Abstract
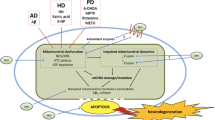
Mitochondria are the powerhouse of the eukaryotic cell through their use of oxidative phosphorylation to generate ATP. Mitochondrial dysfunction is considered an important contributing factor in a variety of physiopathological situations such as aging, heart ischemia/reperfusion injury, diabetes and several neurodegenerative and cardiovascular diseases, as well as in cell death. Increased formation of reactive oxygen species, altered respiratory chain complexes activity and opening of the mitochondrial permeability transition pore have been suggested as possible factors responsible for impaired mitochondrial function. Therefore, preventing mitochondrial dysfunction could be an effective therapeutic strategy against cellular degenerative processes. Cardiolipin is a unique phospholipid located at the level of inner mitochondrial membrane where it plays an important role in mitochondrial bioenergetics, as well as in cell death. Cardiolipin abnormalities have been associated with mitochondrial dysfunction in a variety of pathological conditions and aging. Melatonin, the major secretory product of the pineal gland, is a well-known antioxidant agent and thus an effective protector of mitochondrial bioenergetic function. Melatonin was reported to prevent mitochondrial dysfunction from oxidative damage by preserving cardiolipin integrity, and this may explain, at least in part, the beneficial effect of this compound in mitochondrial physiopathology. In this article, mechanisms through which melatonin exerts its protective role in mitochondrial dysfunction and related disorders are reviewed.
Similar content being viewed by others
References
Abuja PM, Liebmann P, Hayn M, Schauenstein K, Esterbauer H (1997) Antioxidant role of melatonin in lipid peroxidation of human LDL. FEBS Lett 413:289–293
Acuña Castroviejo D, López LC, Escames G, López A, García JA, Reiter RJ (2011) Melatonin-mitochondria interplay in health and disease. Curr Top Med Chem 11:221–240
Acuna-Castroviejo D, Escames G, Rodriguez MI, Lopez LC (2007) Melatonin role in the mitochondrial function. Front Biosci 12:947–963
Acuña-Castroviejo D, Coto-Montes A, Gaia Monti M, Ortiz GG, Reiter RJ (1997) Melatonin is protective against MPTP-induced striatal and hippocampal lesions. Life Sci 60:PL23–PL29
Acuña-Castroviejo D, Escames G, León J, Carazo A, Khaldy H (2003) Mitochondrial regulation by melatonin and its metabolites. Adv Exp Med Biol 527:549–557
Aliev G, Seyidova D, Lamb BT, Obrenovich ME, Siedlak SL, Vinters HV, Friedland RP, LaManna JC, Smith MA, Perry G (2003) Mitochondria and vascular lesions as a central target for the development of Alzheimer’s disease and Alzheimer disease-like pathology in transgenic mice. Neurol Res 25:665–674
Anderson S, Bankier AT, Barrell BG, de Bruijn MH, Coulson AR, Drouin J, Eperon IC, Nierlich DP, Roe BA, Sanger F, Schreier PH, Smith AJ, Staden R, Young IG (1981) Sequence and organization of the human mitochondrial genome. Nature 290:457–465
Andrabi SA, Sayeed I, Siemen D, Wolf G, Horn TF (2004) Direct inhibition of the mitochondrial permeability transition pore: a possible mechanism responsible for anti-apoptotic effects of melatonin. FASEB J 18:869–871
Antolín I, Rodríguez C, Saínz RM, Mayo JC, Uría H, Kotler ML, Rodríguez-Colunga MJ, Tolivia D, Menéndez-Peláez A (1996) Neurohormone melatonin prevents cell damage: effect on gene expression for antioxidant enzymes. FASEB J 10:882–890
Ban T, Heymann JA, Song Z, Hinshaw JE, Chan DC (2010) OPA1 disease alleles causing dominant optic atrophy have defects in cardiolipin-stimulated GTP hydrolysis and membrane tubulation. Hum Mol Genet 19:2113–2122
Bayir H, Kapralov AA, Jiang J, Huang Z, Tyurina YY, Tyurin VA, Zhao Q, Belikova NA, Vlasova II, Maeda A, Zhu J, Na HM, Mastroberardino PG, Sparvero LJ, Amoscato AA, Chu CT, Greenamyre JT, Kagan VE (2009) Peroxidase mechanism of lipid-dependent cross-linking of synuclein with cytochrome C: protection against apoptosis versus delayed oxidative stress in Parkinson disease. J Biol Chem 284:15951–15969
Bazán S, Mileykovskaya E, Mallampalli VK, Heacock P, Sparagna GC, Dowhan W (2013) Cardiolipin-dependent reconstitution of respiratory supercomplexes from purified Saccharomyces cerevisiae complexes III and IV. J Biol Chem 288:401–411
