Abstract
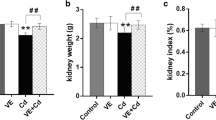
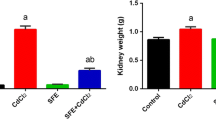
Nephrotoxicity is recognized as a serious disorder affected by chronic cadmium exposure. Imbalance between radical generation and elimination is considered a critical factor involved in the initiation and progression of renal injury caused by this heavy metal. The present study investigated the possible protection by catechin, a natural phenolic antioxidant, against cadmium nephrotoxicity and elucidated its potential mechanism. Male Wistar rats were assigned to receive vehicle, cadmium (CdCl2 2 mg/kg, i.p.) and cadmium plus catechin (25, 50, and 100 mg/kg, orally, respectively). After 4 weeks of treatment, rats exposed to cadmium demonstrated a marked rise in blood urea nitrogen and creatinine, a fall in creatinine clearance, and renal pathologies like severe tubular damage, apoptosis, and abnormal mitochondrial structure. Significant increases in malondialdehyde, nitric oxide, and tumor necrosis factor-alpha, while reductions in antioxidant thiols, superoxide dismutase, and catalase, were also detected in the kidney tissues of cadmium-intoxicated rats. These alterations were associated with mitochondrial dysfunction as supported by an increase in mitochondrial reactive oxygen species production and a decline in mitochondrial membrane potential. Treatment with catechin significantly attenuated all the changes caused by cadmium. These findings suggest that catechin effectively protects the kidney against toxic effect of cadmium, presumably through its antioxidant, anti-inflammation, and mitochondrial protection. The study outcomes not only add evidence to reinforce the medical benefits of catechin but also, most importantly, give rise to a prospect of developing renal preventive strategy for individuals who are at risk of cadmium contamination by means of catechin supplementation.






Similar content being viewed by others
References
Alshatwi AA, Hasan TN, Alqahtani AM, Syed NA, Shafi G, Al-Assaf AH, Al-Khalifa AS (2014) Delineating the anti-cytotoxic and anti-genotoxic potentials of catechin hydrate against cadmium toxicity in human peripheral blood lymphocytes. Environ Toxicol Pharmacol 38(2):653–662. https://doi.org/10.1016/j.etap.2014.07.013
Anjaneyulu M, Tirkey N, Chopra K (2003) Attenuation of cyclosporine-induced renal dysfunction by catechin: possible antioxidant mechanism. Ren Fail 25(5):691–707. https://doi.org/10.1081/JDI-120024285
Blaser H, Dostert C, Mak TW, Brenner D (2016) TNF and ROS crosstalk in inflammation. Trends Cell Biol 26(4):249–261. https://doi.org/10.1016/j.tcb.2015.12.002
Chen L, Zhou J, Gao W, Jiang YZ (2003) Action of NO and TNF-α release of rats with cadmium loading in malfunction of multiple system organ. Sheng Li Xue Bao 55:535–540
Chen Q, Zhang R, Li W, Niu Y, Guoc H, Liu X, Hou Y, Zhao L (2013) The protective effect of grape seed procyanidin extract against cadmium-induced renal oxidative damage in mice. Environ Toxicol Pharmacol 36(3):759–768. https://doi.org/10.1016/j.etap.2013.07.006
Chopra K, Singh D, Chander V (2004) Nephrotoxicity and its prevention by catechin in ferric nitrilotriacetate promoted oxidative stress in rats. Hum Exp Toxicol 23(3):137–143. https://doi.org/10.1191/0960327104ht427oa
Crespy V, Williamson G (2004) A review of the health effects of green tea catechins in in vivo animal models. J Nutr 134(12 Suppl):3431S–3440S
Cuypers A, Plusquin M, Remans T, Jozefczak M, Keunen E, Gielen H, Opdenakker K, Nair AR, Munters E, Artois TJ, Nawrot T, Vangronsveld J, Smeets K (2010) Cadmium stress: an oxidative challenge. Biometals 23(5):927–940. https://doi.org/10.1007/s10534-010-9329
Gheysarzadeh A, Yazdanparast R (2015) STAT5 reactivation by catechin modulates H2O2-induced apoptosis through miR-182/FOXO1 pathway in SK-N-MC cells. Cell Biochem Biophys 71(2):649–656. https://doi.org/10.1007/s12013-014-0244-6
Gobe G, Crane D (2010) Mitochondria, reactive oxygen species and cadmium toxicity in the kidney. Toxicol Lett 198(1):49–55. https://doi.org/10.1016/j.toxlet.2010.04.013
Grzesik M, Naparło K, Bartosz G, Sadowska-Bartosz I (2018) Antioxidant properties of catechins: comparison with other antioxidants. Food Chem 241:480–492. https://doi.org/10.1016/j.foodchem.2017.08.117
Johri N, Jacquillet G, Unwin R (2010) Heavy metal poisoning: the effects of cadmium on the kidney. Biometals 23(5):783–792. https://doi.org/10.1007/s10534-010-9328-y
Kobroob A, Chattipakorn N, Wongmekiat O (2012) Caffeic acid phenethyl ester ameliorates cadmium-induced kidney mitochondrial injury. Chem Biol Interact 200(1):21–27. https://doi.org/10.1016/j.cbi.2012.08.026
