem Cytobiol 32:17–20
Kamo H, Haebara H, Akiguchi I, Kameyama M, Kimura H, McGeer PL (1987) A distinctive distribution of reactive astroglia in the precentral cortex in amyotrophic lateral sclerosis. Acta Neuropathol (Berl) 74:33–38
Khurgel M, Ivy GO (1996) Astrocytes in kindling: relevance to epileptogenesis. Epilepsy Res 26:163–175
Kiefer R, Streit WJ, Toyka KV, Kreutzberg GW, Hartung HP (1995) Transforming growth factor-beta 1: a lesion-associated cytokine of the nervous system. Int J Dev Neurosci 13:331–339
Kondo Y, Nakanishi T, Takigawa M, Ogaw N (1999) Immunohistochemical localization of connective tissue growth factor in the rat central nervous system. Brain Res 834:146–151
Krieglstein K, Strelau J, Schober A, Sullivan A, Unsicker K (2002) TGF-beta and the regulation of neuron survival and death. J Physiol 96:25–30
Krishna M, Smith TW, Recht LD (1995) Expression of bcl-2 in reactive and neoplastic astrocytes: lack of correlation with presence or degree of malignancy. J Neurosurg 83:1017–1022
Krishnamurti U, Chen Y, Michael A, Kim Y, Fan WW, Wieslander J, Brunmark C, Rondeau E, Sraer JD, Delarue F, Tsilibary EC (1996) Integrin-mediated interactions between primary/T-sv40 immortalized human glomerular epithelial cells and type IV collagen. Lab Invest 74:650–657
Kushner PD, Stephenson DT, Wright S (1991) Reactive astrogliosis is widespread in the subcortical white matter of amyotrophic lateral sclerosis brain. Neuropathol Exp Neurol 50:263–277
Lehrmann E, Kiefer R, Finsen B, Diemer NH, Zimmer J, Hartung HP (1995) Cytokines in cerebral ischemia: expression of transforming growth factor beta-1 (TGF-beta 1) mRNA in the postischemic adult rat hippocampus. Exp Neurol 131:114–123
Levine JB, Kong J, Nadler M, Xu Z (1999) Astrocytes interact intimately with degenerating motor neurons in mouse amyotrophic lateral sclerosis (ALS). Glia 28:215–224
McGeer PL, McGeer EG (2002) Inflammatory processes in amyotrophic lateral sclerosis. Muscle Nerve 26:459–470
Morgan TE, Nichols NR, Pasinetti GM, Finch CE (1993) TGF-beta 1 mRNA increases in macrophage/microglial cells of the hippocampus in response to deafferentation and kainic acid-induced neurodegeneration. Exp Neurol 120:291–301
Moussad EE, Brigstock DR (2000) Connective tissue growth factor: what's in a name? Mol Genet Metab 71:276–292
Murayama S, Inoue K, Kawakami H, Bouldin TW, Suzuki K (1991) A unique pattern of astrocytosis in the primary motor area in amyotrophic lateral sclerosis. Acta Neuropathol 82:456–461
Nagy D, Kato T, Kushner PD (1994) Reactive astrocytes are widespread in the cortical gray matter of amyotrophic lateral sclerosis. J Neurosci Res 38:336–347
Nishida T, Nakanishi T, Shimo T, Asano M, Hattori T, Tamatani T, Tezuka K, Takigawa M (1998) Demonstration of receptors specific for connective tissue growth factor on a human chondrocytic cell line (HCS-2/8). Biochem Biophys Res Commun 247:905–909
Pan LH, Beppu T, Kurose A, Yamauchi K, Sugawara A, Suzuki M, Ogawa A, Sawai T (2002) Neoplastic cells and proliferating endothelial cells express connective tissue growth factor (CTGF) in glioblastoma. Neurol Res 24:677–683
Ridet JL, Malhotra SK, Privat A, Gage FH (1997) Reactive astrocytes: cellular and molecular cues to biological function. Trends Neurosci 20:570–577
Schiffer D, Cordera S, Cavalla P, Migheli A (1996) Reactive astrogliosis of the spinal cord in amyotrophic lateral sclerosis. Neurol Sci 139:27–33
Schwab JM, Postler E, Nguyen TD, Mittelbronn M, Meyermann R, Schluesener HJ (2000) Connective tissue growth factor is expressed by a subset of reactive astrocytes in human cerebral infarction. Neuropathol Appl Neurobiol 26:434–440
Schwab JM, Beschorner R, Nguyen TD, Meyermann R, Schluesener HJ (2001) Differential cellular accumulation of connective tissue growth factor defines a subset of reactive astrocytes, invading fibroblasts, and endothelial cells following central nervous system injury in rats and humans. J Neurotrauma 18:377–388
Segarini PR, Nesbitt JE, Li D, Hays LG, Yates JR 3rd, Carmichael DF (2001) The low density lipoprotein receptor-related protein/alpha2-macroglobulin receptor is a receptor for connective tissue growth factor. J Biol Chem 276:40659–40667
Shimo T, Nakanishi T, Nishida T, Asano M, Kanyama M, Kuboki T, Tamatani T, Tezuka K, Takemura M, Matsumura T, Takigawa M (1999) Connective tissue growth factor induces the proliferation, migration, and tube formation of vascular endothelial cells in vitro, and angiogenesis in vivo. J Biochem 126:137–145
Surveyor GA, Brigstock DR (1999) Immunohistochemical localization of connective tissue growth factor (CTGF) in the mouse embryo between days 7.5 and 14.5 of gestation. Growth Factors 17:115–124
Troost D, Van den Oord JJ, Vianney de Jong JM (1990) Immunohistochemical characterization of the inflammatory infiltrate in amyotrophic lateral sclerosis. Neuropathol Appl Neurobiol 16:401–410
Vandeputte DA, Troost D, Leenstra S, Ijlst-Keizers H, Ramkema M, Bosch DA, Baas F, Das NK, Aronica E (2002) Expression and distribution of id helix-loop-helix proteins in human astrocytic tumors. Glia 38:329–338
Wahab NA, Brinkman H, Mason RM (2001) Uptake and intracellular transport of the connective tissue growth factor: a potential mode of action. Biochem J 359:89–97
Yamada T, Kawamata T, Walker DG, McGeer PL (1992) Vimentin immunoreactivity in normal and pathological human brain tissue. Acta Neuropathol 84:157–162
Zhu Y, Yang GY, Ahlemeyer B, Pang L, Che XM, Culmsee C, Klumpp S, Krieglstein J (2002) Transforming growth factor-beta 1 increases bad phosphorylation and protects neurons against damage. J Neurosci 22:3898–3909
Acknowledgements
This work was supported by the Netherlands ALS Research Foundation and Stichting AZUA-funds.
Author information
Authors and Affiliations
Corresponding author
Additional information
The first two authors contributed equally to the present work.
Rights and permissions
About this article
Cite this article
Spliet, W.G.M., Aronica, E., Ramkema, M. et al. Increased expression of connective tissue growth factor in amyotrophic lateral sclerosis human spinal cord. Acta Neuropathol 106, 449–457 (2003). https://doi.org/10.1007/s00401-003-0741-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-003-0741-y