Beal MF (1998) Mitochondrial dysfunction in neurodegenerative diseases. Biochim Biophys Acta 1366:211–223
Beal MF (2005) Mitochondria take center stage in aging and neurodegeneration. Ann Neurol 58:495–505
Bondy SC, Sharman EH (2007) Melatonin and the aging brain. Neurochem Int 50:571–580
Bondy SC, Lahiri DK, Perreau VM, Sharman KZ, Campbell A, Zhou J, Sharman EH (2004) Retardation of brain aging by chronic treatment with melatonin. Ann N Y Acad Sci 1035:197–215
Boveris A, Chance B (1973) The mitochondrial generation of hydrogen peroxide. General properties and effect of hyperbaric oxygen. Biochem J 134:707–716
Boveris A, Navarro A (2008) Brain mitochondrial dysfunction in aging. IUBMB Life 60:308–314
Büeler H (2009) Impaired mitochondrial dynamics and function in the pathogenesis of Parkinson’s disease. Exp Neurol 218:235–246
Caballero B, Vega-Naredo I, Sierra V, Huidobro-Fernández C, Soria-Valles C, De Gonzalo-Calvo D, Tolivia D, Gutierrez-Cuesta J, Pallas M, Camins A, Rodríguez-Colunga MJ, Coto-Montes A (2008) Favorable effects of a prolonged treatment with melatonin on the level of oxidative damage and neurodegeneration in senescence-accelerated mice. J Pineal Res 45:302–311
Camara AK, Bienengraeber M, Stowe DF (2011) Mitochondrial approaches to protect against cardiac ischemia and reperfusion injury. Front Physiol 2:1–34
Cardinali DP, Pagano ES, Bernasconi PAS, Reynoso R, Scacchi P (2013) Melatonin and mitochondrial dysfunction in the central nervous system. Horm Behav 63:322–330
Carretero M, Escames G, López LC, Venegas C, Dayoub JC, García L, Acuña-Castroviejo D (2009) Long-term melatonin administration protects brain mitochondria from aging. J Pineal Res 47:192–200
Casley CS, Land JM, Sharpe MA, Clark JB, Duchen MR, Canevari L (2002) Beta-amyloid fragment 25–35 causes mitochondrial dysfunction in primary cortical neurons. Neurobiol Dis 10:258–267
Catala´ A (2007) The ability of melatonin to counteract lipid peroxidation in biological membranes. Curr Mol Med 7:638–649
Ceraulo L, Ferrugia M, Tesoriere L, Segreto S, Livrea MA, Liveri VT (1999) Interactions of melatonin with membrane models: portioning of melatonin in AOT and lecithin reversed micelles. J Pineal Res 26:108–112
Chen JX, Yan SS (2010) Role of mitochondrial amyloid-beta in Alzheimer’s disease. J Alzheimers Dis 20(Suppl 2):S569–S578
Chen YR, Zweier JL (2014) Cardiac mitochondria and reactive oxygen species generation. Circ Res 114:524–537
Chicco AJ, Sparagna GC (2007) Role of cardiolipin alterations in mitochondrial dysfunction and disease. Am J Physiol Cell Physiol 292:C33–C44
Claypool SM (2009) Cardiolipin, a critical determinant of mitochondrial carrier protein assembly and function. Biochim Biophys Acta 1788:2059–2068
Clementi ME, Marini S, Coletta M, Orsini F, Giardina B, Misiti F (2005) Abeta (31–35) and Abeta (25–35) fragments of amyloid beta-protein induce cellular death through apoptotic signals: role of the redox state of methionine-35. FEBS Lett 579:2913–2918
Crompton M (1999) The mitochondrial permeability transition pore and its role in cell death. Biochem J 341:233–249
Crompton M (2004) Mitochondria and aging: a role for the permeability transition? Aging Cell 3:3–6
Cuzzocrea S, Zingarelli B, Gilad E, Hake P, Salzman AL, Szabó C (1997) Protective effect of melatonin in carrageenan-induced models of local inflammation: relationship to its inhibitory effect on nitric oxide production and its peroxynitrite scavenging activity. J Pineal Res 23:106–116
DeVay RM, Dominguez-Ramirez L, Lackner LL, Hoppins S, Stahlberg H, Nunnari J (2009) Coassembly of Mgm1 isoforms requires cardiolipin and mediates mitochondrial inner membrane fusion. J Cell Biol 186:793–803
DiMauro S, Schon EA (2003) Mitochondrial respiratory-chain diseases. N Engl J Med 348:2656–2668
Dominguez-Rodriguez A, Abreu-Gonzalez P (2010) Myocardial ischemia-reperfusion injury: possible role of melatonin. World J Cardiol 2:233–236
Dominguez-Rodriguez A, Abreu-Gonzalez P, Avanzas P (2012) The role of melatonin in acute myocardial infarction. Front Biosci 17:2433–2441
Dong W, Huang F, Fan W, Cheng S, Chen Y, Zhang W, Shi H, He H (2010) Differential effects of melatonin on amyloid-beta peptide 25–35-induced mitochondrial dysfunction in hippocampal neurons at different stages of culture. J Pineal Res 48:117–125
