Abstract
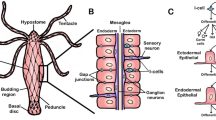
The inner ear is essential for maintaining balance and hearing predator and prey in the environment. Each inner ear contains three CaCO3 otolith polycrystals, which are calcified within an alkaline, K+-rich endolymph secreted by the surrounding epithelium. However, the underlying cellular mechanisms are poorly understood, especially in marine fish. Here, we investigated the presence and cellular localization of several ion-transporting proteins within the saccular epithelium of the Pacific Chub Mackerel (Scomber japonicus). Western blotting revealed the presence of Na+/K+-ATPase (NKA), carbonic anhydrase (CA), Na+-K+-2Cl−-co-transporter (NKCC), vacuolar-type H+-ATPase (VHA), plasma membrane Ca2+ ATPase (PMCA), and soluble adenylyl cyclase (sAC). Immunohistochemistry analysis identified two distinct ionocytes types in the saccular epithelium: Type-I ionocytes were mitochondrion-rich and abundantly expressed NKA and NKCC in their basolateral membrane, indicating a role in secreting K+ into the endolymph. On the other hand, Type-II ionocytes were enriched in cytoplasmic CA and VHA, suggesting they help transport HCO3− into the endolymph and remove H+. In addition, both types of ionocytes expressed cytoplasmic PMCA, which is likely involved in Ca2+ transport and homeostasis, as well as sAC, an evolutionary conserved acid–base sensing enzyme that regulates epithelial ion transport. Furthermore, CA, VHA, and sAC were also expressed within the capillaries that supply blood to the meshwork area, suggesting additional mechanisms that contribute to otolith calcification. This information improves our knowledge about the cellular mechanisms responsible for endolymph ion regulation and otolith formation, and can help understand responses to environmental stressors such as ocean acidification.







Similar content being viewed by others
References
Abbas L, Whitfield TT (2009) Nkcc1 (Slc12a2) is required for the regulation of endolymph volume in the otic vesicle and swim bladder volume in the zebrafish larva. Development 136:2837–2848. https://doi.org/10.1242/dev.034215
Allemand D, Mayer-Gostan N, De Pontual H, Boeuf G, Payan P (2008) Fish otolith calcification in relation to endolymph chemistry. Handb Biominer Biol Asp Struct Form 1:291–308. https://doi.org/10.1002/9783527619443.ch17
Allmon EB, Esbaugh AJ (2017) Carbon dioxide induced plasticity of branchial acid-base pathways in an estuarine teleost. Sci Rep 7:45680. https://doi.org/10.1038/srep45680
Barott K, Venn AA, Perez SO, Tambutté S, Tresguerres M (2015a) Coral host cells acidify symbiotic algal microenvironment to promote photosynthesis. Proc Natl Acad Sci U S A 112:607–612. https://doi.org/10.1073/pnas.1413483112
Barott KL, Perez SO, Linsmayer LB, Tresguerres M (2015b) Differential localization of ion transporters suggests distinct cellular mechanisms for calcification and photosynthesis between two coral species. Am J Physiol Regul Integr Comp Physiol 309:R235–R246. https://doi.org/10.1152/ajpregu.00052.2015
Barott KL, Barron ME, Tresguerres M (2017) Identification of a molecular pH sensor in coral. Proc R Soc B Biol Sci 284:20171769. https://doi.org/10.1098/rspb.2017.1769
