Abstract
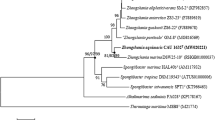
In the course of screening for bacterial predators, a Gram-stain-negative, non-flagellated, gliding, long rod-shaped, and yellow-pigmented bacterium, designated strain HICWT, was isolated from coastal seawater of China. Phylogenetic analysis based on 16S rRNA gene sequences indicated that strain HICWT represented a member of the genus Muricauda and showed the highest sequence similarity to M. aquimarina JCM11811T (98.8%) and M. ruestringensis DSM13258T (98.1%). The average nucleotide identity (ANI) and digital DNA–DNA hybridization (dDDH) values between strain HICWT and M. aquimarina JCM11811T were 79.2% and 34.1%, respectively. NaCl was required for growth. Optimum growth occurred at 25–30 °C, 2.0–3.0% (w/v) NaCl with pH 7.0. Strain HICWT showed some similar characteristics to the nonobligate bacterial predators, and the cells can attach to the prey cells. Strain HICWT contained MK-6 as the predominant respiratory quinone and had iso-C15:0, iso-C15:1 G, and iso-C17:0 3-OH as the major cellular fatty acids. The polar lipids contained phosphatidylethanolamine (PE), three unidentified phospholipids (PL1–PL3), one unidentified amino lipids (AL), and three unidentified polar lipids (L1–L3). The genome size of strain HICWT was approximately 3.8 Mbp, with a G + C content of 41.4%. Based on the polyphasic evidence, strain HICWT is proposed as representing a new species of the genus Muricauda, for which the name Muricauda chongwuensis sp. nov. is proposed. The type strain is HICWT (= JCM 33643 T = MCCC 1K03769T).



Similar content being viewed by others
Data availability
The GenBank/EMBL/DDBJ accession numbers for the draft genome sequence and the 16S rRNA gene sequence of Muricauda chongwuensis HICWT are WYET00000000 and MK920190, respectively.
Code availability
Not applicable.
Abbreviations
- BALOs:
-
Bdellovibrio-and-like organisms
- rRNA:
-
Ribosomal RNA
- MB:
-
Marine broth 2216E
- MA:
-
Marine agar 2216E
- MCCC:
-
Marine Culture Collection of China
- JCM:
-
Japan Collection of Microorganisms
- TEM:
-
Transmission electron microscope
- gANI:
-
Genome average nucleotide identity
- dDDH:
-
Digital DNA–DNA hybridization
- MP:
-
Maximum parsimony
- ML:
-
Maximum likelihood
- NJ:
-
Neighbor joining
- OMVs:
-
Outer membrane vesicles
References
Athalye M, Noble WC, Minnikin DE (1985) Analysis of cellular fatty acids by gas chromatography as a tool in the identification of medically important coryneform bacteria. J Appl Bacteriol 58:507–512
Bae SS, Kwon KK, Yang SH, Lee HS, Kim SJ, Lee JH (2007) Flagellimonas eckloniae gen. nov. sp. nov. a mesophilic marine bacterium of the family Flavobacteriaceae, isolated from the rhizosphere of Ecklonia kurome. Int J Syst Evol Microbiol 57:1050–1054
Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M, Kulikov AS, Lesin VM, Nikolenko SI, Pham S, Prjibelski AD, Pyshkin AV, Sirotkin AV, Vyahhi N, Tesler G, Alekseyev MA, Pevzner PA (2012) SPAdes: a new genome assembly algorithm and its applications to single cell sequencing. J Comput Biol 19:455–477
Bernardet JF, Nakagawa Y, Holmes B (2002) Proposed minimal standards for describing new taxa of the family Flavobacteriaceae and emended description of the family. Int J Syst Evol Microbiol 52:1049–1070
Bruns A, Rohde M, Berthe-Corti L (2001) Muricauda ruestringensis gen. nov., sp. nov., a facultatively anaerobic, appendaged bacterium from German North Sea intertidal sediment. Int J Syst Evol Microbiol 51:1997–2006
Chen Y, Hu Z, Wang H (2021) Muricauda amphidinii sp. nov., a novel marine bacterium isolated from the phycosphere of dinoflagellate Amphidinium carterae. Int J Syst Evol Microbiol. https://doi.org/10.1099/ijsem.0.004764
Collins MD, Costas M, Owen RJ (1984) Isoprenoid quinone composition of representatives of the genus Campylobacter. Arch Microbiol 137:168–170
