Abstract
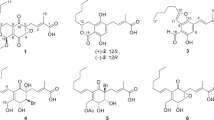
A library of 18 different compounds was synthesized starting from (R)-3-hydroxyoctanoic acid which is derived from the bacterial polymer polyhydroxyalkanoate (PHA). Ten derivatives, including halo and unsaturated methyl and benzyl esters, were synthesized and characterized for the first time. Given that (R)-3-hydroxyalkanoic acids are known to have biological activity, the new compounds were evaluated for antimicrobial activity and in vitro antiproliferative effect with mammalian cell lines. The presence of the carboxylic group was essential for the antimicrobial activity, with minimal inhibitory concentrations against a panel of bacteria (Gram-positive and Gram-negative) and fungi (Candida albicans and Microsporum gypseum) in the range 2.8–7.0 mM and 0.1–6.3 mM, respectively. 3-Halogenated octanoic acids exhibited the ability to inhibit C. albicans hyphae formation. In addition, (R)-3-hydroxyoctanoic and (E)-oct-2-enoic acids inhibited quorum sensing-regulated pyocyanin production in the opportunistic pathogen Pseudomonas aeruginosa PAO1. Generally, derivatives did not inhibit mammalian cell proliferation even at 3-mM concentrations, while only (E)-oct-2-enoic and 3-oxooctanoic acid had IC50 values of 1.7 and 1.6 mM with the human lung fibroblast cell line.







Similar content being viewed by others
References
Albert M, Seebach D, Duchardt E, Schwalbe H (2002) Synthesis and NMR analysis in solution of oligo(3-hydroxyalkanoic acid) derivatives with the side chains of alanine, valine, and leucine (β-depsides): coming full circle from PHB to β-peptides to PHB. Helv Chim Acta 85:633–658
Allen AD, Daley P, Ayorinde FO, Gugssa A, Anderson WA, Eribo BE (2012) Characterization of medium chain length (R)-3-hydroxycarboxylic acids produced by Streptomyces sp. JM3 and the evaluation of their antimicrobial properties. World J Microbiol Biotechnol 28:2791–2800
Anessian S, Dhanoa DS, Beaulieu PL (1987) Synthesis of carbocycles from omega-substituted a, alfa beta-unsaturated esters via radical-induced cyclizations. Can J Chem 65:1859
Arkin AH, Hazer B (2002) Chemical modification of chlorinated microbial polyesters. Biomacromolecules 3:1327–1335
Balducci E, Attolino E, Taddei M (2011) A stereoselective and practical synthesis of (E)-α,β-unsaturated ketones from aldehydes. Eur J Org Chem 2011:311–318
Brandl H, Gross RQ, Lenz RW, Fuller CR (1988) Pseudomonas oleovorans as a source of poly(β-hydroxyalkanoates) for potential applications as biodegradable polyesters. Appl Environ Microbiol 54:1977–1982
Brinkerhoff RC, Tarazona HF, de Oliveira PM, Flores DC, Montes D’Oca CDR, Russowsky D, Montes D’Oca MG (2014) Synthesis of β-ketoesters from renewable resources and Meldrum’s acid. RSC Advances 4:49556–49559
Brown HC, Ramachandran PV (1991) The boron approach to asymmetric synthesis. Pure Appl Chem 63:307–316
Case-Green SC, Davies SG, Roberts PM, Russell AJ, Thomson JE (2008) Asymmetric synthesis of tetrahydrolipstatin and valilactone. Tetrahedron Asymmetry 19:2620–2631
Casey JT, O’Cleirigh C, Walsh PK, O’Shea DG (2004) Development of a robust microtiter plate-based assay method for assessment of bioactivity. J Micro Meth 58:327–334
Chen G-Q (2009) A microbial polyhydroxyalkanoates (PHA) based bio- and materials industry. Chem Soc Rev 38:2434–2446
Chen G-Q (2011) Biofunctionalization of polymers and their applications. Adv Biochem Engin/Biotechnol 125:29–45