Eble KS, Coleman WB, Hantgan RR, Cunningham CC (1990) Tightly associated cardiolipin in the bovine heart mitochondrial ATP synthase as analyzed by 31P nuclear magnetic resonance spectroscopy. J Biol Chem 265:19434–19440
Ellis CE, Murphy EJ, Mitchell DC, Golovko MY, Scaglia F, Barceló-Coblijn GC, Nussbaum RL (2005) Mitochondrial lipid abnormality and electron transport chain impairment in mice lacking alpha-synuclein. Mol Cell Biol 25:10190–10201
Escames G, López A, García JA, García L, Acuña-Castroviejo D, García JJ, López LC (2010) The role of mitochondria in brain aging and the effects of melatonin. Curr Neuropharmacol 8:182–193
Feng Z, Qin C, Chang Y, Zhang JT (2006) Early melatonin supplementation alleviates oxidative stress in a transgenic mouse model of Alzheimer’s disease. Free Radic Biol Med 40:101–109
Fowler G, Daroszewska M, Ingold KU (2003) Melatonin does not “directly scavenge hydrogen peroxide”: demise of another myth. Free Radic Biol Med 34:77–83
Fry M, Green M (1981) Cardiolipin requirement for electron transfer in complex I and III of the mitochondrial respiratory chain. J Biol Chem 256:1874–1880
García JJ, López-Pingarrón L, Almeida-Souza P, Tres A, Escudero P, García-Gil FA, Tan DX, Reiter RJ, Ramírez JM, Bernal-Pérez M (2014) Protective effects of melatonin in reducing oxidative stress and in preserving the fluidity of biological membranes: a review. J Pineal Res 56:225–237
Gautier CA, Corti O, Brice A (2014) Mitochondrial dysfunctions in Parkinson’s disease. Rev Neurol 170:339–343
Genova ML, Lenaz G (2014) Functional role of mitochondrial respiratory supercomplexes. Biochim Biophys Acta 1837:427–443
Ghafourifar P, Richter C (1997) Nitric oxide synthase activity in mitochondria. FEBS Lett 418:291–296
Giacomo CG, Antonio M (2007) Melatonin in cardiac ischemia/reperfusion-induced mitochondrial adaptive changes. Cardiovasc Hematol Disord Drug Targets 7:163–169
Giorgio V, von Stockum S, Antoniel M, Fabbro A, Fogolari F, Forte M, Glick GD, Petronilli V, Zoratti M, Szabó I, Lippe G, Bernardi P (2013) Dimers of mitochondrial ATP synthase form the permeability transition pore. Proc Natl Acad Sci USA 110:5887–5892
Giulivi C, Poderoso JJ, Boveris A (1998) Production of nitric oxide by mitochondria. J Biol Chem 273:11038–11043
Gómez LA, Hagen TM (2012) Age-related decline in mitochondrial bioenergetics: does supercomplex destabilization determine lower oxidative capacity and higher superoxide production? Semin Cell Dev Biol 23:758–767
Gonzalvez F, Gottlieb E (2007) Cardiolipin: setting the beat of apoptosis. Apoptosis 12:877–885
Halestrap AP (2009) What is the mitochondrial permeability transition pore? J Mol Cell Cardiol. 46:821–831
Hardeland R (2005) Antioxidative protection by melatonin: multiplicity of mechanisms from radical detoxification to radical avoidance. Endocrine 27:119–130
Hardeland R (2013) Melatonin and the theories of aging: a critical appraisal of melatonin’s role in antiaging mechanisms. J Pineal Res 55:325–356
Hardeland R, Fuhrberg B (1996) Ubiquitous melatonin presence and effects in unicells, plants and animals. Trends Comp Biochem 2:25–44
Hardeland R, Pandi-Perumal SR, Cardinali DP (2006) Melatonin. Int J Biochem Cell Biol 3:313–316
Harman D (1972) The biologic clock: the mitochondria? J Am Geriatr Soc 20:145–147
Hekimi S, Lapointe J, Wen Y (2011) Taking a “good” look at free radicals in the aging process. Trends Cell Biol 21:569–576
Hibaoui Y, Roulet E, Ruegg UT (2009) Melatonin prevents oxidative stress-mediated mitochondrial permeability transition and death in skeletal muscle cells. J Pineal Res 47:238–252
Hoch FL (1992) Cardiolipins and biomembrane function. Biochim Biophys Acta 1113:71–133
Houtkooper RH, Vaz FM (2008) Cardiolipin, the heart of mitochondrial metabolism. Cell Mol Life Sci 65:2493–2506
Jiang J, Huang Z, Zhao Q, Feng W, Belikova NA, Kagan VE (2008) Interplay between bax, reactive oxygen species production, and cardiolipin oxidation during apoptosis. Biochem Biophys Res Commun 368:145–150
Jou MJ (2011) Melatonin preserves the transient mitochondrial permeability transition for protection during mitochondrial Ca(2+) stress in astrocyte. J Pineal Res 50:427–435
Judge S, Leeuwenburgh C (2007) Cardiac mitochondrial bioenergetics, oxidative stress, and aging. Am J Physiol Cell Physiol 292:C1983–C1992