Barron ME, Roa JNBR, Tresguerres M (2012) Pacific oyster mantle, gill and hemocytes express the bicarbonate-sensing enzyme soluble adenylyl cyclase. FASEB J 26:1070–1072
Barron ME, Thies AB, Espinoza JA, Barott KL, Hamdoun A, Tresguerres M (2018) A vesicular Na+/Ca2+ exchanger in coral calcifying cells. PLoS ONE 13:e0205367. https://doi.org/10.1371/journal.pone.0205367
Beier M, Anken R, Hilbig R (2006) Sites of calcium uptake of fish otoliths correspond with macular regions rich of carbonic anhydrase. Adv Space Res 38:1123–1127. https://doi.org/10.1016/j.asr.2005.10.042
Bignami S, Enochs IC, Manzello DP, Sponaugle S, Cowen RK (2013) Ocean acidification alters the otoliths of a pantropical fish species with implications for sensory function. Proc Natl Acad Sci 110:7366–7370. https://doi.org/10.1073/pnas.1301365110
Borelli G, Guibbolini ME, Mayer-Gostan N, Priouzeau F, De Pontual H, Allemand D et al (2003) Daily variations of endolymph composition: relationship with the otolith calcification process in trout. J Exp Biol 206:2685–2692. https://doi.org/10.1242/jeb.00479
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Campana S (1999) Chemistry and composition of fish otoliths: pathways, mechanisms and applications. Mar Ecol Prog Ser 188:263–297. https://doi.org/10.3354/meps188263
Campana SE, Neilson JD (1985) Microstructure of fish otoliths. Can J Fish Aquat Sci 42:1014–1032
Carvalho ESM, Gregório SF, Power DM, Canário AVM, Fuentes J (2012) Water absorption and bicarbonate secretion in the intestine of the sea bream are regulated by transmembrane and soluble adenylyl cyclase stimulation. J Comp Physiol B Biochem Syst Environ Physiol 182:1069–1080. https://doi.org/10.1007/s00360-012-0685-4
Checkley DM, Dickson AG, Takahashi M, Radich JA, Eisenkolb N, Asch R (2009) Elevated CO2 enhances otolith growth in young fish. Science 324:1683. https://doi.org/10.1126/science.1169806
Chen Y, Cann MJ, Litvin TN, Iourgenko V, Sinclair ML, Levin LR et al (2000) Soluble adenylyl cyclase as an evolutionarily conserved bicarbonate sensor. Science 289:625–628. https://doi.org/10.1126/science.289.5479.625
Cooper CA, Regan MD, Brauner CJ, De Bastos ESR, Wilson RW (2014) Osmoregulatory bicarbonate secretion exploits H+-sensitive haemoglobins to autoregulate intestinal O2 delivery in euryhaline teleosts. J Comp Physiol B Biochem Syst Environ Physiol 184:865–876. https://doi.org/10.1007/s00360-014-0844-x
Cruz S, Shiao JC, Liao BK, Huang CJ, Hwang PP (2009) Plasma membrane calcium ATPase required for semicircular canal formation and otolith growth in the zebrafish inner ear. J Exp Biol 212:639–647. https://doi.org/10.1242/jeb.022798
Dijkgraaf S (1960) Hearing in bony fishes. Proc R Soc B 152:51–54
Elsdon TS, Gillanders BM (2002) Interactive effects of temperature and salinity on otolith chemistry: challenges for determining environmental histories of fish. Can J Fish Aquat Sci 59:1796–1808. https://doi.org/10.1139/f02-154
Esbaugh AJ, Cutler B (2016) Intestinal Na+, K+, 2Cl-cotransporter 2 plays a crucial role in hyperosmotic transitions of a euryhaline teleost. Physiol Rep 4:1–12. https://doi.org/10.14814/phy2.13028
Esbaugh AJ, Heuer R, Grosell M (2012) Impacts of ocean acidification on respiratory gas exchange and acid-base balance in a marine teleost, Opsanus beta. J Comp Physiol B Biochem Syst Environ Physiol 182:921–934. https://doi.org/10.1007/s00360-012-0668-5