Chen G-Q, Wu Q (2005) The application of polyhydroxyalkanoates as tissue engineering materials. Biomaterials 26:6565–6578
Collins JC, Hess WW, Frank FJ (1968) Dipyridine-chromium(VI) oxide oxidation of alcohols in dichloromethane. Tetrahedron Lett 9:3363–3366
de Rijk TC, van de Meer P, Eggink G, Weusthuis RA (2001) Methods for analysis of poly(3-hydroxyalkanoate) composition. In: Doi Y, Steinbuchel A (eds) Biopolymers: polyesters. II Properties and chemical synthesis, Vol 2. Wiley-VCH, pp. pp 1–pp17
de Roo G, Kellerhals MB, Ren Q, Witholt B, Kessler B (2002) Production of chiral R-3-hydroxyalkanoic acids and R-3-hydroxyalkanoic acid methylesters via hydrolytic degradation of polyhydroxyalkanoate synthesized by pseudomonads. Biotechnol Bioeng 77:717–722
Delgado A, Ruiz M, Camps F, Hopital S, Guerrero A (1991) Synthesis of potential inhibitors of the biosynthesis of the sex pheromone of Spodoptera littoralis. Part I: Monofluorinated Fatty aCids Chem Phys Lipids 59:127–135
Deng Y, Boon C, Chen S, Lim A, Zhang L-H (2013) Cis-2-dodecenoic acid signal modulates virulence of Pseudomonas aeruginosa through interference with quorum sensing systems and T3SS. BMC Microbiol 13:231
Desbois AP, Lawlor KC (2013) Antibacterial activity of long-chain polyunsaturated fatty acids against Propionibacterium acnes and Staphylococcus aureus. Mar Drugs 11:4544–4557
Desbois AP, Smith VJ (2010) Antibacterial free fatty acids: activities, mechanisms of action and biotechnological potential. Appl Microbiol Biotechnol 85:1629–1642
Desmaris L, Percina N, Cottier L, Sinou D (2003) Conversion of alcohols to bromides using a fluorous phosphine. Tetrahedron Lett 44:7589–7591
Doi Y, Abe C (1990) Biosynthesis and characterization of a new bacterial copolyester of 3-hydroxyalkanoates and 3-hydroxy-ω-chloroalkanoates. Macromolecules 23:3705–3707
El-Batta A, Jiang C, Zhao W, Anness R, Cooksy AL, Bergdahl M (2007) Wittig reactions in water media employing stabilized ylides with aldehydes. Synthesis of α,β-unsaturated esters from mixing aldehydes, α-bromoesters, and Ph3P in aqueous NaHCO3. J Org Chem 72:5244–5259
Elbahloul Y, Steinbuchel A (2009) Large-scale production of poly(3-hydroxyoctanoic acid) by Pseudomonas putida GPo1 and a simplified downstream process. Appl Environ Microbiol 75:643–651
Follonier S, Goyder MS, Silvestri AC, Crelier S, Kalman F, Riesen R, Zinn M (2014) Fruit pomace and waste frying oil as sustainable resources for the bioproduction of medium-chain-length polyhydroxyalkanoates. Int J Biol Macromol 71:42–52
Gibson J, Sood A, Hogan DA (2009) Pseudomonas aeruginosa-Candida albicans interactions: localization and fungal toxicity of a phenazine derivative. Appl Environ Microbiol 75:504–513
Giddens AC, Nielsen L, Boshoff HI, Tasdemir D, Perozzo R, Kaiser M, Wang F, Sacchettini JC, Copp BR (2008) Natural product inhibitors of fatty acid biosynthesis: synthesis of the marine microbial metabolites pseudopyronines A and B and evaluation of their anti-infective activities. Tetrahedron 64:1242–1249
Grage K, Jahns AC, Parlane N, Palanisamy R, Rasaih IA, Atwood JA, Rehm BHA (2009) Bacterial polyhydroxyalkanoate granules: biogenesis, structure, and potential use as nano-/micro-beads in biotechnological and biomedical applications. Biomacromolecules 10:660–669
Hansen MB, Nielsen SE, Berg K (1989) Re-examination and further development of a precise and rapid dye method for measuring cell growth/cell kill. J Immunol Meth 119:203–210