Kagan VE, Borisenko GG, Tyurina YY, Tyurin VA, Jiang J, Potapovich AI, Kini V, Amoscato AA, Fujii Y (2004) Oxidative lipidomics of apoptosis: redox catalytic interactions of cytochrome c with cardiolipin and phosphatidylserine. Free Radic Biol Med 37:1963–1985
Kagan VE, Tyurin VA, Jiang J, Tyurina YY, Ritov VB, Amoscato AA, Osipov AN, Belikova NA, Kapralov AA, Kini V, Vlasova II, Zhao Q, Zou M, Di P, Svistunenko DA, Kurnikov IV, Borisenko GG (2005) Cytochrome c acts as a cardiolipin oxygenase required for release of proapoptotic factors. Nat Chem Biol 1:223–232
Kajstura J, Cheng W, Sarangarajan R, Li P, Li B, Nitahara JA, Chapnick S, Reiss K, Olivetti G, Anversa P (1996) Necrotic and apoptotic myocyte cell death in the aging heart of Fischer 344 rats. Am J Physiol 271:H1215–H1228
Kirkwood TB (2005) Understanding the odd science of aging. Cell 120:437–447
Klingenberg M (2009) Cardiolipin and mitochondrial carriers. Biochim Biophys Acta 1788:2048–2058
Korshunov SS, Skulachev VP, Starkov AA (1997) High protonic potential actuates a mechanism of production of reactive oxygen species in mitochondria. FEBS Lett 416:15–18
Lange C, Nett JH, Trumpower BL, Hunte C (2001) Specific roles of protein-phospholipid interactions in the yeast cytochrome bc1 complex structure. EMBO J 20:6591–6600
Lenaz G, Genova ML (2010) Structure and organization of mitochondrial respiratory complexes: a new understanding of an old subject. Antioxid Redox Signal 12:961–1008
Lenaz G, Bovina C, Castelluccio C, Fato R, Formiggini G, Genova ML, Marchetti M, Pich MM, Pallotti F, Castelli GP, Biagini G (1997) Mitochondrial complex I defects in aging. Mol Cell Biochem 174:329–333
Leon J, Acuña-Castroviejo D, Sainz RM, Mayo JC, Tan DX, Reiter RJ (2004) Melatonin and mitochondrial function. Life Sci 7:765–790
Leung AW, Halestrap AP (2008) Recent progress in elucidating the molecular mechanism of the mitochondrial permeability transition pore. Biochim Biophys Acta 1777:946–952
Levine RL, Stadtman ER (2001) Oxidative modification of proteins during aging. Exp Gerontol 36:1495–1502
Livrea MA, Tesoriere L, D’arpa D, Morreale M (1997) Reaction of melatonin with lipoperoxyl radicals in phospholipid bilayers. Free Radic Biol Med 5:706–711
Lochner A, Huisamen B, Nduhirabandi F (2013) Cardioprotective effect of melatonin against ischaemia/reperfusion damage. Front Biosci 5:305–315
López A, García JA, Escames G, Venegas C, Ortiz F, López LC, Acuña-Castroviejo D (2009) Melatonin protects the mitochondria from oxidative damage reducing oxygen consumption, membrane potential, and superoxide anion production. J Pineal Res 46:188–198
Madesh M, Hajnoczky G (2001) VDAC-dependent permeabilization of the outer mitochondrial membrane by superoxide induces rapid and massive cytochrome c release. J Cell Biol 155:1003–1015
Maharaj DS, Maharaj H, Daya S, Glass BD (2006) Melatonin and 6-hydroxymelatonin protect against iron-induced neurotoxicity. J Neurochem 1:78–81
Mander P, Brown GC (2004) Nitric oxide, hypoxia and brain inflammation. Biochem Soc Trans 32:1068–1069
Marom M, Safonov R, Amram S, Avneon Y, Nachliel E, Gutman M, Zohary K, Azem A, Tsfadia Y (2009) Interaction of the Tim44 C-terminal domain with negatively charged phospholipids. Biochemistry 48:11185–11195
Martín M, Macías M, Escames G, Reiter RJ, Agapito MT, Ortiz GG, Acuña-Castroviejo D (2000) Melatonin-induced increased activity of the respiratory chain complexes I and IV can prevent mitochondrial damage induced by ruthenium red in vivo. J Pineal Res 28:242–248
Martín M, Macías M, León J, Escames G, Khaldy H, Acuña-Castroviejo D (2002) Melatonin increases the activity of the oxidative phosphorylation enzymes and the production of ATP in rat brain and liver mitochondria. Int J Biochem Cell Biol 34:348–357
Massaad CA, Pautler RG, Klann E (2009) Mitochondrial superoxide: a key player in Alzheimer’s disease. Aging (Albany NY) 1:758–761
Mather M, Rottenberg H (2000) Aging enhances the activation of the permeability transition pore in mitochondria. Biochem Biophys Res Commun 273:603–608
Mekhloufi J, Bonnefont-Rousselot D, Yous S, Lesieur D, Couturier M, Thérond P, Legrand A, Jore D, Gardès-Albert M (2005) Antioxidant activity of melatonin and a pinoline derivative on linoleate model system. J Pineal Res 39:27–33