Esbaugh AJ, Ern R, Nordi WM, Johnson AS (2016) Respiratory plasticity is insufficient to alleviate blood acid–base disturbances after acclimation to ocean acidification in the estuarine red drum, Sciaenops ocellatus. J Comp Physiol B Biochem Syst Environ Physiol 186:97–109. https://doi.org/10.1007/s00360-015-0940-6
Fairbanks MB, Hoffert JR, Fromm PO (1969) The dependence of the oxygen-concentrating mechanism of the teleost eye (Salmo gairdneri) on the enzyme carbonic anhydrase. J Gen Physiol 54:203–211. https://doi.org/10.1085/jgp.54.2.203
Furukawa T, Ishii Y (1967) Neurophysiological studies on hearing in goldfish. J Neurophysiol 30:1377–1403
Georgalis T, Gilmour KM, Yorston J, Perry SF (2006) Roles of cytosolic and membrane-bound carbonic anhydrase in renal control of acid-base balance in rainbow trout, Oncorhynchus mykiss. Am J Physiol 291:407–421. https://doi.org/10.1152/ajprenal.00328.2005
Ghanem TA, Breneman KD, Rabbitt RD, Brown HM (2008) Ionic composition of endolymph and perilymph in the inner ear of the oyster toadfish, Opsanus tau. Biol Bull 214:83–90. https://doi.org/10.2307/25066662
Hiroi J, Yasumasu S, McCormick SD, Hwang P-P, Kaneko T (2008) Evidence for an apical Na–Cl cotransporter involved in ion uptake in a teleost fish. J Exp Biol 211:2584–2599. https://doi.org/10.1242/jeb.018663
Ibsch M, Anken R, Beier M, Rahmann H (2004) Endolymphatic calcium supply for fish otolith growth takes place via the proximal portion of the otocyst. Cell Tissue Res 317:333–336. https://doi.org/10.1007/s00441-004-0930-6
Inokuchi M, Hiroi J, Watanabe S, Lee KM, Kaneko T (2008) Gene expression and morphological localization of NHE3, NCC and NKCC1a in branchial mitochondria-rich cells of Mozambique tilapia (Oreochromis mossambicus) acclimated to a wide range of salinities. Comp Biochem Physiol A: Mol Integr Physiol 151:151–158. https://doi.org/10.1016/j.cbpa.2008.06.012
Kwan GT, Wexler JB, Wegner NC, Tresguerres M (2019) Ontogenetic changes in cutaneous and branchial ionocytes and morphology in yellowfin tuna (Thunnus albacares) larvae. J Comp Physiol B Biochem Syst Environ Physiol 189:81–95. https://doi.org/10.1007/s00360-018-1187-9
Ladich F, Schulz-Mirbach T (2016) Diversity in fish auditory systems: one of the riddles of sensory biology. Front Ecol Evol 4:1–26. https://doi.org/10.3389/fevo.2016.00028
Lebovitz RM, Takeyasu K, Fambrough DM (1989) Molecular characterization and expression of the (Na+ + K+)-ATPase alpha-subunit in Drosophila melanogaster. EMBO J 8:193–202
Litvin TN, Kamenetsky M, Zarifyan A, Buck J, Levin LR (2003) Kinetic properties of “soluble” adenylyl cyclase: synergism between calcium and bicarbonate. J Biol Chem 278:15922–15926. https://doi.org/10.1074/jbc.M212475200
Lytle C, Xu JC, Biemesderfer D, Forbush B (1995) Distribution and diversity of Na–K–Cl cotransport proteins: a study with monoclonal antibodies. Am J Physiol 269:C1496–C1505
Maneja RH, Frommel AY, Geffen AJ, Folkvord A, Piatkowski U, Chang MY et al (2013) Effects of ocean acidification on the calcification of otoliths of larval Atlantic cod Gadus morhua. Mar Ecol Prog Ser 477:251–258. https://doi.org/10.3354/meps10146
Mayer-Gostan N, Kossmann H, Watrin A, Payan P, Boeuf G (1997) Distribution of ionocytes in the saccular epithelium of the inner ear of two teleosts (Oncorhynchus mykiss and Scophthalmus maximus). Cell Tissue Res 289:53–61. https://doi.org/10.1007/s004410050851
