Abstract
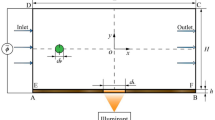
Insulator-based dielectrophoresis (iDEP), an efficient technique with great potential for miniaturization, has been successfully applied for the manipulation of a wide variety of bioparticles. When iDEP is applied employing direct current (DC) electric fields, other electrokinetic transport mechanisms are present: electrophoresis and electroosmotic flow. In order to concentrate particles, iDEP has to overcome electrokinetics. This study presents the characterization of electrokinetic flow under the operating conditions employed with iDEP; in order to identify the optimal conditions for particle concentration employing DC-iDEP, microparticle image velocimetry (μPIV) was employed to measure the velocity of 1-μm-diameter inert polystyrene particles suspended inside a microchannel made from glass. Experiments were carried out by varying the properties of the suspending medium (conductivity from 25 to 100 μS/cm and pH from 6 to 9) and the strength of the applied electric field (50–300 V/cm); the velocities values obtained ranged from 100 to 700 μm/s. These showed that higher conductivity and lower pH values for the suspending medium produced the lowest electrokinetic flow, improving iDEP concentration of particles, which decreases voltage requirements. These ideal conditions for iDEP trapping (pH = 6 and σ m = 100 μS/cm) were tested experimentally and with the aid of mathematical modeling. The μPIV measurements allowed obtaining values for the electrokinetic mobilities of the particles and the zeta potential of the glass surface; these values were used with a mathematical model built with COMSOL Multiphysics software in order to predict the dielectrophoretic and electrokinetic forces exerted on the particles; the modeling results confirmed the μPIV findings. Experiments with iDEP were carried out employing the same microparticles and a glass microchannel that contained an array of cylindrical insulating structures. By applying DC electric fields across the insulating structures array, it was seen that the dielectrophoretic trapping was improved when the electrokinetic force was the lowest; as predicted by μPIV measurements and the mathematical model. The results of this study provide guidelines for the selection of optimal operating conditions for improving insulator-based dielectrophoretic separations and have the potential to be extended to bioparticle applications.

Comparison of experimental measurements and mathematical modeling of electrokinetic and dielectrophoretic effects on microparticles






Similar content being viewed by others
Abbreviations
- AC:
-
Alternating current
- DEP:
-
Dielectrophoresis
- DC:
-
Direct current
- EOF:
-
Electroosmotic flow
- EP:
-
Electrophoresis
- EK:
-
Electrokinetic
- PIV:
-
Particle imaging velocimetry
References
Duffy DC, McDonald JC, Schueller OJA, Whitesides GM (1998) Anal Chem 70:4974–4984
Rheea M, Burns MA (2008) Lab Chip 8:1365–1373
Dittrich PS, Tachikawa K, Manz A (2006) Anal Chem 78:3887–3907
Andersson H, van den Berg A (2003) Sens Actuator B-Chem 92:315–325
Escarpa A, González MC, López-Gil MA, Crevillén AG, Hervás M, García M (2008) Electrophoresis 29:4852–4861
Chung TD, Kim HC (2007) Electrophoresis 28:4511–4520
Hughes MP (2002) Nanoelectromechanics in engineering and biology. CRC, Boca Raton, FL
Castellanos A, Ramos A, González A, Green NG, Morgan H (2003) J Phys D: Appl Phys 36:2584–2597
Chou CF, Tegenfeldt JO, Bakajin O, Chan SS, Cox EC, Darnton N, Duke T, Austin RH (2002) Biophys J 83:2170–2179
Chou CF, Zenhausern F (2003) IEEE Eng Med Biol Mag 22:62–67
Sabounchi P, Morales AM, Ponce P, Lee LP, Simmons BA, Davalos R (2008) Biomed Microdev 10:661–670
Cummings EB, Singh AK (2003) Anal Chem 75:4724–4731
Lapizco-Encinas BH, Simmons BA, Cummings EB, Fintschenko Y (2004) Anal Chem 76:1571–1579
Davalos RV, McGraw GJ, Wallow TI, Morales AM, Krafcik KL, Cummings EB, Simmons BA (2008) Anal Bioanal Chem 390:847–855
Simmons BA, McGraw GJ, Davalos RV, Fiechtner GJ, Fintschenko Y, Cummings EB (2006) MRS Bull 31:120–124
Kwon J-S, Maeng J-S, Chun M-S, Song S (2008) Microfluid Nanofluid 5:23–31
Pysher MD, Hayes MA (2007) Anal Chem 79:4552–4557
Jones TB (1995) Electromechanics of particles. Cambridge University Press, USA
Pohl HA (1951) J Appl Phys 22:869–871
Markx GH, Dyda PA, Pethig R (1996) J Biotechnol 51:175–180
Ozuna-Chacón S, Lapizco-Encinas BH, Rito-Palomares M, Martínez-Chapa SO, Reyes-Betanzo C (2008) Electrophoresis 29:3115–3122
Hawkins BG, Smith AE, Syed YA, Kirby BJ (2007) Anal Chem 79:7291–7300
Johnson AM, Sadoway DR, Cima M, Langera R (2005) J Electrochem Soc 152:H6–H11
Green NG, Morgan H (1999) J Phys Chem B 103:41–50
Sabounchi P, Huber DE, Kanouff MP, Harris AE, Simmons BA (2008) The 12th International Conference on Miniaturized Systems for Chemistry and Life Sciences (MicroTAS 2008), San Diego CA, October 12–16, 2008
Hayes MA, Ketherpal I, Ewing AG (1993) Anal Chem 65:27–31
Kirby BJ, Hasselbrink EF (2004) Electrophoresis 25:187–202
Acknowledgments
The authors would like to acknowledge the financial support provided by the grant CONACYT-CB-2006-53603. The authors are grateful for the financial support provided by Cátedras de Investigación (CAT142 and CAT161) of Tecnológico de Monterrey.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martínez-López, J.I., Moncada-Hernández, H., Baylon-Cardiel, J.L. et al. Characterization of electrokinetic mobility of microparticles in order to improve dielectrophoretic concentration. Anal Bioanal Chem 394, 293–302 (2009). https://doi.org/10.1007/s00216-009-2626-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-009-2626-y