Melov S, Adlard PA, Morten K, Johnson F, Golden TR, Hinerfeld D, Schilling B, Mavros C, Masters CL, Volitakis I, Li QX, Laughton K, Hubbard A, Cherny RA, Gibson B, Bush AI (2007) Mitochondrial oxidative stress causes hyperphosphorylation of tau. Plos One 2:1–12
Menendez-Pelaez A, Reiter RJ (1993) Distribution of melatonin in mammalian tissues: the relative importance of nuclear versus cytosolic localization. J Pineal Res 15:59–69
Messner M, Hardeland R, Rodenbeck A, Huether G (1998) Tissue retention and subcellular distribution of continuously infused melatonin in rats under near physiological conditions. J Pineal Res 25:251–259
Mileykovskaya E, Dowhan W (2014) Cardiolipin-dependent formation of mitochondrial respiratory supercomplexes. Chem Phys Lipids 179:42–48
Miquel J, Economos AC, Fleming J, Johnson JEJR (1980) Mitochondrial role in cell aging. Exp Gerontol 15:575–591
Mizuno Y, Ohta S, Tanaka M, Takamiya S, Suzuki K, Sato T, Oya H, Ozawa T, Kagawa Y (1989) Deficiencies in complex I subunits of the respiratory chain in Parkinson’s disease. Biochem Biophys Res Commun. 163:1450–1455
Mullin S, Schapira AH (2015) Pathogenic mechanisms of neurodegeneration in Parkinson disease. Neurol Clin 33:1–17
Murphy MP (2009) How mitochondria produce reactive oxygen species. Biochem J 417:1–13
Musatov A, Robinson NC (2012) Susceptibility of mitochondrial electron-transport complexes to oxidative damage. Focus on cytochrome c oxidase. Free Radic Res 46:1313–1326
Navarro A, Boveris A (2007) The mitochondrial energy transduction system and the aging process. Am J Physiol Cell Physiol 292:C670–C686
Navarro-Alarcón M, Ruiz-Ojeda FJ, Blanca-Herrera RM, A-Serrano MM, Acuña-Castroviejo D, Fernández-Vázquez G, Agil A (2014) Melatonin and metabolic regulation: a review. Food Funct 5:2806–2832
Nohl H, Stolze K (1992) Ubisemiquinones of the mitochondrial respiratory chain do not interact with molecular oxygen. Free Radic Res Commun 16:409–419
Ong SB, Samangoueia P, Kalkhorana SB, Hausenloy DJ (2015) The mitochondrial permeability transition pore and its role in myocardial ischemia reperfusion injury. J Mol Cell Cardiol 78:23–34
Ortiz GG, Crespo-López ME, Morán-Moguel C, García JJ, Reiter RJ, Acuña-Castroviejo D (2001) Protective role of melatonin against MPTP-induced mouse brain cell DNA fragmentation and apoptosis in vivo. Neuro Endocrinol Lett 22:101–108
Ott M, Gogvadze V, Orrenius S, Zhivotovsky B (2007a) Mitochondria, oxidative stress and cell death. Apoptosis 12:913–922
Ott M, Zhivotovsky B, Orrenius S (2007b) Role of cardiolipin in cytochrome c release from mitochondria. Cell Death Differ 14:1243–1247
Ozawa T, Tanaka M, Wakabayashi T (1982) Crystallization of mitochondrial cytochrome oxidase. Proc Natl Acad Sci USA 79:7175–7179
Öztürk G, Akbulut KG, Güney Ş, Acuna-Castroviejo D (2012) Age-related changes in the rat brain mitochondrial antioxidative enzyme ratios: modulation by melatonin. Exp Gerontol 47:706–711
Pak JW, Herbst A, Bua E, Gokey N, McKenzie D, Aiken JM (2003) Mitochondrial DNA mutations as a fundamental mechanism in physiological declines associated with aging. Aging Cell 2:1–7
Pamplona R (2008) Membrane phospholipids, lipoxidative damage and molecular integrity: a causal role in aging and longevity. Biochim Biophys Acta 1777:1249–1262
Pandi-Perumal SR, BaHammam AS, Brown GM, Spence DW, Bharti VK, Kaur C, Hardeland R, Cardinali DP (2013) Melatonin antioxidative defense: therapeutical implications for aging and neurodegenerative processes. Neurotox Res 23:267–300
Paradies G, Petrosillo G, Pistolese M, Ruggiero FM (2000) The effect of reactive oxygen species generated from the mitochondrial electron transport chain on the cytochrome c oxidase activity and on the cardiolipin content in bovine heart submitochondrial particles. FEBS Lett 466:323–326
Paradies G, Petrosillo G, Pistolese M, Ruggiero FM (2002) Reactive oxygen species affect mitochondrial electron transport complex I activity through oxidative cardiolipin damage. Gene 286:135–141
Paradies G, Petrosillo G, Pistolese M, Di Venosa N, Federici A, Ruggiero FM (2004) Decrease in mitochondrial complex I activity in ischemic/reperfused rat heart: involvement of reactive oxygen species and cardiolipin. Circ Res 94:53–59
Paradies G, Petrosillo G, Paradies V, Ruggiero FM (2009) Role of cardiolipin peroxidation and Ca2+ in mitochondrial dysfunction and disease. Cell Calcium 45:643–650