Michael K, Kreiss CM, Hu MY, Koschnick N, Bickmeyer U, Dupont S et al (2016) Adjustments of molecular key components of branchial ion and pH regulation in Atlantic cod (Gadus morhua) in response to ocean acidification and warming. Comp Biochem Physiol Part B 193:33–46. https://doi.org/10.1016/j.cbpb.2015.12.006
Mugiya Y, Yoshida M (1995) Effects of calcium antagonists and other metabolic on in vitro calcium deposition on otoliths rainbow trout Oncorhynchus mykiss modulators. Fish 61:1026–1030
Munday PL, Hernaman V, Dixson DL, Thorrold SR (2011) Effect of ocean acidification on otolith development in larvae of a tropical marine fish. Biogeosciences 8:1631–1641. https://doi.org/10.5194/bg-8-1631-2011
Murayama E, Takagi Y, Ohira T, Davis JG, Greene MI, Nagasawa H (2002) Fish otolith contains a unique structural protein, otolin-1. Eur J Biochem 269:688–696. https://doi.org/10.1046/j.0014-2956.2001.02701.x
Nelson J, Hanson CW, Koenig C, Chanton J (2011) Influence of diet on stable carbon isotope composition in otoliths of juvenile red drum Sciaenops ocellatus. Aquat Biol 13:89–95. https://doi.org/10.3354/ab00354
Nicolas M-T, Demêmes D, Martin A, Kupershmidt S, Barhanin J (2001) KCNQ1/KCNE1 potassium channels in mammalian vestibular dark cells. Hear Res 153:132–145. https://doi.org/10.1016/S0378-5955(00)00268-9
Pannella G (1971) Fish otoliths : daily growth layers and periodical patterns. Science 173:1124–1127
Payan P, Kossmann H, Watrin A, Mayer-Gostan N, Boeuf G (1997) Ionic composition of endolymph in teleosts: origin and importance of endolymph alkalinity. J Exp Biol 200:1905–1912
Payan P, Edeyer A, de Pontual H, Borelli G, Boeuf G, Mayer-Gostan N (1999) Chemical composition of saccular endolymph and otolith in fish inner ear: lack of spatial uniformity. Am J Physiol 277:R123–R131
Payan P, Borelli G, Priouzeau F, De Pontual H, Boeuf G, Mayer-Gostan N (2002) Otolith growth in trout Oncorhynchus mykiss: supply of Ca2+ and Sr2+ to the saccular endolymph. J Exp Biol 205:2687–2695
Payan P, De Pontual H, Bœuf G, Mayer-Gostan N (2004) Endolymph chemistry and otolith growth in fish. CR Palevol 3:535–547. https://doi.org/10.1016/j.crpv.2004.07.013
Pelster B (2001) The generation of hyperbaric oxygen tensions in fish. Physiology 16:287–291. https://doi.org/10.1152/physiologyonline.2001.16.6.287
Pimentel MS, Faleiro F, Dionisio G, Repolho T, Pousao-Ferreira P, Machado J et al (2014) Defective skeletogenesis and oversized otoliths in fish early stages in a changing ocean. J Exp Biol 217:2062–2070. https://doi.org/10.1242/jeb.092635
Pisam M, Payan P, LeMoal C, Edeyer A, Boeuf G, Mayer-Gostan N (1998) Ultrastructural study of the saccular epithelium of the inner ear of two teleosts, Oncorhynchus mykiss and Psetta maxima. Cell Tissue Res 294:261–270. https://doi.org/10.1007/s004410051176
Qin Z, Lewis JE, Perry SF (2010) Zebrafish (Danio rerio) gill neuroepithelial cells are sensitive chemoreceptors for environmental CO2. J Physiol 588:861–872. https://doi.org/10.1113/jphysiol.2009.184739
Radtke RL, Lenz P, Showers W, Moksness E (1996) Environmental information stored in otoliths: insights from stable isotopes. Mar Biol 127:161–170. https://doi.org/10.1007/BF00993656