Dang YR, Sun YY, Sun LL, Yuan XX, Li Y, Qin QL, Chen XL, Zhang YZ, Shi M, Zhang XY (2019) Muricauda nanhaiensis sp. nov., isolated from seawater of the South China Sea. Int J Syst Evol Microbiol 69:2089–2094
Delong EF (1992) Archaea in coastal marine environments. Proc Natl Acad Sci USA 89:5685–5689
Dong XZ, Cai MY (2001) Determinative manual for routine bacteriology. Scientific Press, Beijing (in Chinese)
Germida JJ, Casida LE (1983) Ensifer adhaerens predatory activity against other bacteria in soil, as monitored by indirect phage analysis. Appl Environ Microbiol 45:1380–1388
Guo LL, Wu D, Sun C, Cheng H, Xu XW, Wu M, Wu YH (2020) Muricauda maritima sp. nov., Muricauda aequoris sp. nov., and Muricauda oceanensis sp. nov., three marine bacteria isolated from seawater. Int J Syst Evol Microbiol 70:6240–6250
Hameed A, Arun AB, Ho HP, Chang CMJ, Rekha PD, Lee MR, Singh S, Young CC (2011) Supercritical carbon dioxide micronization of zeaxanthin from moderately thermophilic bacteria Muricauda lutaonensis CC-HSB-11T. J Agric Food Chem 59:4119–4124
Hwang CY, Kim MH, Bae GD, Zhang GI, Kim YH, Cho BC (2009) Muricauda olearia sp. nov., isolated from crude-oil-contaminated seawater, and emended description of the genus Muricauda. Int J Syst Evol Microbiol 59:1856–1861
Hyatt D, Chen GL, Locascio PF, Land ML, Larimer FW, Hauser LJ (2010) Prodigal: prokaryotic gene recognition and translation initiation site identification. BMC Bioinform 11:119
Jurkevitch E, Davidov Y (2006) Phylogenetic diversity and evolution of predatory prokaryotes. In: Jurkevitch E (ed) Predatory prokaryotes. Microbiology monographs, vol 4. Springer, Berlin
Kates M (1972) Techniques of lipidology: isolation, analysis and identification of lipids. In: Work TS, Work E (eds) Laboratory techniques in biochemistry & molecular biology, vol 3. Elsevier, Amsterdam, pp 151–155
Kim OS, Cho YJ, Lee K, Yoon SH, Kim M, Na H, Park SC, Jeon YS, Lee JH, Yi H, Won S, Chun J (2012) Introducing EzTaxon-e: a prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int J Syst Evol Microbiol 62:716–721
Kim M, Oh HS, Park SC, Chun J (2014) Towards a taxonomic coherence between average nucleotide identity and 16S rRNA gene sequence similarity for species demarcation of prokaryotes. Int J Syst Evol Microbiol 64:346–351
Kim D, Yoo Y, Khim JS, Yang D, Pathiraja D, Choi IG, Kim JJ (2020) Muricauda ochracea sp. nov. isolated from a tidal flat in the Republic of Korea. Int J Syst Evol Microbiol 70:4555–4561
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874
Liu L, Yu M, Zhou S, Fu T, Sun W, Wang L, Zhang XH (2020) Muricauda alvinocaridis sp. nov., isolated from shrimp gill from the Okinawa Trough. Int J Syst Evol Microbiol 70:1666–1671
McCauley EP, Haltli B, Kerr RG (2015) Description of Pseudobacteriovorax antillogorgiicola gen. nov. sp. nov. a bacterium isolated from the gorgonian octocoral Antillogorgia elisabethae, belonging to the family Pseudobacteriovoracaceae fam. nov. within the order Bdellovibrionales. Int J Syst Evol Microbiol 65:522–530
Meier-Kolthoff JP, Auch AF, Klenk HP, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform 14:60
Mu DS, Wang S, Liang QY, Du ZZ, Tian R, Ouyang Y, Wang XP, Zhou A, Gong Y, Chen GJ, Nostrand JV, Yang Y, Zhou J, Du ZJ (2020) Bradymonabacteria, a novel bacterial predator group with versatile survival strategies in saline environments. BMC Microb 8:126
Park JS (2019) Muricauda hymeniacidonis sp. nov., isolated from sponge of Hymeniacidon sinapium. Int J Syst Evol Microbiol 69:3800–3805
Pérez J, Moraleda-Muñoz A, Marcos-Torres FJ, Muñoz-Dorado J (2016) Bacterial predation: 75 years and counting! Environ Microbiol 18:766–779
Reichenbach H (1989) Order I. Cytophagales Leadbetter 1974. In: Staley JT, Bryant MP, Pfennig N, Holt JG (eds) Bergey’s manual of systematic bacteriology, vol 3. Williams & Wilkins, Baltimore, pp 2011–2013