Paradies G, Petrosillo G, Paradies V, Reiter RJ, Ruggiero FM (2010a) Melatonin, cardiolipin and mitochondrial bioenergetics in health and disease. J Pineal Res 4:297–310
Paradies G, Petrosillo G, Paradies V, Ruggiero FM (2010b) Oxidative stress, mitochondrial bioenergetics, and cardiolipin in aging. Free Radic Biol Med 48:1286–1295
Paradies G, Petrosillo G, Paradies V, Ruggiero FM (2011) Mitochondrial dysfunction in brain aging: role of oxidative stress and cardiolipin. Neurochem Int 58:447–457
Paradies G, Paradies V, Ruggiero FM, Petrosillo G (2013) Changes in the mitochondrial permeability transition pore in aging and age-associated diseases. Mech Ageing Dev 134:1–9
Paradies G, Paradies V, De Benedictis V, Ruggiero FM, Petrosillo G (2014a) Functional role of cardiolipin in mitochondrial bioenergetics. Biochim Biophys Acta 1837:408–417
Paradies G, Paradies V, Ruggiero FM, Petrosillo G (2014b) Cardiolipin and mitochondrial function in health and disease. Antioxid Redox Signal 20:1925–1953
Paradies G, Paradies V, Ruggiero FM, Petrosillo G (2014c) Oxidative stress, cardiolipin and mitochondrial dysfunction in nonalcoholic fatty liver disease. World J Gastroenterol 20:14205–14218
Parlakpinar H, Sahna E, Ozer MK, Ozugurlu F, Vardi N, Acet A (2002) Physiological and pharmacological concentrations of melatonin protect against cisplatin-induced acute renal injury. J Pineal Res 33:161–166
Patki G, Lau YS (2011) Melatonin protects against neurobehavioral and mitochondrial deficits in a chronic mouse model of Parkinson’s disease. Pharmacol Biochem Behav 99:704–711
Perier C, Tieu K, Guegan C, Caspersen C, Jackson-Lewis V, Carelli V, Martinuzzi A, Hirano M, Przedborski S, Vila M (2005) Complex I deficiency primes Bax-dependent neuronal apoptosis through mitochondrial oxidative damage. Proc Natl Acad Sci USA 102:19126–19131
Petrosillo G, Ruggiero FM, Pistolese M, Paradies G (2001) Reactive oxygen species generated from the mitochondrial electron transport chain induce cytochrome c dissociation from beef-heart submitochondrial particles via cardiolipin peroxidation. Possible role in the apoptosis. FEBS Lett 509:435–438
Petrosillo G, Ruggiero FM, Di Venosa N, Paradies G (2003a) Decreased complex III activity in mitochondria isolated from rat heart subjected to ischemia and reperfusion: role of reactive oxygen species and cardiolipin. FASEB J 17:714–716
Petrosillo G, Ruggiero FM, Paradies G (2003b) Role of reactive oxygen species and cardiolipin in the release of cytochrome c from mitochondria. FASEB J 15:2202–2208
Petrosillo G, Casanova G, Matera M, Ruggiero FM, Paradies G (2006a) Interaction of peroxidized cardiolipin with rat-heart mitochondrial membranes: induction of permeability transition and cytochrome c release. FEBS Lett 580:6311–6316
Petrosillo G, Di Venosa N, Pistolese M, Casanova G, Tiravanti E, Colantuono G, Federici A, Paradies G, Ruggiero FM (2006b) Protective effect of melatonin against mitochondrial dysfunction associated with cardiac ischemia- reperfusion: role of cardiolipin. FASEB J. 20:269–276
Petrosillo G, Fattoretti P, Matera M, Ruggiero FM, Bertoni-Freddari C, Paradies G (2008a) Melatonin prevents age-related mitochondrial dysfunction in rat brain via cardiolipin protection. Rejuvenation Res 11:935–943
Petrosillo G, Matera M, Casanova G, Ruggiero FM, Paradies G (2008b) Mitochondrial dysfunction in rat brain with aging: involvement of complex I, reactive oxygen species and cardiolipin. Neurochem Int 53:126–131
Petrosillo G, Colantuono G, Moro N, Ruggiero FM, Tiravanti E, Di Venosa N, Fiore T, Paradies G (2009a) Melatonin protects against heart ischemia-reperfusion injury by inhibiting mitochondrial permeability transition pore opening. Am J Physiol Heart Circ Physiol 297:H1487–H1493
Petrosillo G, Matera M, Moro N, Ruggiero FM, Paradies G (2009b) Mitochondrial complex I dysfunction in rat heart with aging: critical role of reactive oxygen species and cardiolipin. Free Radic Biol Med 46:88–94
Petrosillo G, Moro N, Ruggiero FM, Paradies G (2009c) Melatonin inhibits cardiolipin peroxidation in mitochondria and prevents the mitochondrial permeability transition and cytochrome c release. Free Radic Biol Med 47:969–974