Hon Y-S, Hsieh C-H, Liu Y-W (2005) Dibromomethane as one-carbon source in organic synthesis: total synthesis of (±)- and (â)-methylenolactocin. Tetrahedron 61:2713–2723
Kabara J, Swieczkowski DM, Conley AJ, Truant JP (1972) Fatty acids and derivatives as antimicrobial agents. Antimicrob Agents Chemotherapy 2:23–28
Kalia VC, Purohit HJ (2011) Quenching the quorum sensing system: potential antibacterial drug targets. Crit Rev Microbiol 37:121–140
Lau GW, Hassett DJ, Ran H, Kong F (2004) The role of pyocyanin in Pseudomonas aeruginosa infection. Trends Mol Med 10:599–606
Lee SY, Lee Y-H, Wang F (1999) Chiral compounds from bacterial polyesters: sugars to plastics to fine chemicals. Biotechnol Bioeng 65:363–368
Marounek M, Skrivanova E, Rada V (2003) Susceptibility of Escherichia coli to C2-C18 fatty acids. Folia Microbiol 48:731–735
Marris E (2006) Marine natural products: drugs from the deep. Nature 443:904–905
Morales DK, Grahl N, Okegbe C, Dietrich LE, Jacobs NJ, Hogan DA (2013) Control of Candida albicans metabolism and biofilm formation by Pseudomonas aeruginosa phenazines. MBio 4:e00526–e00512
Mordant C, Dunkelmann P, Ratovelomanana-Vidal V, Genet J-P (2004) Dynamic kinetic resolution: an efficient route to anti α-amino-β-hydroxy esters via Ru-SYNPHOS catalyzed hydrogenation. Chem Comm 2004:1296–1297
Nikodinovic-Runic J, Guzik M, Kenny ST, Babu R, Werker A, O’Connor K (2013) Carbon-rich wastes as feedstocks for biodegradable polymer (polyhydroxyalkanoate) production using bacteria. Adv Appl Microbiol 84:139–200
Noyori R, Kitamura M, Ohkuma T (2004) Toward efficient asymmetric hydrogenation: architectural and functional engineering of chiral molecular catalysts. P Natl Acad Sci USA 101:5356–5362
O’Connor S, Szwej E, Nikodinovic-Runic J, O’Connor A, Byrne AT, Devocelle M, O’Donovan N, Gallagher WM, Babu R, Kenny S, Zinn M, Zulian QR, O’Connor KE (2013) The anti-cancer activity of a cationic anti-microbial peptide derived from monomers of polyhydroxyalkanoate. Biomaterials 34:2710–2718
O’Loughlin CT, Miller LC, Siryaporn A, Drescher K, Semmelhack MF, Bassler B (2013) A quorum-sensing inhibitor blocks Pseudomonas aeruginosa virulence and biofilm formation. P Natl Acad Sci USA 110:17981–17986
Ren Q, Grubelnik A, Hoerler M, Ruth K, Hartmann R, Felber H, Zinn M (2005) Bacterial poly(hydroxyalkanoates) as a source of chiral hydroxyalkanoic acids. Biomacromolecules 6:2290–2298
Ren Q, Ruth K, Thony-Meyer L, Zinn M (2007) Process engineering for production of chiral hydroxycarboxylic acids from bacterial polyhydroxyalkanoates. Macromol Rapid Commun 28:2131–2136
Ren Q, Ruth K, Thony-Meyer L, Zinn M (2010) Enatiomerically pure hydroxycarboxylic acids: current approaches and future perspectives. Appl Microbiol Biotechnol 87:41–52
Ruth K, Grubelnik A, Hartmann R, Egli T, Zinn M, Ren Q (2007) Efficient production of (R)-3-hydroxycarboxylic acids by biotechnological conversion of polyhydroxyalkonoates and their purification. Biomacromolecules 8:279–286
Sailer M, Dubicki KI, Sorensen JL (2015) The synthesis of medium-chain-length β-hydroxy esters via the Reformatsky reaction. Synthesis 47:79–82
Sandoval A, Arias-Barrau E, Bermejo F, Canedo L, Naharro G, Olivera E, Luengo J (2005) Production of 3-hydroxy-n-phenylalkanoic acids by a genetically engineered strain of Pseudomonas putida. Appl Microbiol Biotechnol 67:97–105
SDBS Web, http://sdbs.db.aist.go.jp (National Institute of Advanced Industrial Science and Technology, date of access 12/10/2014).