Richter M, Rosselló-Móra R (2009) Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci USA 106:19126–19131
Sasser M (1990) Identification of bacteria by gas chromatography of cellular fatty acids, MIDI technical note 101. MIDI Inc., Newark
Schoeffield AJ, Williams HN (1990) Efficiencies of recovery of bdellovibrios from brackish-water environments by using various bacterial species as prey. Appl Environ Microbiol 56:230–236
Sorokin DY, Mosier D, Zorz JK, Dong X, Strous M (2020) Wenzhouxiangella strain AB-CW3, a proteolytic bacterium from hypersaline Soda Lakes that preys on cells of Gram-positive bacteria. Front Microbiol 11:597686
Svercel M, Saladin B, Moorsel SJ, Wolf S, Bagheri HC (2011) Antagonistic interactions between filamentous heterotrophs and the cyanobacterium Nostoc muscorum. BMC Res Notes 4:357
Wang ZJ, Liu QQ, Zhao LH, Du ZJ, Chen GJ (2015) Bradymonas sediminis gen. nov. sp. nov. isolated from coastal sediment, and description of Bradymonadaceae fam. nov. and Bradymonadales ord. nov. Int J Syst Evol Microbiol 65:1542–1549
Williams HN, Chen H (2020) Environmental regulation of the distribution and ecology of Bdellovibrio and like organisms. Front Microbiol 11:545070
Yang C, Li Y, Guo Q, Lai Q, Wei J, Zheng T, Tian Y (2013) Muricauda zhangzhouensis sp. nov., isolated from mangrove sediment. Int J Syst Evol Microbiol 63:2320–2325
Ye XS, Chen MX, Li HY, He XY, Zhao Y (2019) Halobacteriovorax vibrionivorans sp. nov., a novel prokaryotic predator isolated from coastal seawater of China. Int J Syst Evol Microbiol 69:3917–3923
Yoon BJ, Oh DC (2012) Spongiibacterium flavum gen. nov. sp nov. a member of the family Flavobacteriaceae isolated from the marine sponge Halichondria oshoro, and emended descriptions of the genera Croceitalea and Flagellimonas. Int J Syst Evol Microbiol 62:1158–1164
Yoon JH, Lee MH, Oh TK, Park YH (2005) Muricauda flavescens sp. nov. and Muricauda aquimarina sp. nov., isolated from a salt lake near Hwajinpo Beach of the East Sea in Korea, and emended description of the genus Muricauda. Int J Syst Evol Microbiol 55:1015–1019
Yoon JH, Kang SJ, Jung YT, Oh TK (2008) Muricauda lutimaris sp. nov., isolated from a tidal flat of the Yellow Sea. Int J Syst Evol Microbiol 58:1603–1607
Yoon SH, Ha SM, Lim J, Kwon S, Chun J (2017) A large-scale evaluation of algorithms to calculate average nucleotide identity. Anton Leeuw Int J G 110:1281–1286
Zhang Y, Gao Y, Pei J, Cao J, Xie Z, Liu R, Wang L, Wei Y, Fang J (2020) Muricauda hadalis sp. nov., a novel piezophile isolated from hadopelagic water of the Mariana Trench and reclassification of Muricauda antarctica as a later heterotypic synonym of Muricauda taeanensis. Int J Syst Evol Microbiol 70:4315–4320
Zhu S, Xue Z, Huang Y, Chen X, Ren N, Chen T, Chen Y, Yang J, Chen J (2021) Muricauda sediminis sp. nov., isolated from western Pacific Ocean sediment. Int J Syst Evol Microbiol. https://doi.org/10.1099/ijsem.0.004757
Funding
This work was supported by the National Natural Science Foundation of China (Grant no. 41506179), National Programme on Global Change and Air-Sea Interaction (Grant no. GASI-03-01-03-01), Natural Science Foundation of Fujian Province (Grant no. 2015J01613), and Fujian Provincial Key Laboratory of Marine Ecological Conservation and Restoration (Grant no. EPR2020002).
Author information
Authors and Affiliations
Contributions
M-XC and H-YL conceived the project. M-XC and X-YH experimented and analyzed the data. M-XC wrote the manuscript. All authors have revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, MX., He, XY. & Li, HY. Muricauda chongwuensis sp. nov., isolated from coastal seawater of China. Arch Microbiol 203, 6245–6252 (2021). https://doi.org/10.1007/s00203-021-02591-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-021-02591-1