Petrosillo G, Moro N, Paradies V, Ruggiero FM, Paradies G (2010) Increased susceptibility to Ca(2+)-induced permeability transition and to cytochrome c release in rat heart mitochondria with aging: effect of melatonin. J Pineal Res 48:340–346
Pieri C, Marra M, Moroni F, Recchioni R, Marcheselli F (1994) Melatonin: a peroxyl radical scavenger more effective than vitamin E. Life Sci 55:PL271–PL276
Pieri C, Marra M, Gaspar R, Damjanovich S (1996) Melatonin protects LDL from oxidation but does not prevent the apolipoprotein derivatization. Biochem Biophys Res Commun 2:256–260
Pranke IM, Morello V, Bigay J, Gibson K, Verbavatz JM, Antonny B, Jackson CL (2011) α-Synuclein and ALPS motifs are membrane curvature sensors whose contrasting chemistry mediates selective vesicle binding. J Cell Biol 194:89–103
Protter D, Lang C, Cooper AA (2012) α-Synuclein and mitochondrial dysfunction: a pathogenic partnership in Parkinson’s disease? Parkinsons Dis 2012:829207
Raha S, Robinson BH (2000) Mitochondria, oxygen free radicals, disease and ageing. Trends Biochem Sci 25:502–508
Reiter RJ, Richardson BA, Johnson LY (1980) Pineal melatonin rhythm: reduction in aging Syrian hamsters. Science 210:1372–1373
Reiter RJ, Craft CM, Johnson JE (1981) Age-associated reduction in nocturnal pineal melatonin levels in female rats. Endocrinology 109:1295–1297
Reiter RJ, Tan D, Kim SJ, Manchester LC, Qi W, Garcia JJ, Cabrera JC, El-Sokkary G, Rouvier-Garay V (1999) Augmentation of indices of oxidative damage in life-long melatonin-deficient rats. Mech Ageing Dev 110:157–173
Reiter RJ, Tan DX, Osuna C, Gitto E (2000) Actions of melatonin in the reduction of oxidative stress. A review. J Biomed Sci 7:444–458
Reiter RJ, Paredes SD, Korkmaz A, Jou MJ, Tan DX (2008) Melatonin combats molecular terrorism at the mitochondrial level. Interdiscip Toxicol 2:137–149
Reiter RJ, Tan DX, Galano A (2014) Melatonin reduces lipid peroxidation and membrane viscosity. Front Physiol 5:377–380
Ren M, Phoon CK, Schlame M (2014) Metabolism and function of mitochondrial cardiolipin. Prog Lipid Res 55:1–16
Robinson NC (1993) Functional binding of cardiolipin to cytochrome c oxidase. J Bioenerg Biomembr 25:153–163
Rodríguez MI, Carretero M, Escames G, López LC, Maldonado MD, Tan DX, Reiter RJ, Acuña-Castroviejo D (2007) Chronic melatonin treatment prevents age-dependent cardiac mitochondrial dysfunction in senescence-accelerated mice. Free Radic Res 41:15–24
Rodríguez MI, Escames G, López LC, López A, García JA, Ortiz F, Sánchez V, Romeu M, Acuña-Castroviejo D (2008) Improved mitochondrial function and increased life span after chronic melatonin treatment in senescent prone mice. Exp Gerontol 43:749–756
Rubbo H, Radi R, Trujillo M, Telleri R, Kalyanaraman B, Barnes S, Kirk M, Freeman BA (1994) Nitric oxide regulation of superoxide and peroxynitrite-dependent lipid peroxidation. J Biol Chem 269:26066–26075
Rytomaa M, Mustonen P, Kinnunen PK (1992) Reversible, nonionic, and pH-dependent association of cytochrome c with cardiolipin-phosphatidylcholine liposomes. J Biol Chem 267:22243–22248
Sahna E, Parlakpinar H, Turkoz Y, Acet A (2005) Protective effects of melatonin on myocardial ischemia/reperfusion induced infarct size and oxidative changes. Physiol Res 5(4):91–95
Santos RX, Correia SC, Wang X, Perry G, Smith MA, Moreira PI, Zhu X (2010) Alzheimer’s disease: diverse aspects of mitochondrial malfunctioning. Int J Clin Exp Pathol 3:570–581
Saravanan KS, Sindhu KM, Mohanakumar KP (2007) Melatonin protects against rotenone-induced oxidative stress in a hemiparkinsonian rat model. J Pineal Res 42:247–253
Schagger H (2002) Respiratory chain supercomplexes of mitochondria and bacteria. Biochim Biophys Acta 1555:154–159
Schapira AH, Cooper JM, Dexter D, Clark JB, Jenner P, Marsden CD (1990) Mitochondrial complex I deficiency in Parkinson’s disease. J Neurochem 54:823–827
Schlame M, Ren M (2009) The role of cardiolipin in the structural organization of mitochondrial membranes. Biochim Biophys Acta 1788:2080–2083
Schlame M, Rua D, Greenberg ML (2000) The biosynthesis and functional role of cardiolipin. Prog Lipid Res 39:257–288
Schon EA, DiMauro S, Hirano M, Gilkerson RW (2010) Therapeutic prospects for mitochondrial disease. Trends Mol Med 16:268–276
Sharma R, McMillan CR, Tenn CC, Niles LP (2006) Physiological neuroprotection by melatonin in a 6-hydroxydopamine model of Parkinson’s disease. Brain Res 1068:230–236