Roa JN, Tresguerres M (2016) Soluble adenylyl cyclase is an acid-base sensor in epithelial base-secreting cells. Am J Physiol Cell Physiol 311:C340–C349. https://doi.org/10.1152/ajpcell.00089.2016
Roa JN, Tresguerres M (2017) Bicarbonate-sensing soluble adenylyl cyclase is present in the cell cytoplasm and nucleus of multiple shark tissues. Physiol Rep 5:e13090. https://doi.org/10.14814/phy2.13090
Roa JN, Munévar CL, Tresguerres M (2014) Feeding induces translocation of vacuolar proton ATPase and pendrin to the membrane of leopard shark (Triakis semifasciata) mitochondrion-rich gill cells. Comp Biochem Physiol Part A Mol Integr Physiol 174:29–37. https://doi.org/10.1016/j.cbpa.2014.04.003
Rummer JL, McKenzie DJ, Innocenti A, Supuran CT, Brauner CJ (2013) Root effect hemoglobin may have evolved to enhance general tissue oxygen delivery. Science 340:1327–1329. https://doi.org/10.1126/science.1233692
Saitoh S (1990) Localization and ultrastructure of mitochondria-rich cells in the inner ears of goldfish and tilapia. J Icthyology 37:49–55
Schade FM, Clemmesen C, Mathias Wegner K (2014) Within- and transgenerational effects of ocean acidification on life history of marine three-spined stickleback (Gasterosteus aculeatus). Mar Biol 161:1667–1676. https://doi.org/10.1007/s00227-014-2450-6
Shen SG, Chen F, Schoppik DE, Checkley DM (2016) Otolith size and the vestibulo-ocular reflex of larvae of white seabass Atractoscion nobilis at high pCO2. Mar Ecol Prog Ser 553:173–182. https://doi.org/10.3354/meps11791
Shiao JC, Lin LY, Horng JL, Hwang PP, Kaneko T (2005) How can teleostean inner ear hair cells maintain the proper association with the accreting otolith? J Comp Neurol 488:331–341. https://doi.org/10.1002/cne.20578
Swearer SE, Caselle JE, Lea DW, Warner RR (1999) Larval retention and recruitment in an island population of a coral-reef fish. Nature 402:799–802. https://doi.org/10.1038/45533
Takagi Y (1997) Meshwork arrangement of mitochondria-rich, Na+, K+-ATPase-rich cells in the saccular epithelium of rainbow trout (Oncorhynchus mykiss) inner ear. Anat Rec 248:483–489. https://doi.org/10.1002/(SICI)1097-0185(199708)248:4%3c483:AID-AR1%3e3.0.CO;2-N
Takagi Y (2002) Otolith formation and endolymph chemistry: a strong correlation between the aragonite saturation state and pH in the endolymph of the trout otolith organ. Mar Ecol Prog Ser 231:237–245. https://doi.org/10.3354/meps231237
Thomas ORB, Swearer SE, Kapp EA, Peng P, Tonkin-Hill GQ, Papenfuss A et al (2019) The inner ear proteome of fish. FEBS J 286:66–81. https://doi.org/10.1111/febs.14715
Tohse H, Mugiya Y (2001) Effects of enzyme and anion transport inhibitor on in vitro incorporation of inorganic carbon and calcium into endolymph and otoliths in salmon Oncorhynchus masou. Comp Biochem Physiol A Mol Integr Physiol 128:177–184
Tohse H, Mugiya Y (2008) Sources of otolith carbonate: experimental determination of carbon incorporation rates from water and metabolic CO2, and their diel variations. Aquat Biol 1:259–268. https://doi.org/10.3354/ab00029
Tohse H, Ando H, Mugiya Y (2004) Biochemical properties and immunohistochemical localization of carbonic anhydrase in the sacculus of the inner ear in the salmon Oncorhynchus masou. Comp Biochem Physiol A Mol Integr Physiol 137:87–94. https://doi.org/10.1016/S1095-6433(03)00272-1