Kozlov AV, Lancaster JR, Meszaros AT, Weidinger A (2017) Mitochondria-meditated pathways of organ failure upon inflammation. Redox Biol 13:170–181. https://doi.org/10.1016/j.redox.2017.05.017
Li X, Jiang X, Sun J, Zhu C, Li X, Tian L, Liu L, Bai W (2017) Cytoprotective effects of dietary flavonoids against cadmium-induced toxicity. Ann N Y Acad Sci 1398(1):5–19. https://doi.org/10.1111/nyas.13344
Mehaffey E, Majid DSA (2017) Tumor necrosis factor-α, kidney function, and hypertension. Am J Physiol Renal Physiol 313:F1005–F1008. https://doi.org/10.1152/ajprenal.00535.2016
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2679(79)90738-3
Parvez S, Tabassum H, Rehmana H, Banerjee BD, Athar M, Raisuddin S (2006) Catechin prevents tamoxifen-induced oxidative stress and biochemical perturbations in mice. Toxicology 225(2-3):109–118. https://doi.org/10.1016/j.tox.2006.05.009
Peerapanyasut W, Thamprasert K, Wongmekiat O (2014) Ubiquinol supplementation protects against renal ischemia and reperfusion injury in rats. Free Radic Res 48(2):180–189. https://doi.org/10.3109/10715762.2013.858148
Poontawee W, Natakankitkul S, Wongmekiat O (2016) Protective effect of Cleistocalyx nervosum var. paniala fruit extract against oxidative renal damage caused by cadmium. Molecules 21(2):133–145. https://doi.org/10.3390/molecules21020133
Rani A, Kumar A, Lal M, Pant M (2014) Cellular mechanisms of cadmium-induced toxicity: a review. Int J Environ Health Res 24(4):378–399. https://doi.org/10.1080/09603123.2013.835032
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25(1):192–205. https://doi.org/10.1016/0003-2679(68)90092-4
Shahid A, Ali R, Ali N, Hasan SK, Bernwal P, Afzal SM, Vafa A, Sultana S (2016) Modulatory effects of catechin hydrate against genotoxicity, oxidative stress, inflammation and apoptosis induced by benzo(a)pyrene in mice. Food Chem Toxicol 92:64–74. https://doi.org/10.1016/j.fct.2016.03.021
Silva Santos LF, Stolfo A, Calloni C, Salvador M (2017) Catechin and epicatechin reduce mitochondrial dysfunction and oxidative stress induced by amiodarone in human lung fibroblasts. J Arrhythm 33(3):220–225. https://doi.org/10.1016/j.joa.2016.09.004
Szuster-Ciesielska A, Lokaj I, Kandefer-Szerszen M (2000) The influence of cadmium and zinc ions on the interferon and tumor necrosis factor production in bovine aorta endothelial cells. Toxicology 145(2-3):135–145. https://doi.org/10.1016/S0300-483X(00)00147-5
Tabassum H, Parvez S, Rehman H, Banerjee BD, Raisuddin S (2007) Catechin as an antioxidant in liver mitochondrial toxicity: inhibition of tamoxifen-induced protein oxidation and lipid peroxidation. J Biochem Mol Toxicol 21(3):110–117. https://doi.org/10.1002/jbt.20167
Thevenod F (2003) Nephrotoxicity and the proximal tubule. Insights from cadmium. Nephron Physiol 93(4):87–93. https://doi.org/10.1159/000070241
Valko M, Jomova K, Rhodes CJ, Kuča K, Musílek K (2016) Redox- and non-redox-metal-induced formation of free radicals and their role in human disease. Arch Toxicol 90(1):1–37. https://doi.org/10.1007/s00204-015-1579-5
Vazquez Prieto MA, Bettaieb A, Rodriguez Lanzi C, Soto VC, Perdicaro DJ, Galmarini CR, Haj FG, Miatello RM, Oteiza PI (2015) Catechin and quercetin attenuate adipose inflammation in fructose-fed rats and in 3T3-L1 adipocytes. Mol Nutr Food Res 59(4):622–633. https://doi.org/10.1002/mnfr.201400631
Walker JM (1994) The bicinchoninic acid (BCA) assay for protein quantitation. Methods Mol Biol 32:5–8. https://doi.org/10.1385/0-89603-268-X:5
Zhai W, Zheng J, Yao X, Peng B, Liu M, Huang J, Wang G, Xu Y (2013) Catechin prevents the calcium oxalate monohydrate induced renal calcium crystallization in NRK-52E cells and the ethylene glycol induced renal stone formation in rat. BMC Complemen Altern Med 13(1):228–251. https://doi.org/10.1186/1472-6882-13-228
Zhang T, Mu Y, Yang M, Al Maruf A, Li P, Li C, Dai S, Lu J, Dong Q (2017) (+)-Catechin prevents methylglyoxal-induced mitochondrial dysfunction and apoptosis in EA. hy926 cells. Arch Physiol Biochem 123(2):121–127. https://doi.org/10.1080/13813455.2016.1263868
Funding
This study was supported by the Faculty of Medicine Endowment Fund for Research, Chiang Mai University, Chiang Mai, Thailand (Grant Number: 034/2557).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards and approved by the Ethical Animal Research Committee of Chiang Mai University (project number 34/2556).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wongmekiat, O., Peerapanyasut, W. & Kobroob, A. Catechin supplementation prevents kidney damage in rats repeatedly exposed to cadmium through mitochondrial protection. Naunyn-Schmiedeberg's Arch Pharmacol 391, 385–394 (2018). https://doi.org/10.1007/s00210-018-1468-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-018-1468-6