Abstract
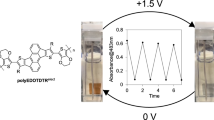
A kind of π-conjugated poly(EDOT-ethynylene-arylene)s P1–P5 were synthesized by Sonogashira coupling reaction between DBEDOT and five arylenes substituted with ethynyl groups. Among them, P5 was a polymer with TTF as pendants. Their TGA results exhibited their good thermal stability and their electrochemical and spectroscopic properties were investigated by CV, UV–Vis, and FL. The results indicated that there were intramolecular interactions between TTF group and polymer backbone as well as between the EDOT and arylene moieties. After doping with TCNQ, P5 could form stable CT-complex with a conductivity of 1.3 × 10−3 S cm−1, while its conductivity could be up to 3.5 × 10−2 S cm−1 after doping with I2, due to its increased conductive dimensions. Such results indicated that this polymer had potential application in developing conducting materials.
Graphical Abstract
A kind of π-conjugated EDOT-ethynylene-fluorene polymer with TTF as pendants was synthesized, which exhibited good solubility and thermal stability. When doping with I2, its conductivity was improved significantly due to its increased conductive dimensions.








Similar content being viewed by others
References
Roncali J (1992) Conjugated poly(thiophenes): synthesis, functionalization, and applications. Chem Rev 92:711–738
Roncali J (1997) Synthetic principles for bandgap control in linear π-conjugated systems. Chem Rev 97:173–206
Groenendaal LB, Zotti G, Aubert PH, Waybright SM, Reynolds JR (2003) Electrochemistry of poly(3,4-alkylenedioxythiophene) derivatives. Adv Mater 15:855–879
Groenendaal LB, Jonas F, Freitag D, Pielarzik H, Reynolds JR (2000) Poly(3,4-ethylenedioxythiophene) and its derivatives: past, present, and future. Adv Mater 12:481–494
Wang F, Wilson MS, Rauh D, Schottland P, Thompson BC, Reynolds JR (2000) Electrochromic linear and star branched poly(3,4-ethylenedioxythiophene-didodecyloxybenzene) polymers. Macromolecules 33:2083–2091
Bao Z, Chan W, Yu L (1993) Synthesis of conjugated polymer by the stille coupling reaction. Chem Mater 5:2–3
Aubert PH, Knipper M, Groenendaal L, Lutsen L, Manca J, Vanderzande D (2004) Copolymers of 3,4-ethylenedioxythiophene and of pyridine alternated with fluorene or phenylene units: synthesis, optical properties, and devices. Macromolecules 37:4087–4098
Segura JL, Martin M (2001) New concepts in tetrathiafulvalene chemistry. Angew Chem Int Ed 40:1372–1409
Inagi S, Naka K, Chujo Y (2007) Functional polymers based on electron-donating TTF and derivatives. J Mater Chem 17:4122–4135
Balog M, Rayah H, Derf FL, Sallé M (2008) A versatile building block for EDOT or PEDOT functionalization. New J Chem 32:1183–1188
Skabara PJ, Roberts DM, Serebryakov IM, Pozo-Gonzalo C (2000) The development of an electropolymerisable unit for TTF-thiophene fused monomers. Chem Commun 1005–1006
Liu YB, Wang CY, Li MJ, Lai GQ, Shen YJ (2008) Synthesis and spectroscopic and electrochemical properties of TTF-derivatized polycarbazole. Macromolecules 41:2045–2048
Liu YB, Wang CY, Li MJ, Lai GQ, Shen YJ (2008) Synthesis and properties of polysilanes with tetrathiafulvalene as pendant group. New J Chem 32:505–510
Huchet L, Akoudad S, Roncali J (1998) Electrosynthesis of highly electroactive tetrathiafulvalene-derivatized polythiophenes. Adv Mater 10:541–545