Tohse H, Murayama E, Ohira T, Takagi Y, Nagasawa H (2006) Localization and diurnal variations of carbonic anhydrase mRNA expression in the inner ear of the rainbow trout Oncorhynchus mykiss. Comp Biochem Physiol B Biochem Mol Biol 145:257–264. https://doi.org/10.1016/j.cbpb.2006.06.011
Tresguerres M (2014) sAC from aquatic organisms as a model to study the evolution of acid/base sensing. Biochim Biophys Acta Mol Basis Dis 1842:2629–2635. https://doi.org/10.1016/j.bbadis.2014.06.021
Tresguerres M, Katoh F, Fenton H, Jasinska E, Goss GG (2005) Regulation of branchial V-H(+)-ATPase, Na(+)/K(+)-ATPase and NHE2 in response to acid and base infusions in the Pacific spiny dogfish (Squalus acanthias). J Exp Biol 208:345–354. https://doi.org/10.1242/jeb.01382
Tresguerres M, Buck J, Levin LR (2010a) Physiological carbon dioxide, bicarbonate, and pH sensing. Pflugers Arch Eur J Physiol 460:953–964. https://doi.org/10.1007/s00424-010-0865-6
Tresguerres M, Levin LR, Buck J, Grosell M (2010b) Modulation of NaCl absorption by [HCO3-] in the marine teleost intestine is mediated by soluble adenylyl cyclase. AJP Regul Integr Comp Physiol 299:R62–R71. https://doi.org/10.1152/ajpregu.00761.2009
Tresguerres M, Parks SK, Salazar E, Levin LR, Goss GG, Buck J (2010c) Bicarbonate-sensing soluble adenylyl cyclase is an essential sensor for acid/base homeostasis. Proc Natl Acad Sci USA 107:442–447. https://doi.org/10.1073/pnas.0911790107
von Biela V, Newsome S, Zimmerman C (2015) Examining the utility of bulk otolith δ13C to describe diet in wild-caught black rockfish Sebastes melanops. Aquat Biol 23:201–208. https://doi.org/10.3354/ab00621
Watanabe K, Ogawa A (1984) Carbonic anhydrase activity in stria vascularis and dark cells in vestibular labyrinth. Ann Otol Rhinol Laryngol 93:262–266. https://doi.org/10.1177/000348948409300315
Wilson JM, Randall DJ, Donowitz M, Vogl AW, Ip AK (2000) Immunolocalization of ion-transport proteins to branchial epithelium mitochondria-rich cells in the mudskipper (Periophthalmodon schlosseri). J Exp Biol 203:2297–2310
Wilson JM, Whiteley NM, Randall DJ (2002) Ionoregulatory changes in the gill epithelia of coho salmon during seawater acclimation. Physiol Biochem Zool 75:237–249. https://doi.org/10.1086/341817
Zdebik AA, Wangemann P, Jentsch TJ (2009) Potassium ion movement in the inner ear: Insights from genetic disease and mouse models. Physiology 24:307–316. https://doi.org/10.1152/physiol.00018.2009
Acknowledgements
GTK was supported by the National Science Foundation (NSF) Graduate Research Fellowship Program and the NSF Graduate Research Internship Program. This research was supported by grant NSF IOS #1754994 to M.T. We thank Jake Munich for his assistance in capturing Pacific Chub Mackerel. The authors would like to thank the two anonymous reviewers for their helpful comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Bernd Pelster.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kwan, G.T., Smith, T.R. & Tresguerres, M. Immunological characterization of two types of ionocytes in the inner ear epithelium of Pacific Chub Mackerel (Scomber japonicus). J Comp Physiol B 190, 419–431 (2020). https://doi.org/10.1007/s00360-020-01276-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-020-01276-3