Shafreen RM, Muthamil S, Pandian SK (2014) Inhibition of Candida albicans virulence factors by novel levofloxacin derivatives. Appl Microbiol Biotechnol 98:6775–6785
Skrivanova E, Savka OG, Marounek M (2004) In vitro effect of C2-C18 fatty acids on salmonellas. Folia Microbiol 49:199–202
Smith SM, Uteuliyev M, Takacs JM (2011) Catalytic asymmetric hydroboration of β,γ-unsaturated Weinreb amides: striking influence of the borane. Chem Comm 47:7812–7814
Steinbüchel A, Valentin HE (1995) Diversity of bacterial polyhydroxyalkanoic acids. FEMS Microbiol Lett 128:219–228
Szwej E, Devocelle M, Kenny S, Guzik M, O’Connor S, Nikodinovic-Runic J, Radivojevic J, Maslak V, Byrne AT, Gallagher WM, Zulian QR, Zinn M, O’Connor KE (2015) The chain length of biologically produced (R)-3-hydroxyalkanoic acid affects biological activity and structure of anti-cancer peptides. J Biotechnol 204:7–12
Takashi M, Daisuke I, Akira A (2003) Process for producing optically active 3-halogenocarboxylic acid esters and 3-azide-carboxylic acid esters. EPO Patent 1344763-A1.
Tortajada M, da Silva LF, Prieto MA (2013) Second-generation functionalized medium-chain-length polyhydroxyalkanoates: the gateway to high-value bioplastic applications. Int Microbiol 16:1–15
Wang Z, Zhao C, Pierce ME, Fortunak JM (1999) Enantioselective synthesis of β-hydroxy carboxylic acids: direct conversion of β-oxocarboxylic acids to enantiomerically enriched β-hydroxy carboxylic acids via neighboring group control. Tetrahedron: Asymm 10:225–228
Watanabe S, Fujita T, Sakamoto M, Arai T, Kttazume T (1989) Fluorination of hydroxyesters with N,N-diethyl-1,1,2,3,3,3-hexafluoro propaneamine. J Am Oil Chem Soc 66:1312–1315
Watanabe S, Fujita T, Suga K, Nasuno I (1983) Fluorination of fatty alcohols with 1,1,2,3,3,3-hexafluoropropyl diethylamine. J Oil Fat Ind 60:1678–1679
Whooley MA, McLoughlin AJ (1982) The regulation of pyocyanin production in Pseudomonas aeruginosa. Eur J Appl Microbiol Biotechnol 15:161–166
Widmer KW, Soni KA, Hume ME, Beier RC, Jesudhasan P, Pillai SD (2007) Identification of poultry meat-derived fatty acids functioning as quorum sensing signal inhibitors to autoinducer-2 (AI-2). J Food Sci 72:M363–M368
Acknowledgments
This work has been financially supported by the Ministry of Education and Science, Republic of Serbia, under Grant No. 173048. JR was partially supported by research project BP2013 (Bioplastech Ltd., Dublin, Ireland).
Conflict of interest
The authors declare that they have no competing interests.
Compliance with ethical standards
ᅟ
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
ESM 1
Electronic Supplementary Information (ESI) available: [Spectral Data 1H NMR, 13C NMR and IR]. (PDF 1614 kb)
Rights and permissions
About this article
Cite this article
Radivojevic, J., Skaro, S., Senerovic, L. et al. Polyhydroxyalkanoate-based 3-hydroxyoctanoic acid and its derivatives as a platform of bioactive compounds. Appl Microbiol Biotechnol 100, 161–172 (2016). https://doi.org/10.1007/s00253-015-6984-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6984-4