Huang YQ, Fan QL, Li SB, Lu XM, Cheng F, Zhang GW, Chen Y, Wang LH, Huang W (2006) Para-linked and meta-linked cationic water-soluble fluorene-containing poly(aryleneethynylene)s: conformational changes and their effects on iron–sulfur protein detection. J Polym Sci, Part A: Polym Chem 44:5424–5437
Huang YQ, Fan QL, Lu XM, Fang C, Liu SJ, Hui L, Wen Y, Wang L, Huang W (2006) Cationic, water-soluble, fluorene-containing poly(arylene-ethynylene)s: effects of water solubility on aggregation, photoluminescence efficiency, and amplified fluorescence quenching in aqueous solutions. J Polym Sci, Part A: Polym Chem 44:5778–5794
Huang YQ, Fan QL, Zhang GW, Chen Y, Lu XM, Huang W (2006) A fluorene-containing water-soluble poly(p-phenyleneethynylene) derivative: highly fluorescent and sensitive conjugated polymer with minor aggregation in aqueous solution. Polymer 47:5233–5238
Zhang XC, Wang CY, Lai GQ, Zhang L, Shen YJ (2010) Conjugated ethynylene–fluorene polymers with electro-donating TTF as pendant groups: synthesis, electrochemical and spectroscopic properties. New J Chem 34:318–324
Tran-Van F, Garreau S, Louarn G, Froyer G, Chevrot C (2001) Fully undoped and soluble oligo(3,4-ethylenedioxythiophene)s: spectroscopic study and electrochemical characterization. J Mater Chem 11:1378–1382
Wu YT, Bandera D, Maag R, Linden A, Baldridge KK, Siegel JS (2008) Multiethynyl corannulenes: synthesis, structure, and properties. J Am Chem Soc 130:10729–10739
Svenstrup N, Rasmussen KM, Hansen TK, Becher J (1994) The chemistry of TTFTT; 1: new efficient synthesis and reactions of tetrathiafulvalene-2,3,6,7-tetrathiolate (TTFTT): an important building block in TTF-syntheses. Synthesis 8:809–812
Christensen CA, Bryce MR, Becher J (2000) New multi(tetrathiafulvalene) dendrimers. Synthesis 12:1695–1704
Frenzel S, Arndt S, Ma R, Müllen K (1995) Synthesis of tetrathiafulvalene polymers. J Mater Chem 5:1529–1537
Casado J, Ortiz RP, Ruiz Delgado MC, Hernandez V, Lo’pez Na’varrete JT, Raimundo JM, Blanchard P, Allain M, Roncali J (2005) Alternated quinoid/aromatic units in terthiophenes building blocks for electroactive narrow band gap polymers. Extended spectroscopic, solid state, electrochemical, and theoretical study. J Phys Chem B 109:16616–16627
Guo XF, Zhang DQ, Zhang HJ, Fan QH, Xu W, Ai XC, Fan LZ, Zhu DB (2003) Donor–acceptor–donor triads incorporating tetrathiafulvalene and perylene diimide units: synthesis, electrochemical and spectroscopic studies. Tetrahedron 59:4843–4850
Skabara PJ, Berridge R, McInnes EJ, West DP, Coles SJ, Hursthouse MB, Müllen K (2004) The electroactivity of tetrathiafulvalene vs. polythiophene: synthesis and characterisation of a fused thieno–TTF polymer. J Mater Chem 14:1964–1969
Elsenbaumer RL, Jen KY, Oboodi R (1986) Processible and environmentally stable conducting polymers. Synth Met 15:169–174
Roehrich J, Müllen K (1992) A donor-type cyclophane with a strongly bent tetrathiafulvalene unit. J Org Chem 57:2374–2379
Acknowledgments
This study was supported by National Natural Science Foundation of China (No. 21076078 and 20872035).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, L., Li, M., Wang, C. et al. Conjugated poly(EDOT-ethynylene-fluorene) with TTF as pendants. Polym. Bull. 70, 353–369 (2013). https://doi.org/10.1007/s00289-012-0841-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-012-0841-8