Sharpley MS, Shannon RJ, Draghi F, Hirst J (2006) Interactions between phospholipids and NADH: ubiquinone oxidoreductase (complex I) from bovine mitochondria. Biochemistry 45:241–248
Shen J, Du T, Wang X, Duan C, Gao G, Zhang J, Lu L, Yang H (2014) α-Synuclein amino terminus regulates mitochondrial membrane permeability. Brain Res 1591:14–26
Skulachev VP (1996) Role of uncoupled and non-coupled oxidations in maintenance of safely low levels of oxygen and its one-electron reductants. Q Rev Biophys 29:169–202
Srinivasan V, Cardinali DP, Srinivasan US, Kaur C, Brown GM, Spence DW, Hardeland R, Pandi-Perumal SR (2011a) Therapeutic potential of melatonin and its analogs in Parkinson’s disease: focus on sleep and neuroprotection. Ther Adv Neurol Disord 4:297–317
Srinivasan V, Spence DW, Pandi-Perumal SR, Brown GM, Cardinali DP (2011b) Melatonin in mitochondrial dysfunction and related disorders. Int J Alzheimers Dis 2011:1–16
Stadtman ER (2002) Importance of individuality in oxidative stress and aging. Free Radic Biol Med 33:597–604
Stadtman ER, Levine RL (2003) Free radical-mediated oxidation of free amino acids and amino acid residues in proteins. Amino Acids 25:207–218
Takeda T (1999) Senescence-accelerated mouse (SAM): a biogerontological resource in aging research. Neurobiol Aging 20:105–110
Tan DX, Pöeggeler B, Reiter RJ, Chen LD, Chen S, Manchester LC, Barlow-Walden LR (1993) The pineal hormone melatonin inhibits DNA-adduct formation induced by the chemical carcinogen safrole in vivo. Cancer Lett 70:65–71
Tan DX, Manchester LC, Reiter RJ, Plummer BF, Hardies LJ, Weintraub ST, Vijayalaxmi, Shepherd AM (1998) A novel melatonin metabolite, cyclic 3-hydroxymelatonin: a biomarker of in vivo hydroxyl radical generation. Biochem Biophys Res Commun 253:614–620
Tan DX, Manchester LC, Reiter RJ, Plummer BF, Limson J, Weintraub ST, Qi W (2000) Melatonin directly scavenges hydrogen peroxide: a potentially new metabolic pathway of melatonin biotransformation. Free Radic Biol Med 29:1177–1185
Tan DX, Reiter RJ, Manchester LC, Yan MT, El-Sawi M, Sainz RM, Mayo JC, Kohen R, Allegra M, Hardeland R (2002) Chemical and physical properties and potential mechanisms: melatonin as a broad spectrum antioxidant and free radical scavenger. Curr Top Med Chem 2:181–197
Tan DX, Manchester LC, Terron MP, Flores LJ, Reiter RJ (2007) One molecule, many derivatives: a never-ending interaction of melatonin with reactive oxygen and nitrogen species? J Pineal Res 1:28–42
Teixeira A, Morfim MP, de Cordova CA, Charão CC, de Lima VR, Creczynski-Pasa TB (2003) Melatonin protects against pro-oxidant enzymes and reduces lipid peroxidation in distinct membranes induced by the hydroxyl and ascorbyl radicals and by peroxynitrite. J Pineal Res 35:262–268
Turrens JF, Alexandre A, Lehninger AL (1985) Ubisemiquinone is the electron donor for superoxide formation by complex III of heart mitochondria. Arch Biochem Biophys 237:408–414
Urata Y, Honma S, Goto S, Todoroki S, Iida T, Cho S, Honma K, Kondo T (1999) Melatonin induces gamma-glutamylcysteine synthetase mediated by activator protein-1 in human vascular endothelial cells. Free Radic Biol Med 27:838–847
Wallace DC (1999) Mitochondrial diseases in man and mouse. Science 283:1482–1488
Wang X (2009) The antiapoptotic activity of melatonin in neurodegenerative diseases. CNS Neurosci Ther 15:345–357
Wei YH, Lee HC (2002) Oxidative stress, mitochondrial DNA mutation, and impairment of antioxidant enzymes in aging. Exp Biol Med 227:671–682
Yang Y, Sun Y, Yi W, Li Y, Fan C, Xin Z, Jiang S, Di S, Qu Y, Reiter RJ, Yi D (2014) A review of melatonin as a suitable antioxidant against myocardial ischemia-reperfusion injury and clinical heart diseases. J Pineal Res 57:357–366
Zhang M, Mileykovskaya E, Dowhan W (2002) Gluing the respiratory chain together. Cardiolipin is required for supercomplex formation in the inner mitochondrial membrane. J Biol Chem 277:43553–43556
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Paradies, G., Paradies, V., Ruggiero, F.M. et al. Protective role of melatonin in mitochondrial dysfunction and related disorders. Arch Toxicol 89, 923–939 (2015). https://doi.org/10.1007/s00204-015-1475-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-015-1475-z